A>D II I IV A B Progress of Reaction Progress of Reaction Progress of Reaction Progress of Reaction (a) Which diagram if any represents the slower endergonic reaction? (b) Which diagram if any represents the faster endergonic reaction? (c) Which diagram if any represents the slower exergonic reaction? (d) Which diagram if any represents a faster exergonic reaction? (e) Which reaction has the lowest activation energy? (f) Based on the energy diagrams, which reaction leads to the least stable product? (g) Based on the energy diagrams, which reactant is thermodynamically least stable? (h) Based on the energy diagrams, which product is themodynamically most stable? Free Energy
A>D II I IV A B Progress of Reaction Progress of Reaction Progress of Reaction Progress of Reaction (a) Which diagram if any represents the slower endergonic reaction? (b) Which diagram if any represents the faster endergonic reaction? (c) Which diagram if any represents the slower exergonic reaction? (d) Which diagram if any represents a faster exergonic reaction? (e) Which reaction has the lowest activation energy? (f) Based on the energy diagrams, which reaction leads to the least stable product? (g) Based on the energy diagrams, which reactant is thermodynamically least stable? (h) Based on the energy diagrams, which product is themodynamically most stable? Free Energy
Introduction to General, Organic and Biochemistry
11th Edition
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Chapter7: Reaction Rates And Chemical Equilibrium
Section: Chapter Questions
Problem 7.51P
Related questions
Question
Consider the energy diagrams (I, II, and III) for the three different reactions shown below when answering questions 18(a) – (f). Write NA if an applicable answer is not given.
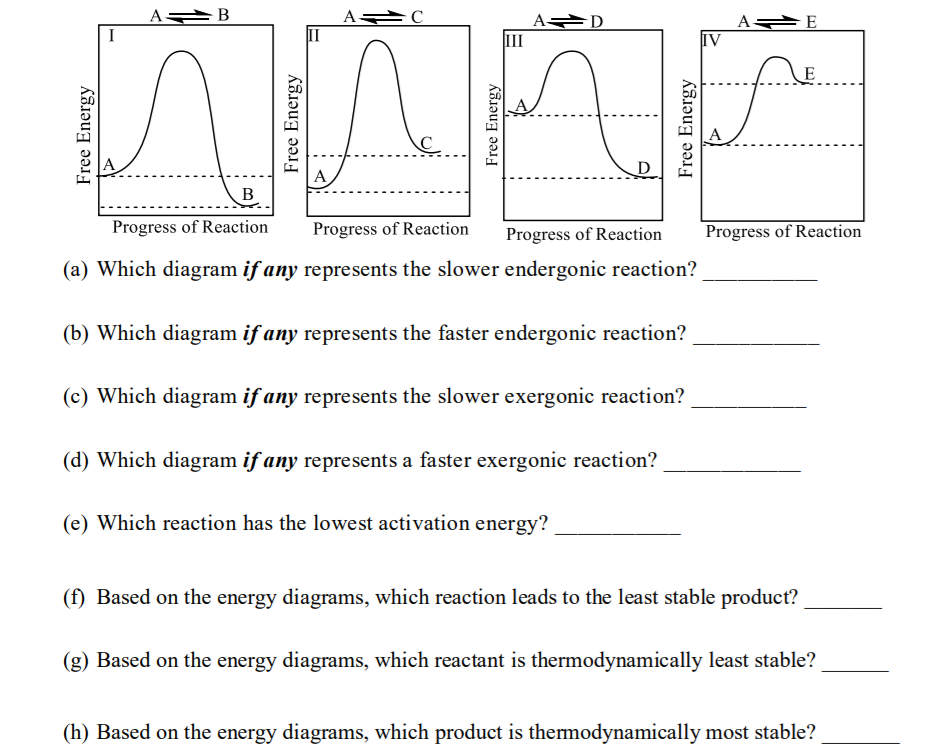
Transcribed Image Text:A B
A D
III
II
IV
E
C.
A
B
Progress of Reaction
Progress of Reaction
Progress of Reaction
Progress of Reaction
(a) Which diagram if any represents the slower endergonic reaction?
(b) Which diagram if any represents the faster endergonic reaction?
(c) Which diagram if any represents the slower exergonic reaction?
(d) Which diagram if any represents a faster exergonic reaction?
(e) Which reaction has the lowest activation energy?
(f) Based on the energy diagrams, which reaction leads to the least stable product?
(g) Based on the energy diagrams, which reactant is thermodynamically least stable?
(h) Based on the energy diagrams, which product is thermodynamically most stable?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax