Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
ChapterL4: Proton (1h) Nmr Spectroscopy
Section: Chapter Questions
Problem 18CTQ
Related questions
Question
Add the unshared (lone) electron pairs to the following structure.
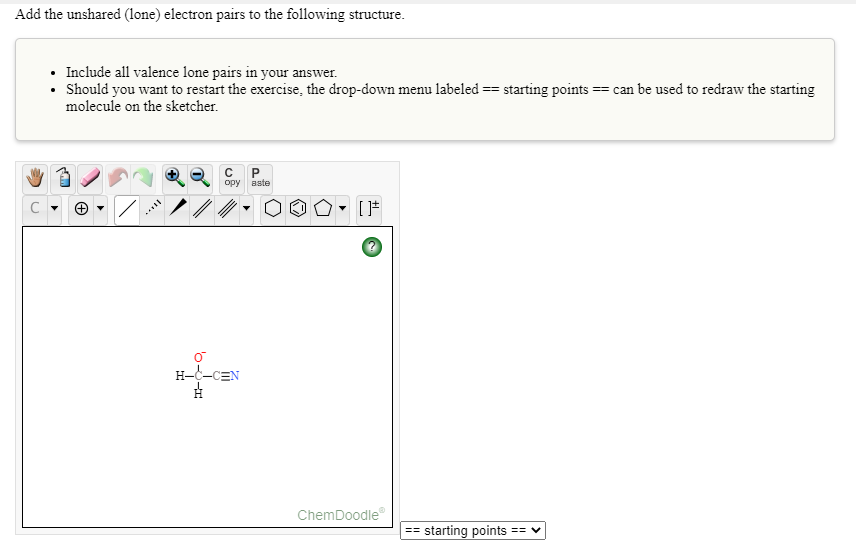
Transcribed Image Text:Add the unshared (lone) electron pairs to the following structure.
• Include all valence lone pairs in your answer.
• Should you want to restart the exercise, the drop-down menu labeled == starting points == can be used to redraw the starting
molecule on the sketcher.
P.
opy
aste
H--CEN
ChemDoodle
== starting points
==
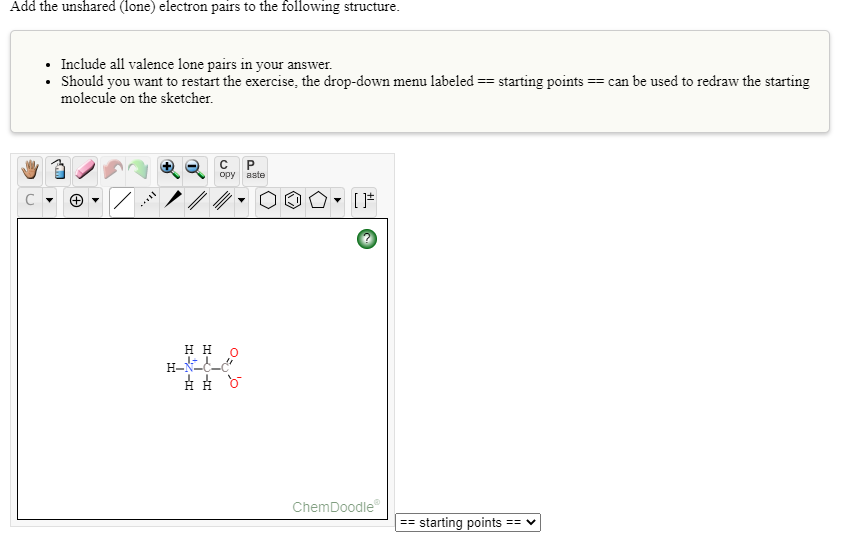
Transcribed Image Text:Add the unshared (lone) electron pairs to the following structure.
• Include all valence lone pairs in your answer.
• Should you want to restart the exercise, the drop-down menu labeled == starting points == can be used to redraw the starting
molecule on the sketcher.
P
ору
aste
нн
ChemDoodle
== starting points == v
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning