Q: Which compound is not a possible product in the reaction below ( F2 is in excess F2 and UV light the…
A: Given here the chemical reaction and reaction condition and we are asked which compound is not a…
Q: For the reaction below, draw the major SN1 organic product or products. There is not a shortcut for…
A: A question based on organic chemistry, which is to be accomplished.
Q: 1) Write a detailed, stepwise mechanism for the following reaction. In your mechanism, provide three…
A:
Q: 4 pts Provide the structure of the major organic product. in the following reaction sequence. 1.…
A: Answer:- This question is answered by using the simple concept of chemical reaction of ketone with…
Q: Chemistry Please answer A and B of the Following question and provide the Name for B too. Predict…
A: The solutions are given below -
Q: The reaction below produces two neutral organic products that are formed in a 1:1 ratio. Provide the…
A: Nucleophilic substitution reactions are those reactions in which nucleophile attacks the alkyl…
Q: The completion of the synthesis of the second compound is provided below. B Br Using the list below,…
A:
Q: Analyze the reaction below, and then provide the reagents necessary to carry out the conversion. Be…
A:
Q: Complete the statement below by writing the number (from the table below) that represents the set of…
A: The starting material is toluene which is first converted to an intermediate, Z before being…
Q: Part 3: Strategy 5. Enantioselective epoxidations are useful reactions when used in multi-step…
A:
Q: Provide the structure of the major organic product in the reaction below. CI NH2 Draw the molecule…
A: Given reaction is :
Q: II. Give a multi-step synthesis proposal to start with the molecule on left and use any reagents…
A:
Q: Provide the major organic product of the following reaction sequence. 1. NAOCH2CH3 .Co,CH2CH3 2.…
A:
Q: (CH3),CHCI AlCl3 CH2 CH3
A: Given,
Q: (a) Illustrate the following name reactions :(i) Reimer-Tiemann Reaction (ii) Williamson…
A: The 2 named reactions, reamer-Tiemann reaction is the conversion of phenol to salicylaldehyde. And…
Q: 1. HNO3, H,SO, 2. Fe, HCI 3. NANO,, HCI 4. HBF, 5. KMN04, A CH(CHS2
A:
Q: 2. (4) Show the synthetic steps necessary to carry out the conversion below. You may use any organic…
A: In this synthesis process, first we will acylate benzene, then react it with grignatd reagent…
Q: Explain each of the following true statements briefly: 1. Hexane (C6H14, a hydrocarbon) is not a…
A: Nucleophile is a rich electron species that contains a lone pair of electrons and can donate to a…
Q: Write a detailed, stepwise mechanism for the following reaction. CH3 Cl2 -CH2CI + HC1
A: The structure in which the electrons are delocalized on the three or more atoms which cannot be…
Q: Part A Provide the major organic product of the reaction shown below. 1. CH,MgBr 2. H,0", A Draw the…
A: Nitriles (RC≡N) react with Grignard reagents to produce ketones.
Q: 1. H2CrO4 2. SOCI2, pyr. 3. (CH,CH2)2NH (xs) HO-
A: The reaction given is,
Q: 3-) Show the mechanism by using curved arrows for the reaction between following lactone and…
A: Grignard reagent first attacks the C=O of the cyclic lactone through nucleophilic addition reaction…
Q: Draw the organic products of the reaction shown. Draw hydrogens on oxygen and nitrogen where…
A: The products of the given reaction is
Q: write mechanism step by step for the reaction ?
A: The given reaction is as follows,
Q: Problems 1 and 3 for part a (3-methyl-3-pentanol with phosphoric acid)
A: The alcohol taken in part A) is 3-Methyl-3-pentanol. The alkenes formed during the experiment are as…
Q: Show the synthesis of 4-iodotoluene starting from the starting material benzene. The usage of other…
A:
Q: Identify the starting material in attached reaction.
A: This is reaction is an example of an ozonolysis reaction. In this reaction, the double bond in the…
Q: Suggest a synthesis for the molecule shown below. Only starting materials containing six (6) carbons…
A: We can get the final product by taking terminal alkyne as a starting material and we can expect…
Q: Draw the structure(s) of the major organic product(s) of the following reaction. „NH, NANO aqueous…
A: Aromatic amine reacts with sodium nitrite and hydrochloric acid at 0oC temperature to form a…
Q: [1] LDA [2] CH3I
A:
Q: Provide the missing structures of products A and B for the following reaction sequence. NH2 1)…
A: → Aniline Aniline is primary amine
Q: H* OH
A: This is an Esterification Reaction. In this, acid react with alcohols to form ester in presence of…
Q: ОН O: H3C- H3C- ОН HO ОН
A: The products in the given reaction are obtained by the ozonolysis of given reactant, but-3-ene-1-oic…
Q: Write out the detailed step-wise mechanism for this esterification. 3-methylbutyl propionate using…
A: Given reaction scheme: We have to write the step-wise mechanism of this esterification reaction.
Q: 2. Write the structural formula of the major organic product formed in each of the following…
A: a) The product formed from the given reaction is 2-methyl pentan-3-ol.
Q: Provide a plausible arrow pushing mechanism for the reaction below. 1 eq. H20 + МеОн HO, OMe cat.…
A:
Q: Provide the reagent that is needed to perform the transformation shown below. (Use the Reagent…
A: Reagent:-Reagent is a substance or mixture for use in chemical analysis or other reactions.
Q: .From the two synthetic routes shown below which one would be the best route for the desired…
A: In this question, we have to find out the correct answer of given problem by the help of the…
Q: Provide a synthesis for the following reaction. If more than one step is required be sure to number…
A:
Q: 4. Provide a detailed, stepwise mechanism for the reaction below. (CH3)2CHCH2CH2CH21 + CN- →…
A: Primary alkyl halide reacts with cyanide ion to form alkyl cyanide. The reaction follows the SN2…
Q: Question: Which of the following procedures would be best for achieving the following reaction? *…
A: Since, you have asked multiple questions, we will solve the first question for you. Question-1 : In…
Q: - Provide the structure of the major organic product for the reaction sequence shown below. 1.…
A: It is a 3 step reaction as shown in the following step.
Q: Tunicates are marine animals that are called "sea squirts" because when they are taken out of water,…
A:
Q: H OCH2CH3 5) Provide the structure of the major organic product in the reaction shown below. 1. 2…
A:
Q: 5. Provide a complete curved arrow mechanism for the most straightforward conversion of 5 and 6 to…
A:
Q: 1.6 Complete the following reactions by providing the missing starting material, reagent(s),…
A:
Q: 1. The following diagram shows the partial synthesis of an important antihistamine drug currently…
A: To write the oxidation state of *C in all the three structures.
Q: of CH3 CH3 1. LDA/THF, -78 °C CH3 H3C. -CH3 + 2. CH3-Br I or II
A:
Q: Provide a synthesis for the following reaction. If more than one step is required be sure to number…
A:
Q: What is/are the best reagents (i.e., chemicals for the box below) to carry out the following…
A:
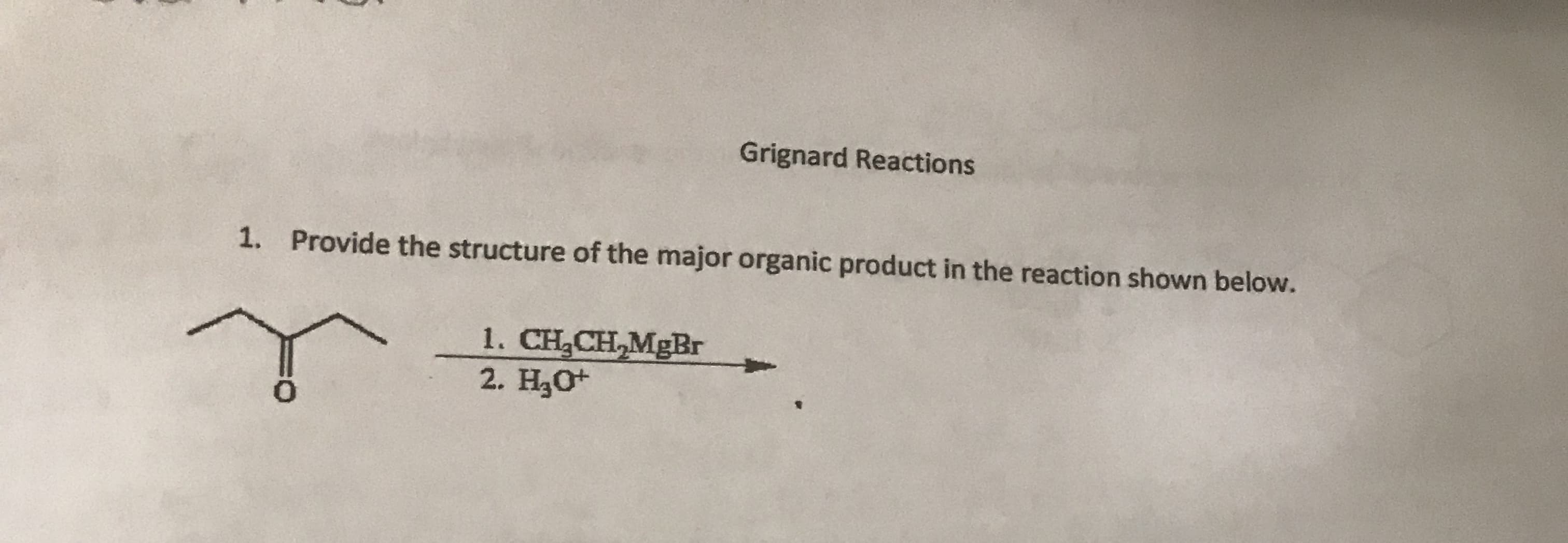
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

- I have this task in organic chemistry (book: Brown's introduction to organic chemisty, global edition). Task 10:42. In (a) I have to tell what the funcion of K2CO3 is in step 1. Is it that CO32- take the hydrogen atom in 1-napthol? Will it then be a SN2 mechanism? In (b) I have to name the amine used in step 2 to form Propanolol. But I can't really find out how to come up with an amine that will make that reaction. Here are two pictures of the task:Which compound is not a possible product in the reaction below ( F2 is in excess F2and UV light the catalyst).CHA + F2 -->O CFAO CHFaO CHF-O CH2F2O CHaFTunicates are marine animals that are called "sea squirts" because when they are taken out of water, they tend to contract and expel seawater. Lepadiformine is a cytotoxic agent (toxic to cells) isolated from a marine tunicate. During a recent synthesis of lepadiformine, the investigators observed the formation of an interesting by-product (3) while treating diol 1 with a reagent similar in function to PBr3 (J. Org. Chem. 2012, 77, 3390–3400):
- Please show the steps to the organic synthesis reactions below:Which reagent is required to complete the reaction in Figure 11? [Generate a SMILES notation: https://jsme-editor.github.io/dist/JSME_test.html. Copy the notation (CTRL+C) and paste it as the answer to this question (CTRL+V).] * Your answerFor 20.7 the solutions for part c and d dont seem right. Shouldn't reacting with 1 eq of H2 just take away the C=C bond and not the C=O bond. (H2 reduces C=C selectively to form a ketone according to page 738. Also the both the C=C and C=O bond should be reduced if excess H2 is used
- I recovered 3 mL of water, how many grams of water was produced in the reaction please show how? Cyclohexanol can attack the carbocation intermediate formed during the synthesis of cyclohexene. Draw this side product with the molecular formula C12H22O.C3H6 + H2O + H2SO4 --> C3H8O + H2SO4 what is the catalyst in the reaction above?Reaction with Strong Acids Table 9.8 Observations on the reaction of aniline with strong acids. Sample Observations +/- Aniline + H2SO4 Aniline + HCl Explain, in 1 – 3 sentences, the difference in the reaction of aniline with H2SO4 and HCl. ___________________________________________________________________________ ___________________________________________________________________________ ___________________________________________________________________________ Reactions: Aniline + H2SO4 Aniline + HCl
- In the reaction from compound 10 to compound 11, why the C=C bond is retained and is not hydrogenated? f) LiAlH4, Et2O, 08C; g) Ac2O, py, DMAP, CH2Cl2, RT, 77% over 2 steps;10.14 Complete the following reactions: Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be. Encode (not hand-written)!What is the major organic product in the reaction below?

