Account for the formation of the product and for the cis stereochemistry of its ring junc- tion. (The function of silver carbonate is to enhance the rate of reaction.) COOME COOME Pd(OAc)g, PPh, Ag,COg, CH,CN H 86%
Account for the formation of the product and for the cis stereochemistry of its ring junc- tion. (The function of silver carbonate is to enhance the rate of reaction.) COOME COOME Pd(OAc)g, PPh, Ag,COg, CH,CN H 86%
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter24: Catalytic Carbon-carbon Bond Formation
Section: Chapter Questions
Problem 24.13P
Related questions
Question
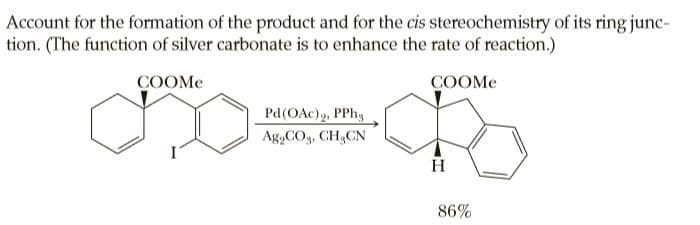
Transcribed Image Text:Account for the formation of the product and for the cis stereochemistry of its ring junc-
tion. (The function of silver carbonate is to enhance the rate of reaction.)
COOME
COOME
Pd(OAc)g, PPh,
Ag,COg, CH,CN
H
86%
Expert Solution
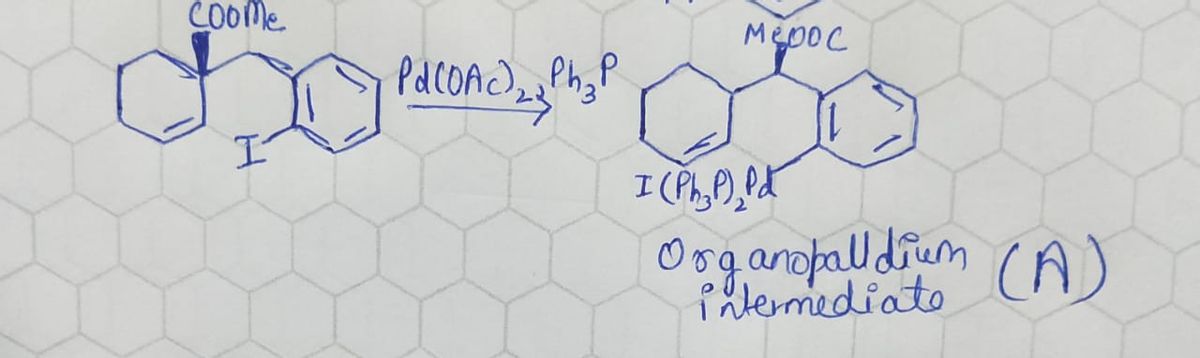
Step 1
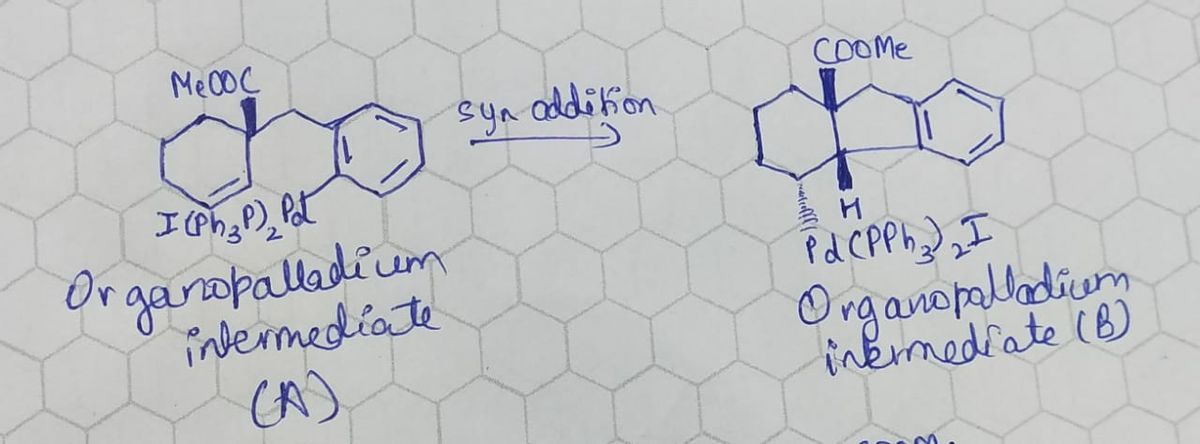
Step 2
Step by step
Solved in 3 steps with 3 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning