Q: An object with a mass of 0.255 kg and density of 2.89 g/cm3 measures 34 mm in length and 46 mm in…
A: The mass of the object is = 0.255 kg The density of the object is = 2.89 g/cm3 The length and width…
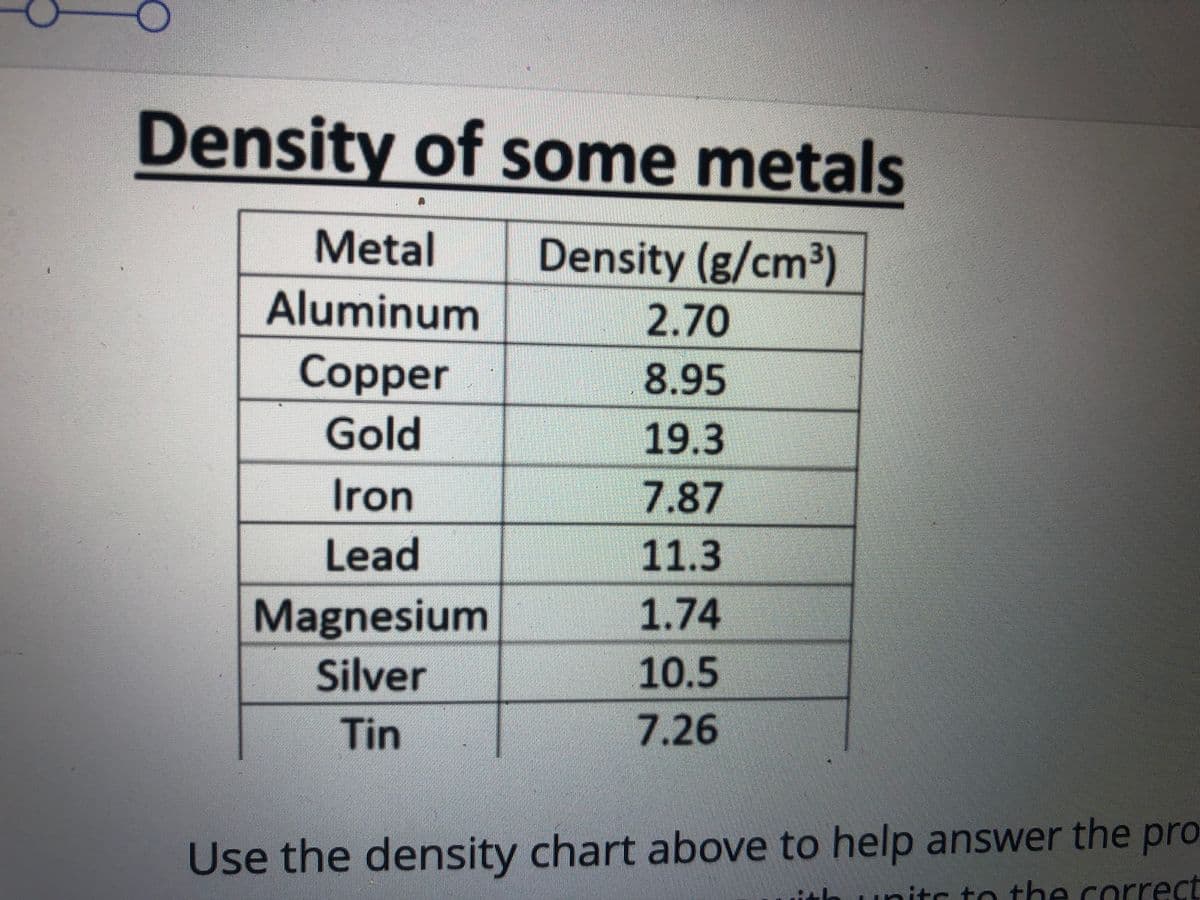
Q: 9. If 86 g of a substance has a volume of 125 cm³, what is the density of the substance?
A:
Q: The mass of an empty container is 85.50 g . The mass of the container and a liquid with a density of…
A: Given data:- * mass of empty container = 85.50 g * mass of container and liquid = 150.25 g So, *…
Q: if a substance has a density of 19.1 g/cm3 , what is the density?
A: The density of a substance is defined as a mass of a unit volume of the substance. The density of a…
Q: What is the volume in L of a cube with an edge length of 7.0 dm?
A: Volume can be defined as the amount of the space taken by an object. It represents the ability of an…
Q: a rectangular block of cadmium has a length of 10cm and a width of 5cm. if the mass of the bloc is…
A: We have Cadmium block, Length=10 cm Width=5 cm height=h cm Density=8.64 g/cm3 mass=50 g
Q: A new penny has a mass of 2.55 g and a volume of 0.345 cm3. What is the average density of the…
A: The average density of the penny is to be calculated
Q: A steel cylinder has a length of 2.16 in, a radius of 0.22 in, and a mass of 41 g. What is the…
A: Given: The length of steel cylinder(h)=2.16 inch=5.48 cm The radius of the steel cylinder(r)=0.22…
Q: A spherical metal ball has a mass of 3.641 g and a diameter of 8.14 mm. What is the density of the…
A: diameter of spherical ball = 8.14 mm∴ radius of spherical ball = diameter2…
Q: An irregularly-shaped piece of aluminum has a mass of 88.8 grams. What is the volume of this piece…
A: Concept introduction: Density is equal to the ratio of mass to volume. Formula: If the density of…
Q: A cube of silver is 5.00 cm on a side and has a mass of 1312.5 g. What is the density of silver?
A: Density is given by the ratio of mass to volume. The volume of the cube is given by Volume=(side)3…
Q: 2. The density of human blood is 1.06 g/mL. What is the volume of 0.6822 g of blood? A bar of gold…
A: 2) Given: Density of human blood = 1.06 g/mL Mass of blood = 0.6822 g Calculation for volume of…
Q: What is the volume of 40.0 kg of gold, which has a density of 19.3 g/ml?
A:
Q: 248
A: Density= mass/volume Mass = density× volume Density=10.5 g/ cm3 Volume= 23.6cm3
Q: An irregularly-shaped piece of chromium (Cr) has a mass of 65.0 grams. What is the volume in cm³ of…
A: givenmass of Chromium=65.0 gramsdensity of Chromium=7.19 g/cm3
Q: An object has density of 19.3 g/ml. What is the mass of 25.0 mL of this object?
A: Given : density of object = 19.3 g/mL And volume of object = 25.0 mL
Q: you have a rock that displaces a volume of 15 cm (cubed) and a mass of 45 g. what is its density?
A: It is given that a rock displaces a volume of 15 cm3 and has a mass of 45 g.Since, the volume…
Q: The density of liquid mercury is 13.5 g/cm3. What mass of mercury will fill a 0.500 L container?
A: Given Density = 13.5 g/cm3 Volume = 0.500 L
Q: The density of mercury is 13.6g/cm3. What is the mass in pounds of 1.00 quart of mercury
A: 1 quart=946.53cm3 Density=mass÷volume 13.6=mass÷946.53 Mass=13.6×946.53 Mass=12872.8gm…
Q: what volume will 88.0 g of an object with a density of 3.44 g/mL occupy?
A: The given data contains, mass =88.0 g density=3.44 g/mL
Q: A cube of lead has a mass of 54.3g. If the density of lead is 13.5g/cm3, what is the edge length of…
A: Given :- mass of cube of lead = 54.3 g Density of lead = 13.5 g/cm3 To calculate :- Edge length…
Q: Osmium has a density of 22.6 g/cm3. What volume (in cm3) would be occupied by a 21.8 g sample of…
A: Given, Density of osmium = 22.6 g / cm3 Mass of a osmium sample = 21.8 g Required,…
Q: What would the volume (in cm) of an aluminum cube be if the density is 2.70 g/cm3 and the mass is…
A: Density = Mass / Volume Mass in grams(g) Volume in cm3 Unit of Density = g/cm3
Q: A rectangular block of cadmium has a length of 10 cm and a width of 5 cm. If the mass of the block…
A:
Q: Bromine has a density of 3.12 g/cm3. What is the volume of 25.0 grams of bromine
A: Given question is completely formula based problem, which asking about the volume of bromine. We…
Q: An irregularly-shaped piece of iron (Fe) has a mass of 60.0 grams. What is the volume in cm³ of this…
A:
Q: What is the density of an unknown metal of 45.36 g of metal occupies a volume of 3.62 cm^3?
A: Density can be calculated by dividing mass( in gram) by Volume in cm3
Q: A cube of aluminum weighs 72.90 g and has a density of 2.70 g/cm3. What is the length of each side…
A:
Q: 5. A rectangular block of an unknown solid has a mass of 441.26 grams and measures 12.00 cm by 3.00…
A: Given, The mass of an unknown solid is 441.26 grams and measures 12.00 cm by 3.00 cm by 1.50 cm.
Q: An object with a mass of 0.255kg and a density of 2.89 g/cm^3 measures 34mm in length and 46mm in…
A: Given : Mass of object = 0.255 Kg Density of object = 2.89 g/cm3 Length of object = 34 mm = 3.4 cm…
Q: The density of mercury is 13.6 g/cm3. What is its density in mg/mm3?
A: Given: Density of mercury is 13.6 g/cm3
Q: Calculate the density of mercury if 1.00 x 10² g occupies a volume of 7.36 cm3.
A: Solution: Given that, Mass = 1.00 * 102 g Volume = 7.36 cm3 We know that, Density = mass / volume…
Q: If lead has a density of 11.4 g/cm3, what is the volume of 5,200 g of lead
A:
Q: A block of ice has edge lengths of 8.00 cm3 each and a mass of 476g. What is the density of ice in…
A: Answer :- Density Of Ice Block = 0.930 (g/cm3) Density of Ice block having edge length 8.00 cm and…
Q: Calculate the density mercury if 1.00x 10 squared g occupies a volume of 7.36cm cubed
A:
Q: A cylindrical rod of iron metal has a density of 7.87 g/cm3. If the mass of the rod is 54.00 g, then…
A: density = mass / volume
Q: A solid metal sphere has a radius of 3.84 cm and a mass of 1.765 kg. What is the density of the…
A: density is related to the mass (g) and volume (cm^3) of a substance by the formula,density = mass…
Q: What is the density of a ball with a mass of 561 g and a radius of 5.00 cm?
A: Given;
Q: Lithium is the least dense metal known (density: 0.53 g/cm3). What is the volume occupied by 1.20…
A: Given : Density of Li = 0.53 g/cm3 And mass of Li = 1.20 X 103 g
Q: An object has a volume of 4.3 x 103 cm3, and a density of 13.27 g/cm3. WHAT IS THE MASS OF THE…
A: The volume of the object is given as 4.3 x 103 cm3 and the density is given as 13.27 g/cm3. The mass…
Q: An irregularly-shaped piece of aluminum (Al) has a mass of 57.2 grams. What is the volume in cm³ of…
A: Density (g/cm3) = Mass(gram)/volume(cm3) So, volume= mass/density So by the above formula…
Q: A 0.418 kg sample of methylene chloride has a density of 1.326 g/cm3. Calculate its volume.
A: Weight of the sample is 0.418 kg = 418 g Density is 1.326g/cc Volume = 418/1.326 = 315.233 cc
Q: What is the volume of an osmium sphere that has a mass of 9.991 g? The density of osmium (Os) is 22…
A: The density of a substance is defined as the ratio of mass to volume or in other words, the volume…
Q: The average density of a planet is 0.500 g/cm3. What is its density in kg/m3? (Enter your answer in…
A: The ratio of mass to volume is known as density
Q: A 5.73g cube measures 2cm in length,12cm in width, and 0.25cm in height. What is the density of the…
A: Density of an object is the mass of an object per volume of an object. It is expressed as,…
Q: The density of silver is 10.49 g/cm3. What is the volume of a silver coin that has a mass of 31.103…
A: The mass of the unit volume of the substance is known as the density of the substance. The density…
Q: A sample of metal has a mass of 19.13g, and a volume of 5.18mL. What is the density of this metal?
A: Given: Mass of metal = 19.13 g. And the volume of metal = 5.18 mL.
Q: A 17.0 kg iron weightlifting plate has a volume of 2160 cm³ . What is the density of the iron plate…
A: Introduction The density of any particular metal can be derived by calculating the ratio between the…
Q: A sample of mercury is found to have a mass of 524.5 g and to have a volume of 38.72 cm3. What is…
A:
Q: Calculate the density of mercury if 1.00 * 102 g occupies a volume of 7.36 cm3.
A: Density of a substance is equal to the mass of substance divided by its volume. Write the expression…
Question # 2
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- The active ingredient of aspirin tablets is acetylsalicylic acid, which, has a density of 1.4 g/cm3. In a lab class, a student used paper chromatography to isolate another common ingredient of headache remedies. The isolated sample had a mass of 0.384 g and a volume of 0.32 cm3. Given the data in the following table, what was the other ingredient in the headache remedy? Density Values for Potential Headache Remedies Compound Density (g/cm3) White table sugar 0.70 Caffeine 1.2 Acetylsalicylic acid 1.4 Sodium chloride 2.2The density of an irregularly shaped object was determined as follows. The mass of the object was found to be 28.90 g 0.03 g. A graduated cylinder was partially filled with water. The reading of the level of the water was 6.4 cm3 0.1 cm3. The object was dropped in the cylinder, and the level of the water rose to 9.8 cm3 0.1 cm3. What is the density of the object with appropriate error limits? (See Appendix 1.5.)1.) A dense plastic-like shape has a mass of 42.985 g and has a volume of 41.96 mL. What is the density of the substance? 2.) Another plastic-like piece has a mass of 65.389 grams and displaces 53.652 grams of water (measured at 24.6 ⁰C). Is this piece likely to be composed of the same material as the sample in problem 1? Explain using the Claim-Evidence-Reasoning process.
- Calculate the density of a spherical metal, in g/cm3, with a diameter of 4.77 in and and a mass of 2.41 lbs. 1lb=454g *1 in=2.54cm Vol of sphere= 4/3 ??3(a) To identify a liquid substance, a student determined itsdensity. Using a graduated cylinder, she measured out a 45-mLsample of the substance. She then measured the mass of thesample, finding that it weighed 38.5 g. She knew that the substancehad to be either isopropyl alcohol (density 0.785 g/mL)or toluene (density 0.866 g/mL). What are the calculated densityand the probable identity of the substance? (b) An experimentrequires 45.0 g of ethylene glycol, a liquid whose densityis 1.114 g/mL. Rather than weigh the sample on a balance, achemist chooses to dispense the liquid using a graduated cylinder.What volume of the liquid should he use? (c) Is a graduatedcylinder such as that shown in Figure 1.21 likely to afford theaccuracy of measurement needed? (d) A cubic piece of metalmeasures 5.00 cm on each edge. If the metal is nickel, whosedensity is 8.90 g/cm3, what is the mass of the cube?Composite decking is a manufactured substitute for woodcompounded from post-consumer plastic and wood. It isfrequently used in outdoor decks. The density of a particularcomposite decking is reported as 60.0 lb>ft3. What isthe density in kg/L? 0.961 kg/L
- To identify a liquid substance, a student determined itsdensity. Using a graduated cylinder, she measured out a 45-mLsample of the substance. She then measured the mass of thesample, finding that it weighed 38.5 g. She knew that the substancehad to be either isopropyl alcohol 1density 0.785 g>mL2or toluene 1density 0.866 g>mL2. What are the calculated densityand the probable identity of the substance?A manufacturer of paint claims that a gallon (3.78 L) of latex paint can cover 365 ft2 of the surface of a wall in a single coating. The paint manufacturer also claims that one single coating of an entire wall will be 3.40 mm thick. Is this a case of false advertising? Yes or No? Elaborate your answer by providing a calculation (handwritten, upload a pdf). You may use the following conversions: 12 in = 1 ft, 2.54 cm = 1 inYou are asked to determine the mass of a piece of copper using itsreported density, 8.96 g/mL, and a 150-mL graduated cylinder.First, you add 105 mL of water to the graduated cylinder; thenyou place the piece of copper in the cylinder and record a volumeof 137 mL. What is the mass of the copper reported with the correctnumber of significant figures? (a) 287 g (b) 3.5 * 10-3 g/mL(c) 286.72 g/mL (d) 3.48 * 10-3 g/mL (e) 2.9 * 102 g/mL
- The physician of patient AJ (50 kg, M) admitted in your hospital ordered that the patient receives 0.25 mL of drug A per kg of body weight. This must be mixed with 250 mL of D5W. The IV admixture must be administered over a six-hour period only using a drip set capable of delivering 15 drops per mL. Your pharmacy has stocks of 2.0% w/v of drug A solution (density of drug A: 1.2 g/mL). Answer all questions below. 1. How many mL of drug A must the patient receive to obtain the needed dose? 12.5 mL B. 25.0 mL C. 50.0 mL D. 75 mL 2. From the pharmacy’s stocks of drug A solution, how many mL must be added to 250 mL D5W to prepare the IV admixture? 250 mL B. 500 mL C. 750 mL D. 1000 mL 3. What is the total volume of the admixture that will be administered to the patient? 262.50 mL B. 500 mL C. 750 mL D. 1000 mL 4. Given the capability of the hospital’s IV set, how many drops of the admixture should be administered to the patient? 3,936.5 drops B. 7,500 drops C. 11,250 drops D.…You are asked to determine the mass of a piece of copper using itsreported density, 8.96 g/mL, and a 150-mL graduated cylinder.First, you add 105 mL of water to the graduated cylinder; thenyou place the piece of copper in the cylinder and record a volumeof 137 mL. What is the mass of the copper reported with the correctnumber of significant figures? (a) 287 g (b) 3.5 * 10-3 g/mLA steel ball-bearing with a circumference of 26.1 mm weighs 200 mg. What is the density of the steel in g/cm3? (V of a sphere = 4/3 πr3; circumference of a circle = 2πr)?

