An unknown sugar is found to have a molecular weight of 180.18 amu. The sugar contains 40.O % carbon, 6.7 % hydrogen and 63.3% oxygen. Find the molecular formula of this sugar. Enter your answer with the elements in the same order as they appear in the question. Also, you will not be able to make subscripts in the answer. The formula for water would be entered as H20 rather than H2O Answer
An unknown sugar is found to have a molecular weight of 180.18 amu. The sugar contains 40.O % carbon, 6.7 % hydrogen and 63.3% oxygen. Find the molecular formula of this sugar. Enter your answer with the elements in the same order as they appear in the question. Also, you will not be able to make subscripts in the answer. The formula for water would be entered as H20 rather than H2O Answer
Chapter1: Molecular Reasons
Section: Chapter Questions
Problem 38E: Several samples of methane gas, the primary component of natural gas, are decomposed into carbon and...
Related questions
Question
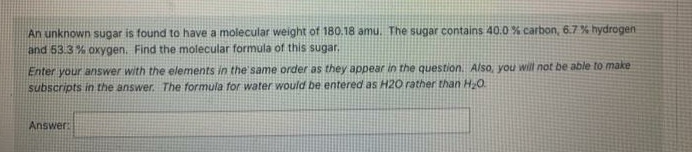
Transcribed Image Text:An unknown sugar is found to have a molecular weight of 180.18 amu. The sugar contains 40.0 % carbon, 6.7% hydrogen
and 53.3 % oxygen. Find the molecular formula of this sugar.
Enter your answer with the elements in the same order as they appear in the question. Also, you will not be able to make
subscripts in the answer. The formula for water would be entered as H20 rather than H20
Answer:
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning