All are properties of ideal solvent for recrystallization except: Has a boiling point below the sample's melting point Does not react with the target compound Immiscible with the impurity Dissolves the sample when hot Previous
All are properties of ideal solvent for recrystallization except: Has a boiling point below the sample's melting point Does not react with the target compound Immiscible with the impurity Dissolves the sample when hot Previous
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.13QAP
Related questions
Question
1 & 2
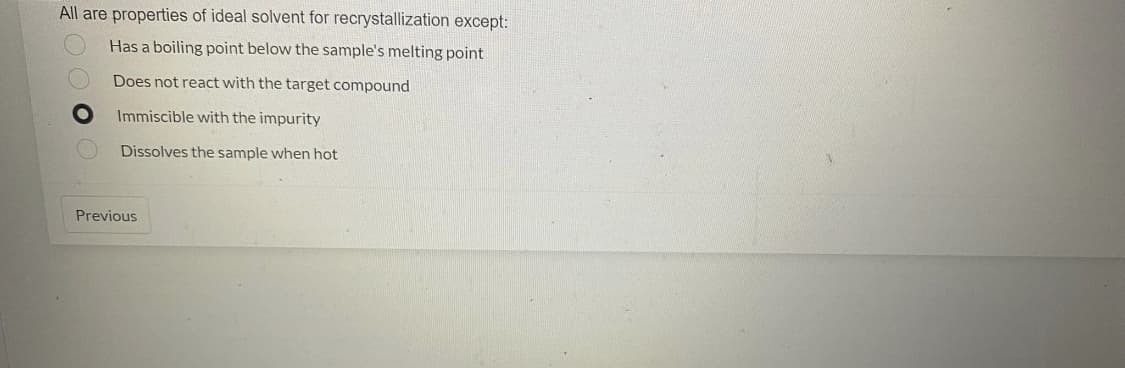
Transcribed Image Text:All are properties of ideal solvent for recrystallization except:
Has a boiling point below the sample's melting point
Does not react with the target compound
Immiscible with the impurity
Dissolves the sample when hot
Previous

Transcribed Image Text:Student A is trying to purify a sample stained blue by impurities. Which of the following should they use to remove the color?
Activated carbon
Gram decolorizer
Sodium hypochlorite
Acetone
0000
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

