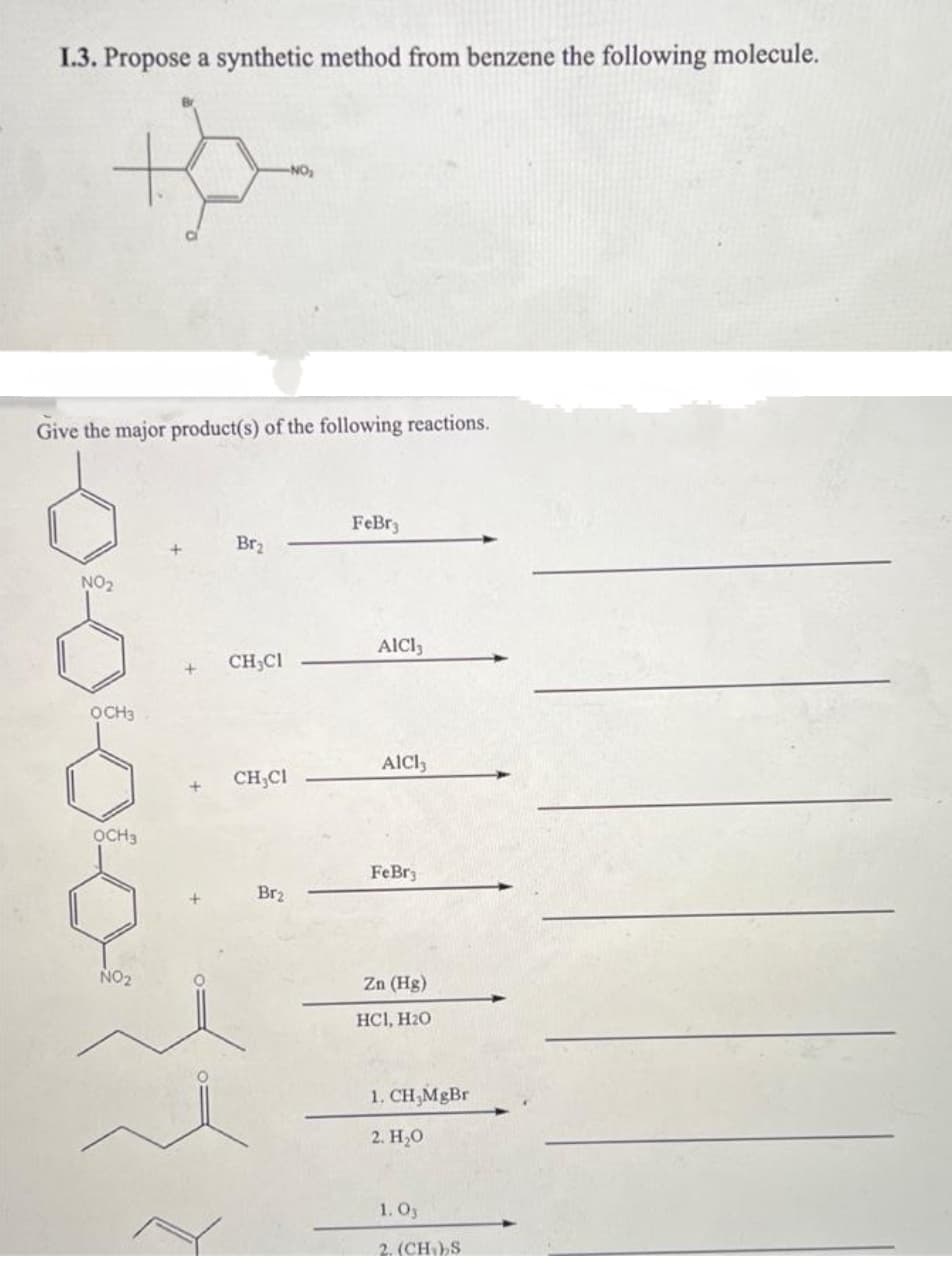
1.3. Propose a synthetic method from benzene the following molecule. NO Give the major product(s) of the following reactions. FeBr3 Br2 NO2 AIC3 CH,CI OCH3 AlCl, CH;CI OCH3 FeBr Br2
Q: Calculations and Analysis: 1. If vinegar is 5% acetic acid and the density of vinegar is 1.05 g/mL, ...
A: Since you have posted multiple questions, as per our company guidelines we are supposed to answer on...
Q: Synthesis Here are the targets. Starting compounds will be provided. 0. NHCH, starting Compound: sta...
A:
Q: Consider the following reaction where K. = 6.50x10-3 at 298 K. 2NOBR(g) 2NO(g) + Br2(g) A reaction m...
A:
Q: Determine the percent composition by mass of a 100 g salt solution which contains 20g of salt.
A: Weight of salt = 20 gm Weight of solution = 100 gm
Q: If 12.0 g Pb (c = 0.295 J/g °C) at 115°C is placed in 85 ml of water at 22°C, determine the final te...
A:
Q: Draw tructu f he ajor rganic roduct f he ollowing reaction. CH2 1. Hg(OAc)2, H20 2. NaBH4 CH3 • In c...
A: Hg(OAc)2 along with water and followed by NaBH4 are used to oxidize an alkene to alcohol.
Q: Use the following information for the following three questions. AH --524 kJ/mol AS- -250 J/K-mol D ...
A: Given, ∆H° = -524 kJ/mol = 524000 J/mol ∆S° = -250 J/k.mol
Q: Predict the products of the reaction below. That is, complete the right-hand side of the chemical eq...
A:
Q: Br ÓH end here start here
A:
Q: Calculate the mass of Li necessary to bind to 2 moles of nitrogen.
A: Mole can be defined as a standard unit for measuring large quantities of very small entities such as...
Q: An imaginary ionic salt Na 5X has a molar solubility of 4.43*10^-12. What is the Ksp value for this...
A: molar solubility of salt = 4.43 * 10-12
Q: How does how the molecular identity of the element change in space and/or time? Be sure to make the ...
A: A name that uniquely identifies a compound, molecule or substance is known as chemical identity. In ...
Q: At a particular temperature, Kp = 0.27ó for the reaction %3D N204(9) = 2NO2(g) A flask containing on...
A: Answer: Given chemical equation is: N2O4(g)↔2NO2(g) Equilibrium constant for this equilibrium is: Kp...
Q: 6. Determine which mechanism is used (SN1, SN2, E1, E2) and draw all steps for the following transfo...
A: Here we have to predict the types mechanism i.e substitution or elimination occur in the following g...
Q: Why is - CN a weaker leaving group than - NHCH3.
A: Weak Bases are the Best Leaving Groups. (Recall that the stronger the acid, the weaker the conjugate...
Q: Answer the following for the titration of 50.00 mL of 0.050 M nitric acid with 0.10 M NaOH. Report a...
A: Balanced equation for the reaction of nitric acid, HNO3(aq) and NaOH(aq) is: HNO3(aq) + NaOH(aq) ---...
Q: and
A: (a) The Se contains 3P ground state and a 3D and 3F excited state. The electronic transition takes p...
Q: a) b) c) Calculate the M oxidation states: Draw the structures: What are the colours and why? K„[Mo,...
A:
Q: How many moles of potassium would be necessary to produce 23.5g of potassium oxide?
A: Mass of potassium oxide produced = 23.5 g Here, we have to calculate the moles of potassium that are...
Q: Temperature (K) Rate Constant (L mol- s1) Ea = 23.0 kJ mol¬1 90 0.00357 Submit Previous Answers 100 ...
A: Given, Ea = 23.0 kJ T = 90 K k = 0.00357 M-1 s-1
Q: A sample containing both HCl (FW 36.45) and HNO3 (FW 62.98) had a density of 1.083 g/mL. A 10.00-mL ...
A: Given: Volume of sample = 10.00 mL. Concentration of NaOH = 0.1163 M Volume of NaOH solution require...
Q: Questions 6a, b, c, d is finked to one another and focus on the molecule shown below. O: H C2 H. I-Z...
A: Here we have to determine the proper geometrtical arrangements about the given central atoms by appl...
Q: If 7.43 L of gas at 15°C and 4.66 atm is compressed at a pressure of 132 atm and 45°C, calculate the...
A:
Q: 5. The enthalpy of formation for ethanol (CHẠO (1) is AHº = -200.7 kJ/mol Which of the reactions bel...
A: Now consider combustion of methanol CH4O CH3OH (l) + 3/2 O2 (g) →CO2 (g) + 2H2O (l) .........(1) Δ...
Q: Calculate the pI of the Glycine,Valine,Leucine,Proline, Histidine
A: Here, it is given to find out the iso electric point of the given aminoacids. They are Glycine,Valin...
Q: Ammonium perchlorate (NH,clo,) is the solid rocket fuel used by the U.S. Space Shuttle. It reacts wi...
A: Given-> Weight of NH4ClO4 = 8.0 gm
Q: Determine the pH of 5.416 x 10-8 M of HCl. (use the systematic treatment) Determine the pH of 7.814 ...
A: 1) Concentration of HCl solution = [HCl] = 5.416 x 10-8 M pH of the solution = ? Note: HCl is a st...
Q: Does Kl and Na2S form a precipitate? If so describe this solid or aqueous precipitate.
A: Na2S + KI —> Na2S= Sodium sulphide KI= Potassium iodide In the given reaction, Sodium sulphide re...
Q: Need both sets of compounds ranked strongest to weakest
A:
Q: 1, (e) +1,e)2e) 1st attempt Jhd See Periodic Table O See Hint Initially, 0.146 mole of H. 0.146 mole...
A: Given, Volume of the vessel = 1 L Concentration of H2 [H2] = 0.146/1 = 0.146 M Concentration of I...
Q: Predict the products of the reaction below. That is, complete the right-hand side of the chemical eq...
A:
Q: Adding a solute will change the normal boiling point or freezing point of a solution. Is this correc...
A:
Q: 10. Draw the following compounds. Make sure to describe the hybridization, the shape, whether it is ...
A: Hybridization is determined with the help of number of sigma bonds formed by atom and number of lone...
Q: CH3 is mixed with H,SO4 at high temperature OH
A: Protonation of alcoholic oxygen Loss of leaving group Carbocation rearrangement Deprotonation
Q: In an experiment involving the extraction of essential oils from a plant, 7.5 mL of benzene (C6H6, 7...
A:
Q: Part A Concentration-time data for the conversion of A and B to D are listed in the following table:...
A: The data given is,
Q: Identify the stronger acid between each pair and justify your answer. 1. HI аnd HF 2. ΗΝΟ, and HNO, ...
A: Given : Identify the stronger acid between each pair 1. HI and HF 2. HNO3 and HNO2 3. HBrO₂ and HC...
Q: Consider the hydrogen-oxygen fuel cell where H2(g) + O2(g) = H20() DG° = -237.18 kJ/mol H2 Which of ...
A: Since you asked multiple questions so, as per Q&A guidelines of portal I solved first image ques...
Q: Enter the number of electrons in each energy level (shell) for each of the elements. If the energy l...
A:
Q: HC2H3O2 (aq) ↔ C2H3O2- (aq) + H+ (aq) At equilibrium at 25 ºC, a 0.100 M solution of acetic ac...
A: From the given chemical equation , first we would write the expression for Keq, the using given equi...
Q: Describe the cleaning action of soap
A:
Q: formulas formed between the given ions. Barium and Chlorine Show solutions. Follow the format in an...
A: a) electronic configuration Ba = 1s2,2s2,2p6,3s2,3p6,3d10,4s2,4p6, 4d10, 5s2 ,5p6,6s2 Cl = 1s2,2s2,...
Q: A mixture of a gas in a 300 L container at 273 K with a pressure of 0.75 atm contains 6.7 mol of H₂ ...
A: Total pressure (PTotal) = 0.75 atm Moles of H2 (nH2 ) = 6.7 mol Moles of O2 (nO2) = 3.3 mol Total ...
Q: helpp asap!! An aqueous solution of hydrofluoric acid is 16.7% HF, by mass, and has a density of 1.1...
A:
Q: 2. Determine the pH for the following 0.100 M LiF (Kа — 6.6 х 104) а) b) 3.2x 10° М Вa(ОН)2
A:
Q: A. Write the formula of the conjugate acid or base of the following substances: 1. Acids a. HC1O b. ...
A:
Q: What weight of pyrite ore (impure FeS2) should be taken for analysis that the no. of centigram of pr...
A: A question based on mole concept that is to be accomplished.
Q: 7) Determine the point group for each of the following molecules. For each molecule, list which symm...
A:
Q: 4) Would you expect cis-1,2-dimethylcyclohexane and trans-1,2-dimethylcyclohexane to have a good sep...
A: TLC is a type of chromatography used in separation of non volatile Mixture. Its stationary phase is ...
Q: A chemical reaction takes place inside a flask submerged in a water bath. The water bath contains 3....
A: Given : mass of water = 3 kg
5
Step by step
Solved in 3 steps with 2 images

- For butenafine, develop a synthesis that starts with napthalene and benzene. (aromatic methyl groups can be converted to alkyl bromides using Br2/light, and aldehydes by heating the alkyl bromide in DMSO, eg. ArCH3 to ArCH2Br then to ArCHO).Propose a mechanism for the followingreaction.Answer in 2 sig figs, thank you!
- Propose a structure consistent with each set of data.plz provide DETAILED written explanation and AVOID USING MECHANISMS.Fast pls in 5 min will give u like for sure Create a synthesis of the heterocyclic compound 5 from starting materials each containing no more than six carbon atoms.In the solution you should use retrosynthetic analysis to justify your choice.
- Given this organic synthesis, is there any limiting and excess reagents? also describe the procedure is run, how is the reaction monitored?Is the order fo addition important? N-ETHYLALLENIMINE[Aziridine, 1-ethyl-2-methylene-] Submitted by Albert T. Bottini and Robert E. Olsen1.Checked by Thomas H. Lowry and E. J. Corey. 1. ProcedureCaution! This preparation should be carried out in a good hood to avoid exposure to ammonia. The operator should wear rubber gloves and protective goggles because 2-haloallylamines and ethylenimines can cause severe skin and eye irritation. A 2-l. three-necked flask is fitted with a sealed mechanical stirrer, a gas-inlet tube, and a dry ice condenser protected from the air by a soda-lime drying tube (Note 1). The system is flushed thoroughly with dry ammonia, and 32.8 g. (0.84 mole) of sodium amide (Note 2) is added to the flask. The system is again flushed with ammonia, the condenser is provided with dry ice covered by acetone, and 1.2 l. of liquid…Synthesize 3,5-dibromotoluene from benzene and any suitable inorganic and organic reagents. Assume you CANNOT separate ortho and para products and both isomers have to be carried through in the entire synthesis.Cleavage of oct-1-ene with hot, KMnO4 will yield _____________. octanol and carbon dioxide octanone hexanol and 2 carbon dioxides heptanal and formaldehyde heptanoic acid and carbon dioxide

