An electrochemical cell is made from the electrode systems shown by these half-equations. Ag (aq) + e = Ag(s) E° = +0.80V VO;(aq) + 2H'(aq) + e = vo"(aq) + H,O() E° = +1.00V The apparatus used to measure the value for E under standard conditions is shown. voltmeter silver platinum 1 mol dm Agʻ(aq) electrolyte salt bridge (a) Which silver compound could be used as the electrolyte in the left-hand half-cell? O A silver nitrate O B silver hydroxide I C silver chloride O D silver carbonate (b) The electrolyte in the right-hand half-cell is prepared using equal volumes of O A 1 moldmacidified VO (aq) and 1 mol dm' acidified Vo"(aq) O B 2moldmacidified VO; (aq) and 2 mol dm acidified Vo²"(aq) C ImoldmVo;(aq) and 1 mol dm3 HCl(aq) 1 mol dmvo"(aq) and 1 mol dm HCl(aq)
An electrochemical cell is made from the electrode systems shown by these half-equations. Ag (aq) + e = Ag(s) E° = +0.80V VO;(aq) + 2H'(aq) + e = vo"(aq) + H,O() E° = +1.00V The apparatus used to measure the value for E under standard conditions is shown. voltmeter silver platinum 1 mol dm Agʻ(aq) electrolyte salt bridge (a) Which silver compound could be used as the electrolyte in the left-hand half-cell? O A silver nitrate O B silver hydroxide I C silver chloride O D silver carbonate (b) The electrolyte in the right-hand half-cell is prepared using equal volumes of O A 1 moldmacidified VO (aq) and 1 mol dm' acidified Vo"(aq) O B 2moldmacidified VO; (aq) and 2 mol dm acidified Vo²"(aq) C ImoldmVo;(aq) and 1 mol dm3 HCl(aq) 1 mol dmvo"(aq) and 1 mol dm HCl(aq)
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter17: Electrochemistry
Section: Chapter Questions
Problem 160MP
Related questions
Question
Please give me handwritten answer?
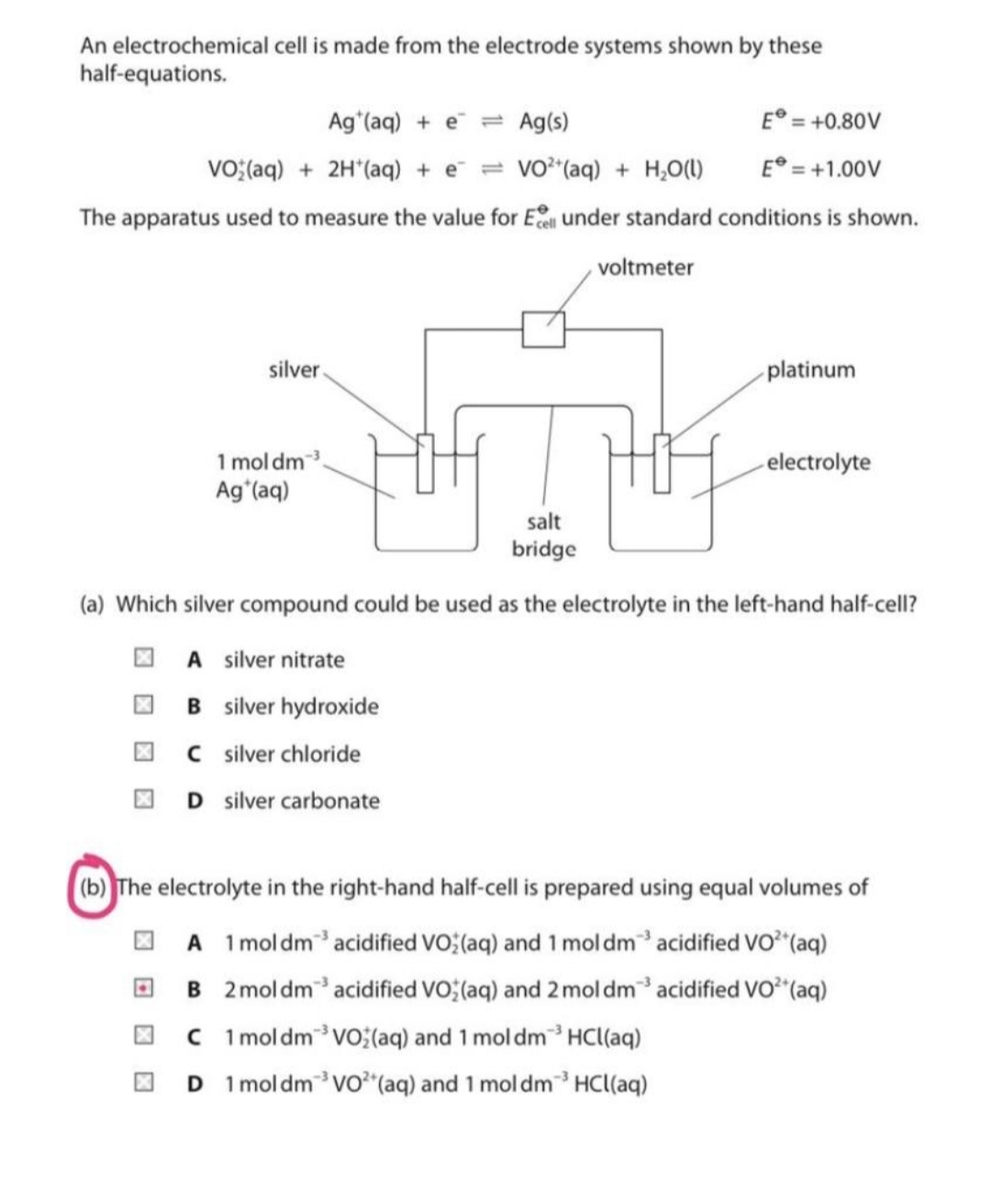
Transcribed Image Text:An electrochemical cell is made from the electrode systems shown by these
half-equations.
Ag (aq) + e = Ag(s)
E° = +0.80V
VO;(aq) + 2H*(aq) + e¯ = vo²(aq) + H,O(1)
E° = +1.00V
The apparatus used to measure the value for E under standard conditions is shown.
voltmeter
silver
platinum
1 mol dm3
Ag'(aq)
electrolyte
salt
bridge
(a) Which silver compound could be used as the electrolyte in the left-hand half-cell?
O A silver nitrate
I B silver hydroxide
O C silver chloride
D silver carbonate
(b) The electrolyte in the right-hand half-cell is prepared using equal volumes of
O A 1moldmacidified VO; (aq) and 1 mol dm acidified vo (aq)
O B 2moldm acidified VO (aq) and 2 mol dm acidified vo*"(aq)
O C 1 moldmVo(aq) and 1 mol dm³HCl(aq)
O D 1moldmvo*"(aq) and 1 mol dm³ HCl(aq)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning