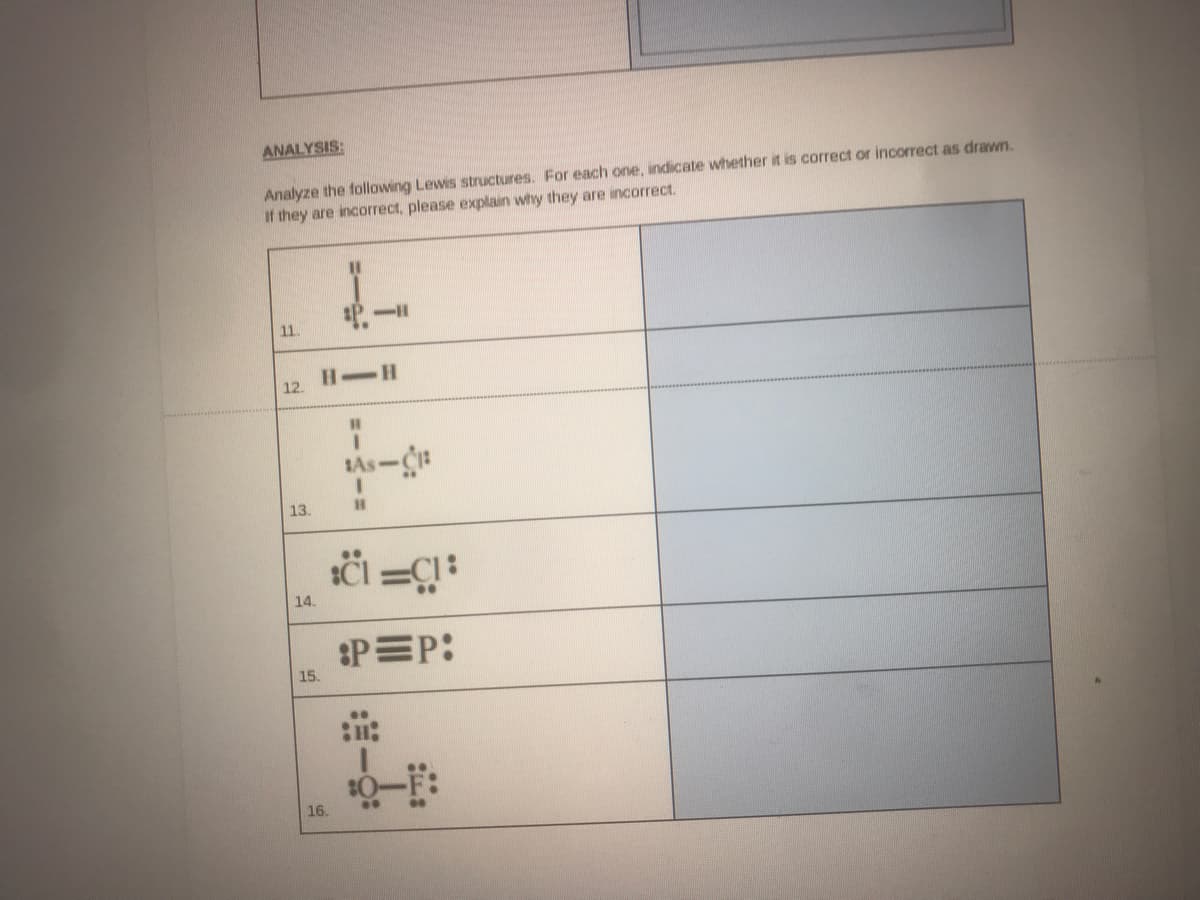
Analyze the following Lewis structures. For each one, indicate whether it is correct or incorrecl If they are incorrect, please explain why they are incorrect. %3D 11. H-H 12. SAs-CH 13.
Q: snipp
A: Since you have posted a question with multiple subparts, we will solve the first three subparts for…
Q: H H-C- C-H H H エー0ーエ
A: Name of the given Lewis structure is Dimethyl ether.
Q: D. Structure of Molecules and Molecular lons On a separate sheet of paper, set up the following…
A: As per the bartleby guidlines , I can solve only first three questions of multipart or only first…
Q: How many valence electrons does ClO2- get in its Lewis Dot diagram? a) 20 b) 19…
A: Please find below the Lewis structure of ClO2-
Q: 41. Which species has a Lewis diagram and structure most like that of the carbonate ion? NO3 c.…
A: Lewis structure of the given compounds:
Q: 45. The Lewis structures for CH4, N2, and CO2, along with the corresponding space-filling molecular…
A: According to Lewis theory of bonding atoms can complete their octet of valence electrons by shared…
Q: Valence Shell Bonding Nonbonding Approx. Bond Molecule or Lewis Electron Electron Electron VSEPR…
A: The total number of electrons present in the outermost shell of an atom are known as valence…
Q: Please draw the Lewis structure for each of the follow 1). HCN Of all the atoms in HCN, which is the…
A:
Q: The bond length of NaF is 231.00 pm. What would be the dipole moment, in D, of this compound by…
A:
Q: 35. Many monatomic ions are found in seawater, including the ions formed from the following list of…
A: Lewis symbol :- Lewis symbol is a representation of an element symbol along with its valence…
Q: Nonbonding Bonding Electron Valence Shell Approx. Bond Molecule or Lewis Electron Electron VSEPR…
A: The molecules given are PCl2F3, BrF3, XeF2.
Q: Which of the following Lewis structures is/are written correctly? H. H. H. :Cl: :F: Structure I…
A: ,
Q: Write the Lewis structure for each molecule. PH3
A: P belongs to N-family and thus it has 5 valence electron in its outermost valence shell while H has…
Q: Construct the Lewis structure of each of the following: (Show your solution.) a) HPO4²- b) H2SO4 c)…
A: Given molecules:
Q: Draw the Lewis structure of each molecule or ion and place the following in order of increasing…
A: BrCl3 Total valence electron = 7+7(3) =28 Central atom = Br Central atom Bromine has 3 bond pairs…
Q: Draw the Lewis structure for BrF with an arrow representing the dipole moment. Use Figure 5.5 to…
A:
Q: 9.61 Use Figure 9.21 to indicate the polarity of each bond with partial charges: (а) Br—CI (d) Se-H…
A: We know, Greater is the electronegativity value more polar is the atom/molecule.
Q: Choose the best Lewis structure for Xel2 (T A) -Xe–i: B) 1=Xe=r: D) -Xe-T: NF The shape of the…
A: Lewis Structure is a very simplified representation of the valence shell electrons in a molecule
Q: The pentafluoride I5^- drawing a lewis dot structure. I understand that the chemical formula is…
A: In this scenario of I5- The ion forms a V shape and it is also called to have a bent shape. These 5…
Q: smallest? Mg2+ Sr2+ Be2+ Ca2+ Which of the following is the best Lewis structure? :0=Si -0: ö=o= O A…
A:
Q: Consider the Lewis Structure for the molecule, PBR3. On your own, make a sketch of the VSEPR shape,…
A: VSEPR theory:The structure of a molecule can be determined by using the VSEPR theory. According to…
Q: Which statement is false given the following Lewis structures? 13- :Br :ö: :0-As-ö: В :Br Н-Se-H ..…
A:
Q: For PCL4F what are the: Number of lone pairs? Number of bonding pairs? AXE notation? Electronic…
A: Electronic geometry can be predicted by using the number of electron pairs sorrounding the central…
Q: Consider the Lewis structure for ozone (03) proposed by a student in General Chemistry: Which of the…
A:
Q: 10. The following Lewis structures for (a) HCN, (b) C>H2, (c) SnOz. (d) BF, (e)HOF are incorrect.…
A: Lewis Dot structures is the representation of the valence electrons of the atom. Total number of…
Q: 1. 8 6 OSHO) sbyrdsblamo bas ('e) noi muiboed This attraction happens between polar molecules. The…
A: As you posted questions with multiple questions, we will solve the first question for you. To get…
Q: You must show your Lewis structure in your work to receive credit. Complete the sentence using whole…
A: Lewis structure : A lewis structure is the simplified representation of valence electrons in a…
Q: 45. The Lewis structures for CH4, N2, and CO2, along with the corresponding space-filling molecular…
A: As you already mentioned that you need help in question number -45 so I am solving here your…
Q: Here are the molecular structures of C Br, and CH,Br. Use the images and the electronegativity chart…
A: We know that the electronegativity order for the atoms C, Br and H are Br > C > H Since there…
Q: Molecule Number Lewis Structure How many electrons are Does every atom have its version of or of…
A: Geometry:- Molecular geometry is the three-dimensional arrangement of the atoms that constitute a…
Q: Florine and Sodium ions form a polar covalent bond. Use a periodic table and your knowledge of…
A: Ionic bond is formed by transferring of electrons from one atom to another atom and formation of…
Q: In the BEST Lewis structure for the molecule XeF2, how many unshared pairs are there on the central…
A: The centre atom will be the one which is less electronegative And since F is more electronegative…
Q: Valence Shell Bonding Nonbonding Approx. Molecule or Lewis Electron Electron Electron VSEPR Bond…
A: From given Each molecule details and structures is assigned as per table as follows
Q: IERE? of electrons are required for 6.6x10-2 mol Ni? x mol e-
A: Given, Mass of Ni(m) = 3.9 g Current (i) = 108.2 A Number of electrons transferred (n) = 2 Faraday's…
Q: Below is the Lewis structure of the chloroform (CHCI,) molecule. .. : Cl:C: Cl: : Cl: Count the…
A: Please find the solution attached below:
Q: Table 1: Lewis Structures using Determined Formal Charges and Bond Polarity Molecul Lewis Lewis…
A:
Q: Bonding Electron Valence Shell Nonbonding Approx. Bond Molecule or Lewis Electron Electron VSEPR…
A: Three questions based on Lewis structure, which are to be accomplished.
Q: 1. Write a Lewis structure for each of the following: a) CHCI3 b) SeBr2 c) NS*
A:
Q: Consider the Lewis Structure for the molecule, SeF4. On your own, make a sketch of the VSEPR shape,…
A: The given structure of the molecule is,
Q: H. H-C-Ci : H. Based on the Lewis structure above, the central atom of this molecule has pairs of…
A:
Q: Nonbonding Electron Valence Shell Bonding Electron Approx. Molecule or Lewis Electron VSEPR Bond…
A: Lewis octet rules :- The octet rule is the tendency of atom to prefer to have…
Q: Valence Shell Bonding Nonbonding Approx. Molecule or Lewis Electron Electron Electron VSEPR Bond…
A: In Lewis dot structure all the valence electrons which are present in bonds as well as unshared…
Q: The Lewis structure for carbon dioxide (CO2) shown below is incorrect. Starting from this structure,…
A: The given structure is represented as follows:
Q: Draw a Lewis structure for the molecúles listed and correctly identify whether they are polar or…
A:
Q: The Lewis structure for H3BO3 is Select one: H-ö=B-Ö-H O a. :0: H H-Ö-B–Ö–H O b. :0: H H-Ö=B-Ö–H O…
A: Lewis structure: Lewis structures show the bonding between atoms of a molecule and the lone pairs of…
Q: mmetrical. 15. Electronegativity can influence the polarity of a molecule. 16. When electrons in a…
A: Answer of 15,16,17,18
Q: Consider the Lewis Structure for the molecule, [BrCl4]. On your own, make a sketch of the VSEPR…
A: VSEPR theory:The structure of a molecule can be determined by using the VSEPR theory. According to…
Q: Is there anything wrong with this Lewis Structure of ICI4 ? If no, explain why not. If yes, explain…
A: Answer: We have a WRONG lewis structure of ICL4- ion given above. The total valence electrons of…
Q: How many carbons are in the following structure? A) 8 How many carbons are in the following…
A: "Since you've asked multiple questions, we'll solve the first three parts for you and for the rest…
Q: Nonbonding Approx. Bond Valence Shell Bonding Molecule or Lewis Electron Electron Electron VSEPR…
A: The given question has subparts so I have solved three subparts.
Step by step
Solved in 4 steps with 3 images

- Draw the lewis structure of SO2 ( best resonance) and 2nd best resonance showing the shape and bond angles of each one, the 3D structure with polar bonds or bonds dipole of each one and the molecular polarity. Here is an example of what I want the answer to be like:Derive Lewis structures for the compounds below. Ethyl crotonate (For the purpose of drawing Lewis structures of compounds with more than one functional group, each group can be treated independently.)1. Please explain how an FTIR works 2. Why it's so important and useful for organic chemistry
- If the bond in the AB molecule is considered 100% ionic, the absolute value of the partial charge would be equal to the electron charge (1.602 x 10-19 C). If the distance between A and B atoms is taken as 10-10 m (1Å), the dipole moment between two elementary charges of opposite sign at atomic magnitude (1Å) distance between them is = 1.602x10-19 C x 10-10 m = 1.602 x 10- It is available in 29 Cm (or 4.8 x 10-18 esbcm). Dipole moment is given by debye (D) and 1D = 3.34x10-30 Cm (or 1D = 10-18 esbcm) (esb = electrostatic unit). 1. As can be seen, when the HCl covalent molecule is considered ionic and the bond length is 1.27x10-10 m, calculate the dipole moment. The experimental dipole moment of an HCl polar option is = 1.03 D. Calculate the value character of the H-Cl bond according to the bulletin. How many% covalent is the bond, how many% is ionic? SpecifyHow many LONE pairs of electron does ONE water molecule have? a. two b. three c. one d. none I need explanation in a research way. Please don't base the answer to any articles that can be seen in google instead base it with a research paper or any studies with authors. Book-supported is also okay.Calculate delta H for: a. 2 C2H2 (g) + 5 O2 (g) ---> 4 CO2 (g) + 2 H2O (g) Given: (Delta Hf in KJ/mol) [ C2H2 = +227, C02 = - 394. H2O = - 242 and O2 = O ]
- Write the lewis structures for the following three molecules and ions, including the dots for external electrons and lines for single, double, and triple bonds. I realize it is easy to look them up online but try them on your own first. I recommend writing them on paper, scan them, and submit them as a pdf or jpeg file. 1) BH2- 2) N2H2 3) CNH3Use VSEPR theory to predict the shape of this molecule (POC13) and indicate any deviation from the bond angle(s) expected from a regular arrangement of repulsion axes. Show all the steps took to arrive at answer 4 to five sentencesHow can we identify whether the compounds are ionic or covalent for part 1?
- 14) Although I3- is known, F3- is not known to occur. Draw the Lewis structures for these ins and explain why F3- does not form.provide the fllowing information including the lewis structure and the 3d sketch for NO2- and BrF3Put this from most polar for 1 and least 3 I keep asking on Bartleby and they keep getting it wrong wasting my questions to ask



