Give IUPAC names of the following compounds and identify the alcohols as primary, secondary or tertiary. (b) H3C-CH2-CH-OH ČH3 (а) CH3 H3C-Ć-OH ČH3 CH3 H3C-CH2-CH2-Ċ-OH H,C-CH, CH2 ČH3 (с) (d) CH3 Но CH2-CH3 H3C C=CH CH3 (е) (f) но-сн, HO
Give IUPAC names of the following compounds and identify the alcohols as primary, secondary or tertiary. (b) H3C-CH2-CH-OH ČH3 (а) CH3 H3C-Ć-OH ČH3 CH3 H3C-CH2-CH2-Ċ-OH H,C-CH, CH2 ČH3 (с) (d) CH3 Но CH2-CH3 H3C C=CH CH3 (е) (f) но-сн, HO
General, Organic, and Biological Chemistry
7th Edition
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:H. Stephen Stoker
Chapter14: Alcohols, Phenols And Ethers
Section: Chapter Questions
Problem 14.71EP: Three isomeric pentanols with unbranched carbon chains exist. Which of these isomers, upon...
Related questions
Question
100%
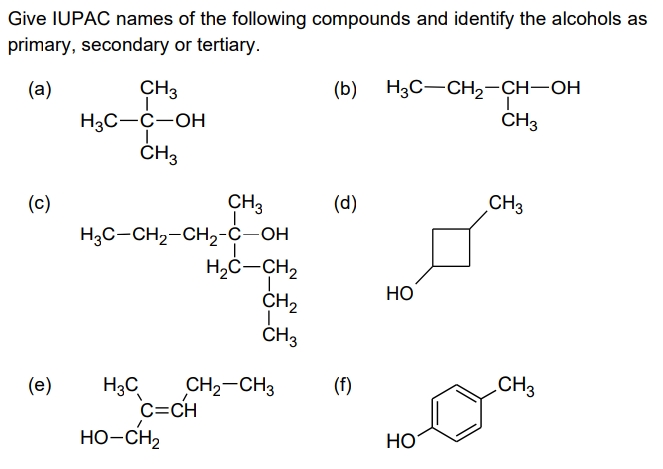
Transcribed Image Text:Give IUPAC names of the following compounds and identify the alcohols as
primary, secondary or tertiary.
(а)
CH3
(b) H3C-CH2-CH-OH
H3C-C-OH
ČH3
ČH3
CH3
H3C-CH2-CH2-Ċ-OH
H,Ċ-CH,
ČH2
ČH3
(с)
(d)
CH3
НО
(е)
.CH3
CH2-CH3
C=CH
HO-CH2
H3C
(f)
НО
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 7 steps with 11 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:
9781305081079
Author:
STOKER, H. Stephen (howard Stephen)
Publisher:
Cengage Learning,