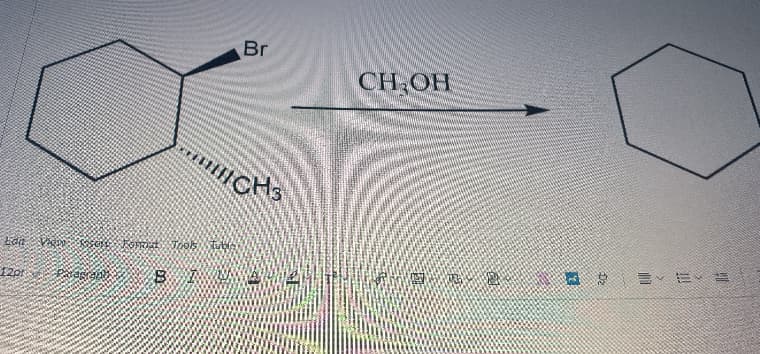
Q: Write the major products A- P for each of the following reactions.
A: In this question, we have to find out the correct answer of given problem by the help of the con...
Q: need answer
A:
Q: When glutamic acid dissolve in aqueous sodium hydroxide, monosodium glutamate (MSG) is formed, which...
A: 1) 2) Firstly glutamic acid changes into its ionic form (glutamate) and then finally addition of ...
Q: need answers
A: In given question chany the temperature, mass and specific heat required
Q: Br KMNO/ OH" Zn b) 2) Zn/ CH;COOH Br
A:
Q: If 32.0 g of NaOH is added to 0.550 L of 1.00 M Ni(NO,),, how many grams of Ni(OH), will be formed i...
A:
Q: In general, ionization energy increases down a group because atoms have more protons. O True False
A: Ionization energy refers to the amount of energy needed to remove an electron from an atom. Ionizati...
Q: Direct currents are uniform and have uniform voltage. O True O False
A: True
Q: 7. Compound A, C„H160 is optically active and gives a negative results with Iodoform tust and forms ...
A:
Q: What mass of NH3 is needed to completely react with 0.600 moles of O2 (g) based on the reaction belo...
A:
Q: How many grams are needed to make a solution that is 5 M HCl when you have 500 milliliters of water?
A: Molarity (M) :- The number of moles of solute dissolved in one litre of solution is called molarity...
Q: 4. Provide reagents necessary to transform the given starting materials to the indicated products. I...
A: The conversion of the substrate to the desired product is done by different reagents. If we use 1 e...
Q: How many moles of H2O2 are produced in the production of hydrogen peroxide when 13 grams of oxygen a...
A: The given reaction of hydrogen peroxide production is , 2H2O +O2 →2H2O2 Weight of oxygen is us...
Q: 3. What would the pH at the half-equivalence point be for the first ionization of the triprotic acid...
A: • The Handerson Hasselbalch equation for an acid buffer is- pH = pKa + log [salt]/[acid...
Q: (see image)
A: The nitric oxide reacts with the oxygen to produce nitrogen dioxide is a heterogeneous reaction. The...
Q: At the end of the school year a high school chemistry teacher asks one of his/her students to stay a...
A: The given unknown compound is rectangular in shape and the compound should be identified by density ...
Q: 1) Calculate the pH of a solution prepared by dissolving 1.15 g of sodium acetate, CH3COONa, in 71.0...
A: Find the moles of Sodium Acetate find the moles of Acetic acid then find the ph using henderson eq...
Q: Question is attached.
A: Ksp expression is written as products of concentration of ions raise to the power the coefficients .
Q: please show all work Please answer very soon will give rating surely
A: Organic compounds are compounds that are mainly composed C and H atoms. The branch of chemistry that...
Q: Classify the functional groups in the following steroid (choose all that apply).CHOOSE ALL THAT APPL...
A: This this question, we will see all the functional group in the given Compound. How we can identify...
Q: Classify the functional groups in the following steroid (choose all that apply).CHOOSE ALL THAT APPL...
A: If OH group is attached to the carbon atom which is directly attached to 3 carbons then it is known...
Q: 2) Write the major products A- P for each of the following reactions. HBr A B CH3CH,0'K* CH;CH,OH t-...
A: The product A is formed as a result of SN2 reaction (more favored) of the given secondary alkyl hali...
Q: A gas with a volume of 3.50L at a pressure of 200kPa is allowed to expand to 14.0L. If the temperatu...
A: V1 = 3.50L P1 = 200kPa V2 = 14.0L P2 = ? At constant temperature the equation f...
Q: Pick all of the combinations of atomic quantum numbers that cannot occur: (Can be more than 1 answer...
A: n=0, l=0, m=0 n=2, l=2, m=0
Q: Which would NOT protect iron from corrosion? O Covering with paint or oil Attaching to Ni Electropla...
A: Corrosion is defined as the process by which refined metals are transformed into more durable compou...
Q: In the sketch of the structure of CH2B12 label all bonds. Drag the appropriate labels to their respe...
A: All the bonds formed in CH2Br2 are sigma bonds. The hybridisation of C is sp3 Both, the C-Br bonds a...
Q: please give the correct name and structure of 1-bromo-6-chloro-5-phenol and 1-octanone. Give their 2...
A: Isomers: Two molecules have the same molecular weight but different in their spatial arrangement or ...
Q: In which one of the following reactions does sulfur disproportionate? O A. H2S2 - H2S + S B. H2S(aq)...
A: In a disproportionation reaction, the same element undergoes oxidation as well as reduction Example ...
Q: Substance Reaction to flame Phase at room temperature Reaction with water Hydrogen Colorless Gas Non...
A: Strontium chloride
Q: Match each phrase with its correct term. Record your responses onto the Google Form. 13. -log (H,O']...
A:
Q: In this equation H2O +HF- H3O+ +F-1 H20 is a base and HF is its conjugated acid HF is an acid and F-...
A: THe answer to the following question is
Q: In a flask containing granules of aluminium, we added a solution of diluted hydrochloric acid, that ...
A: The reactivity series follows the order :Al>Zn>Fe>Pb>H>CuThus, aluminium being more r...
Q: 2. Is it possible to obtain diazonium chloride from secondáry and tertiary amine? Explain your answe...
A: the answer is no.
Q: 2
A: Electron geometry is the arrangement of electron groups. Given molecule is IF3.
Q: Classify the following lipid (choose all that apply).CHOOSE ALL THAT APPLY a. triglyceride b. pol...
A: There are different types of lipids - Fats, oils, wxes, steroids, triglycerides, phospholipids etc....
Q: 2) Write the major products A- P for each of the following reactions. HBr A B CH;CH,O K* а) CH;CH,OH...
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you. ...
Q: Identify what is undergoing oxidation-reduction in the following reactions (some of the following re...
A:
Q: What process is used to remove unwanted elements from freshly mined iron ore?
A: in metallurgy or during the extraction of metal beneficiation process is used. Beneficiation is a pr...
Q: 2.3
A: Metal:Metals are situated on the left-hand side of the periodic table. The metals are electropositiv...
Q: What is the mole ratio of oxygen to carbon dioxide in the combustion reaction for propane? C3H8 + 5 ...
A: The mole ratio of two elements is the ratio of the moles of the two elements in the balanced chemica...
Q: give UV-vis spectrum showing (n to pi*) transition in addition to the main one and HNMR spectrum of ...
A: The desired product that formed in the reaction is shown below.
Q: Write the major products A- P for each of the following reactions.
A:
Q: 1. Using the curved-arrow formalism, provide a mechanism that accounts for the following transformat...
A:
Q: What is the major product obtained from the following reaction? Heat 3 O 3 O 2 O 1 O 4
A: The given compound is a β keto acid. β keto acids undergo decarboxylation reaction in presence of he...
Q: What is the hybridization of the nitrogen atom in this structure? N. sp2 O sp sp3 O sp³d
A: In Nitrate ion ( NO3-), there are total 24 valence electrons. The central metal Nitrogen has 5 valen...
Q: explain what happens in the electron transport chain and the formation of ATP
A: The electron transport chain is basically a collection of series of steps. When the electrons trave...
Q: A student made 10.0 mL of a 0.750 M solution in the following way. First, the student took some 16.0...
A:
Q: One vessel contains 3.00 mol of an ideal gas at 100 kPa. Another vessel contains 4.00 mol of the sam...
A: Given: For container 1st: No of moles, n1 = 3 mol Pressure, P1 =100kPa For container 2st: No of mole...
Q: Task 3: In the following problems, you will need to identify the real metals in the electrochemical ...
A: In an electrochemical cell, the reactions take place at the two electrode placed in the cell that re...
Q: What is the enthalpy change when 8.20 moles ammonium chloride form in the following equation? N...
A: The given balanced chemical equation is as follows --- N2 (g) + 4 H2 (g) + Cl2 (g) --> 2 NH4C...
Step by step
Solved in 2 steps with 1 images

- El poder de rotación específico de la sacarosa a 20 °C es [alfa]D20 = +66,53 grados/(dm g mL-1). ¿Cuál es su poder de rotación molar, expresado en grados/(dm mol L-1)? (Peso molecular de la sacarosa: 342,3). 1). 5.14 2). 22.77 3). 194.4 4). 22773What does a refractometer reading of 2 mean when distilling ethanol?pls answer the next 3 pls FAB ion mass detection quadrupole

