All of these alkenes will form this alcohol. Figure 10 |BH3 сн, он CH3-CH- CH-CH, 2 H,02, NAOH A B CH, CH2 CH;=c-CH2-CH, CH3-C-CH,- CH, CH, CH,-c=CH-CH, CH, CH3-CH- CH=CH2 09:00 25°C Sunny 2021/11/19
All of these alkenes will form this alcohol. Figure 10 |BH3 сн, он CH3-CH- CH-CH, 2 H,02, NAOH A B CH, CH2 CH;=c-CH2-CH, CH3-C-CH,- CH, CH, CH,-c=CH-CH, CH, CH3-CH- CH=CH2 09:00 25°C Sunny 2021/11/19
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter2: Matter And Energy
Section: Chapter Questions
Problem 7E: 7.The word pour is commonly used in reference to liquids but not to solids or gases. Can you pour a...
Related questions
Question
Transcribed Image Text:LU eFu X
NCH X
E NCH X
Ь Му х
LU Oef x
LU Oef x
LU Oef x
U Oef x
n Oef x
n Oef x
O 202 x
n Oef x
U Mic x
A docs.google.com/forms/d/e/1FAlpQLSfSGcE8gf218c6JoHMWME1A8Nydf8M4g1yG93D-2LPpoMj_g/formResponse
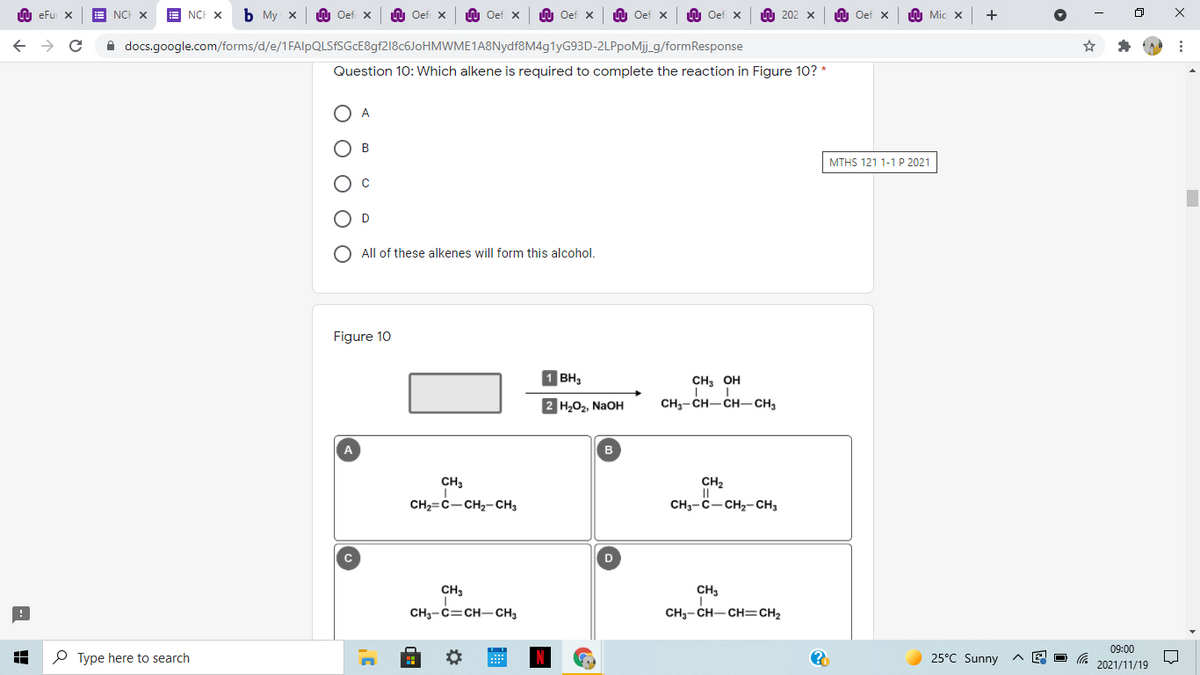
Question 10: Which alkene is required to complete the reaction in Figure 10? *
A
B
MTHS 121 1-1 P 2021
D
All of these alkenes will form this alcohol.
Figure 10
1 BH,
CH, OH
2 H202, NAOH
CH3- CH-CH-CH3
A
B
CH3
CH2
CH2=C-CH2-CH3
CH3-C-CH2- CH3
D
CH3
CH3
CH3-C=CH-CH3
CH3- CH-CH=CH2
09:00
P Type here to search
25°C Sunny
2021/11/19
O O O O
Transcribed Image Text:LU eFu X
NCH X
E NCH X
Ь Му х
U Oef x
n Oef X
U Oef x
O Oef x
LU Oef x
U Oef x
A 202 x
n Oef x
U Mic x
A docs.google.com/forms/d/e/1FAlpQLSfSGcE8gf218c6JoHMWME1A8Nydf8M4g1yG93D-2LPpoMj_g/formResponse
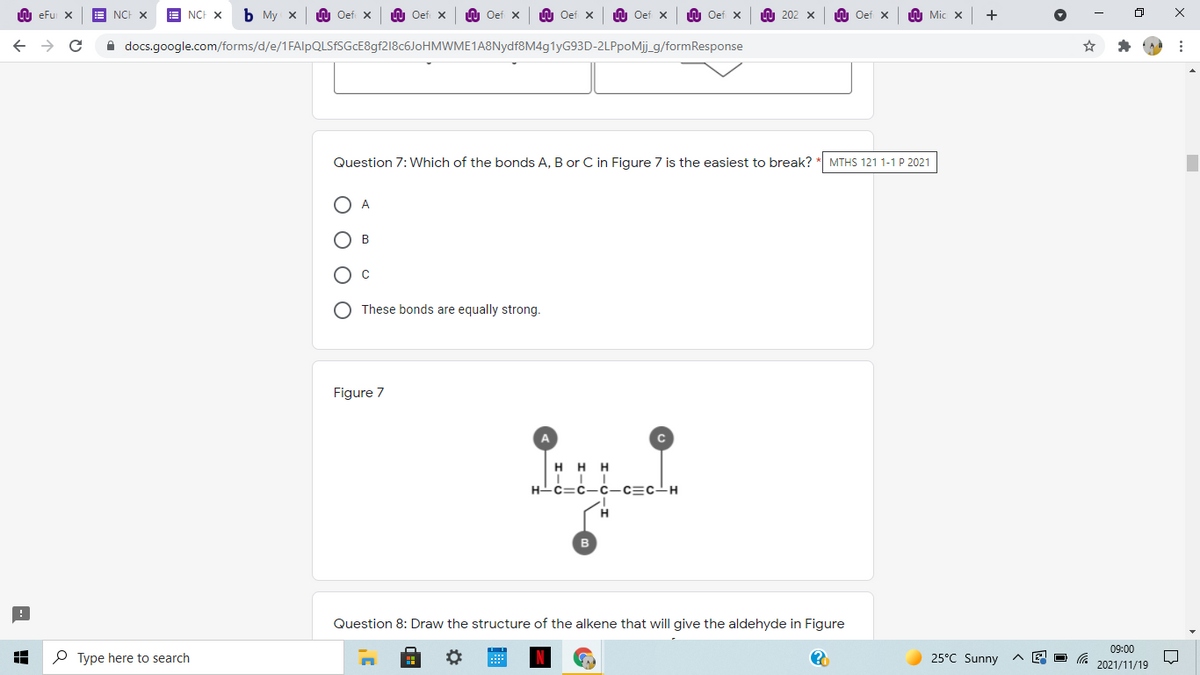
Question 7: Which of the bonds A, B or C in Figure 7 is the easiest to break?
MTHS 121 1-1 P 2021
A
В
These bonds are equally strong.
Figure 7
A
H H H
H-c=c-c-c=c-H
H.
Question 8: Draw the structure of the alkene that will give the aldehyde in Figure
09:00
P Type here to search
25°C Sunny
2021/11/19
оооо
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning