Q: (a) The amount of work done on the water. = 35x -9 Force
A: Work done on water = change in potential energy of weight
Q: What is the total vapor pressure of a solution containing 35.8 g benzene (C6H6, molar mass = 78.11…
A: Given-> Weight of benzene = 35.8 gm Weight of toluene = 56.7 gm T = 25°C = 25 + 273 = 298 K…
Q: What type of chromatogtaphy (RPLC, NPLC, HILIC, SEC, IEX) could be used for analyzing monosodium…
A: The answer is attached below-
Q: A mixture containing only AgCl (FW 143.32) and AgBr (FW 187.77) weighs 2.000g. It is quantitatively…
A:
Q: 2. Write the name(s) of the product(s) for the following reactions. If no reaction can occur, write…
A: Here we are required to find the product of the reaction.
Q: In which type of point defect, positive and negative ions are missing from a crystal? Vacancy defect…
A:
Q: Calculate the number of unsaturations in: a. C8H12O3 b. C14H22BRNO C. C10H22N₂O d. C16H32Br2O3 e.…
A:
Q: 1.963-g sample of an alloy is dissolved in HNO3 and diluted to volume in a 100-mL volumetric flask.…
A:
Q: A 6.63 x 10-4 mol sample of CO₂ takes 615 seconds to effuse through a small hole. Under the same…
A:
Q: Determine whether an example is spontaneous process (SP) or a non-spontaneous process (NSP). 6. ball…
A: In this question we have given some process. We have to classify them as spontaneous or non…
Q: A chemist prepared a reagent blank and 6 Fe standards with the following concentrations: 20, 30, 40,…
A:
Q: Why can’t we use a simple distillation for the solution containing ethyl acetate and butyl acetate?
A: Ethyl acetate, CH3COOC2H5 is a simple ester, with boiling point of 77.1°C while butyl Acetate has…
Q: Given the absolute entropy values, calculate the standard entropy changes in the following reaction.…
A:
Q: c) Jack and Helen wish to sketch a weak acid strong base titration curve. Before they can do so,…
A:
Q: A student has synthesised propyl acetate and collected spectroscopic evidence for a successful…
A:
Q: 10. Give the structure corresponding to each name. a. 7,7-dimethyl-4-octanol b.…
A:
Q: Draw and name the structures of the isomeric 5-carbon saturated alcohols. Stipulate their reactions…
A: Given statement is : Draw and name the structures of the isomeric 5-carbon saturated alcohols.…
Q: 1. Determine if the corresponding pairs are conjugates: a. HCI & CI- b. CH3NH₂ & CH3NH3* c. H3PO4 &…
A: A question based on equilibrium concept that is to be accomplished.
Q: Hd 10.0 mL of a weak monoprotic acid is titrated with 0.100 M NaOH and the pH is measured. The…
A:
Q: The benzene radical anion C₂H, has a g-value of 2.0025. At what field would you search for resonance…
A:
Q: Which of the following will show a Tyndall Effect? Select the correct response: Aqueous solution of…
A: Tyndall effect is an optical property of a colloidal solution. When a beam of light passed through a…
Q: How do pyrethrins insecticides produce in the industries in details with all the chemical reactions…
A: The pyrethrins are a class of terpenoids a class of organic compounds normally derived from…
Q: theoretical yeild of this Chemiluminescent Reaction? The reaction uses 4ml of a 0.6M phenol…
A: Chemiluminescence is the emission of light from a chemical reaction.
Q: A 3.00 mol sample of an ideal diatomic gas expands adiabatically from a volume of 0.1610 m³ to 0.719…
A:
Q: To what Celsius temperature must 580 ml of oxygen at 17°C be raised to increase its volume to 700.0…
A:
Q: #55. Distinguish between isotonic and isometric contractions.??
A: There are two types of muscle contractions - 1- isotonic muscle contraction 2- isometric muscle…
Q: Balance the equation and indicate what type of chemical reaction is shown in each letter: a.…
A:
Q: 3. A mixture containing only AgCl (FW 143.32) and AgBr (FW 187.77) weighs 2.000g. It is…
A: Given that - Weight of a mixture containing AgCl + AgBr = 2.000 g Weight of silver obtained after…
Q: (b) Draw a more accurate representation of one unit of C,H,O, dissolved in water.
A:
Q: Which of the following is true of reduction reaction? It is the removal of electrons from an atom…
A: Since you have posted more than one question, we can solve only first question for you. If you want…
Q: 1. Show the arrow pushing in predicting the product (sigmatropic rxns) H3C 1.1 1.2 H3C- D A [3,3] A…
A:
Q: How would you make 1L of 0.5M total strength phosphate buffer (pKas are 2.1, 7.2, 12.7) at pH = 8…
A:
Q: Given reaction rate date for: Trial اجرای اب 1 2 3 2NO(g) + Br₂(g) → 2NOBr(g) [Br2] (M) 0.020 0.050…
A: The order of the reaction can be found by taking the concentration of one reactant constant at one…
Q: What is the milliequivalents of 0.980 g of Na2CO3 (MW=105.99)?
A: Given the milliequivalents of 0.980 g of Na2CO3 molar mass of Na2CO3, MW=105.99 g
Q: 7. Draw any lone pairs associated with the nitrogen atoms in the following structure: X b 겨 NH,
A: ->Nitrogen has 5 valence electrons but it can form tree bond without any charge to complete…
Q: Consider the phase diagram of substance J below: 4500 J(s) J(1) 760 82 53 0.1 12 93 200 Temperature…
A:
Q: Determine whether an example is spontaneous process (SP) or a non-spontaneous process (NSP). 1.…
A: Determination of process either spontaneous or non spontaneous with explanation.
Q: . In a 2.000-Liter vessel at 226 °C, a mixture of 0.2000 mol of CO2, 0.1000 mol of H₂ and 0.1600 mol…
A: 1. The initial pressure PV = nRT P = (nRT)/V Pco2 =(nco2* R* T)/ V Pco2 = (0.2* 0.0821* 500)/ 2…
Q: A mixture containing only AgCl (FW 143.32) and AgBr (FW 187.77) 2.000g. It is quantitatively reduced…
A:
Q: What is the percentage of Na2CO3 in the mix?
A:
Q: Carla and her friends were tasked with determining the heat of reaction for the neutralization of…
A: Given: Concentration of NaOH = 1.00 M Volume of NaOH = 2.00 mL Concentration of HX = 1.00 M Volume…
Q: Choose the word from the word bank below that best matches each statement. Constant Equilibrium…
A: Please find the below answer. Please rate if it is helpful for you. thank you.
Q: Identify the endothermic process(es) below. You may choose one or more processes. If there are more…
A:
Q: In Fourier's law, the conduction heat flux is: Constant A scalar A vector A tensor A cross product
A:
Q: Carla and her friends were tasked with determining the heat of reaction for the neutralization of…
A: Given: Concentration of NaOH = 1.00 M Volume of NaOH = 2.00 mL Concentration of HX = 1.00 M Volume…
Q: 27. SYNTHESIS! Propose a multistep synthesis for the following transformation. Use the starting…
A: Here we are required to synthesize the given compound.
Q: Molecular Formula Total # Valence Electrons Lewis Structure Bond Electron Angles Geometry Molecular…
A:
Q: H₂C H₂N Choices: OH H₂C CH3 HO. CH3 Enantiomers Geometric Isomers CH 3 CH 3 NH₂ Skeletal Isomers…
A: Given compounds are : Match the items = ?
Q: A chemist designs a galvanic cell that uses these two half-reactions: half-reaction 210₂(aq)+12…
A: First we will choose cathode and anode, then write half cell reactions at both. Write overall…
Q: A reactant in a certain first-order reaction is 56 % consumed in 63 s. What is the half-life for…
A: In this question, we will determine the half-life for the first order reaction. You can see the…
Complete the following reaction
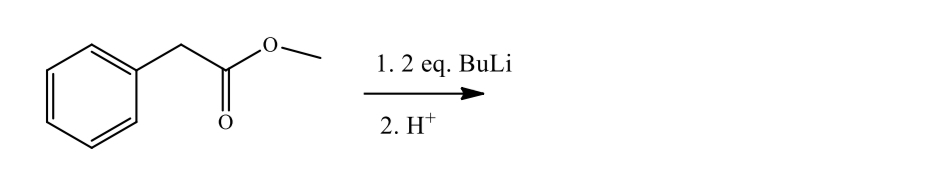
Step by step
Solved in 3 steps with 2 images

- Suppose you want to prepare a solution of 2.50L of 1.00M HCL solution from concentrated 12.0 M HCl (aq.) How many liters of concentrated acid is needed and how many liters of water is needed?[Dimensional Analysis is appreciated!]3 q101 Please put solutions thanksA student determined the average molarity of their sample of acetic acid to be 0.608 M. Calculate the (w/w)% of the solution. Assume the density of this solution is that of pure water, dH2O = 1000. g/L. Enter a number without the '%' symbol.
- 2. PLease don't provide handwritten solution ...a) Calculate [H3O+] of the following polyprotic acid solution: 0.115 M H2CO3. Express your answer using two significant figures. b)Calculate [H3O+] of the following polyprotic acid solution: 0.135 M H3C6H5O7. Express your answer using two significant figures.Acetic acid Ka= 1.75 x 10^-5, 5.00 M

