Q: The structure of methyl benzoate is shown in the figure below. In the El mass spectrum to methyl…
A: Mass spectra is an analytical technique that is used to analyse the mass to charge ratio of ions.
Q: A molecule produces an IR spectrum with key peaks at 3100 and 2100 cm-1. The mass spectrum has a…
A: IR and mass spectrometry are important tools for the determination of structures of the unknown…
Q: * In mass spectroscopy, intensity of each signal represents molecular mass abundance Relative…
A: Two multiple choice questions based on spectroscopy, which are to be accomplished.
Q: . Consider the mass spectrum of 2-bromopentane, which exhibits a molecular ion peak at m/z = 150…
A: A question based on mass spectrum that is to be accomplished.
Q: Select the most reasonable formula for the compounds with the following mass spectral data. M m/z…
A:
Q: what is the fragment produced at 137 M/Z (mass spectrum) ? Draw the structure and provide the…
A: In mass spectrum some molecular ion is characterized by their specific m/z peak. m/z =137 molecular…
Q: give the mass spectroscopy interpretation of the peaks Propan-1-amine / Propylamine
A: First , we will do the molecular ion fragments and then assign the peaks to the particular…
Q: 3 pts) Predict the number of signals expected and state their splitting pattern in the 1H NMR…
A:
Q: The mass spectrum of compound A shows the molecular ion at m/z 85, an M + 1 peak at m/z 86 of…
A: The corresponding molecular formula C5H11N contains either one pi bond or one ring.
Q: In a 300-MHz spectrometer, the protons in iodomethane absorb at a position 650 Hz downfield from…
A:
Q: Predict the masses and the structures of the most abundant fragments observed in the mass spectra of…
A: SOLUTION: Step 1: The most abundant fragment peaks of 4-methylpentan-2-ol are shown below: 1) First…
Q: Predict the masses and the structures of the most abundant fragments observed in the mass spectra of…
A: Fragmentation gives the resonance stabilized cations whenever possible. Loss of a small molecule is…
Q: Predict the masses and the structures of the most abundant fragments observed in the mass spectra of…
A: (a) The mass and the structure of the most abundant fragments observed in the mass spectra of the…
Q: Propylbenzene, C6H5CH2CH2CH3, and isopropyl benzene, C6H5CH(CH3)2, are constitutional isomers with…
A:
Q: How many peaks can be expected in the 13C spectrum of acetylsalicylic acid (shown below)?
A:
Q: Draw the structure of the compound shown in the mass spectra given its molecular formula C8H9NO.…
A: There are different types of fragmentation methods- 1- alpha cleavage 2- heterolytic cleavage 3-…
Q: What would distinguish the mass spectrum of 2,2-dimethylpropane from those of 2 methylbutane and…
A: Ans: E The base peak at 57 will distinguish the mass spectrum of 2,2-dimethylpropane from those of…
Q: Primary (1°) alcohols often show a peak in their mass spectra at m/z = 31. Suggest a structure for…
A: Given information, Primary alcohols often show peak in their mass spectra at m/z = 31.
Q: The base peak appears at m/z 105 for one of the following compounds and at m/z 119 for the other…
A: The base peak of m/z 105 for one of the compound among the three given compound and m/z 119 for the…
Q: Draw all four resonance forms of the fragment at m>z 73 in the mass spectrum of pentanoic acid
A:
Q: HC I| HC CH CH
A:
Q: Based on the mass spectrum shown below, 100 73 HO 80 A 58 20 100 10 20 30 40 50 100 60 70 m/z 80 90…
A:
Q: 7 Predict the masses and the structures of the most abundant fragments observed in the mass spectra…
A: Mass spectrometry- Mass spectrometry is an analytical technique which measures mass to charge (m/z)…
Q: Which of the following will produce an even M* and (M+2) peaks of equal intensity and a base peak of…
A: Mass spectrometry is an analytical tool used to identify different fragments in the molecule.
Q: A mass peak at m/z = 59 appears in the mass spectrum of an amide, C5H11NO. Draw the structure of a…
A: Mass spectrometry helps to determine the presence of the compounds in the given mixture. The m/z…
Q: The mass spectrum of an aldehyde shows a parent peak at m/z = 58 and a base peak at m/z 29. Propose…
A: Mass spectra helps in determination of the molecular formula and a possible structure.
Q: how many peaks would be expected from the ¹³C NMR of the compound?
A: We have to find out the number of 13c NMR peaks for the given compound.
Q: 2-Methyl-3-pentanol reacts with PCC to form compound F. () Draw the structure of F. (ii) In the mass…
A: Since you are posted with multiple sub-parts. As per the rule, I am answering first question only.…
Q: Does ethyl radical be detected in Mass Spectrometry as a peak at m/z = 29. True or False?
A:
Q: The mass spectrum of a ketone (C7H14O) exhibits a base peak at m/z = 57. Is the ketone most likely…
A: Base peak in carbonyl group is controlled by α-cleavage. While doing α-cleavage bigger group has to…
Q: Ater analyzing a mass spectrum you conclude that the molecule being analyzed has the molecular…
A: The possible isomer of the given molecular formula is as follows:
Q: Can the exact mass obtained in a high-resolution mass spectrum distinguish between two isomers such…
A: CH2CHCH2CH2CH2CH3 is n-hexane (CH3)2CHCH2CH2CH3 is 2-methylpentane
Q: The base peak in the mass spectrum of propanone (acetone) occurs at m/z 43. What cation does this…
A: Propanone is a compound containing a ketone group. Draw the structure of propanone.
Q: In the mass spectrum of the following compounds, which is the tallest—the peak at m/z = 57 or the…
A: Solution: a) part Draw the structure of molecular ion of 3-methyl pentane and follow two…
Q: Which of the following has the highest intensity of molecular ion peak in its mass spectrum? Select…
A: Mass spectrometry is an important tool for the determination of the molecular mass of the compound…
Q: The mass spectrum of t-butylcyclohexane is show below. Draw the molecular ion, M+, for…
A: A question based on Mass spectrum, which is to be accomplished.
Q: The mass spectrum of compound A shows the molecular ion at m/z 85, an M + 1 peak at m/z 86 of…
A: Based on nitrogen rule, even nominal mass is found for molecules that contains even number of…
Q: Mass Spectrum 43 4이 29 M+' = 88 2아 C4H8O2 40 80 120 160 200 240 280 m/e 8 8 8 % of base peak
A:
Q: Determining the Molecular Ions for an Alkyl Chloride What molecular ions will be present in a mass…
A: Different types of spectroscopy can be used to identify the structure of unknown compound. Some…
Q: b) Given the structure of the hydrocarbon below: CH3CH2CI i. How many peaks will this compound…
A: NMR is one of the spectroscopy method used for identification of compound.
Q: 2-Methylhexane shows an intense peak in the mass spectrum at m/z = 85, 57 and 43. Propose the…
A: We have to predict the structure for given fragments.
Q: Identify the hydrocarbon that has a molecular ion with an m/z value of 128, a base peak with an m/z…
A: Analyzing for rule 13: The mass spectrum rule of 13 allows possible molecular formula to be…
Q: Assigning Possible Structures to Fragments in a Mass Spectrum The mass spectrum of…
A: In mass spectroscopy, spectra spectra show so many peaks, the peak at right side is the molecular…
Q: Based on the mass spectrum shown below, 100 73 N° 80 A 40 58 20 100 10 20 30 40 50 60 70 m/z 80 90…
A: In the mass spectrum, the greatest m/z value is likely to be the molecular ion.…
Q: Which of the following has the highest intensity of mass fragment peak in its mass spectrum? Select…
A: more the stability of the fragment, more the intensity of the mass fragment peak in the mass…
Q: Identify the key fragment structures of 4'-Bromoacetophenone based on the peaks of its attached mass…
A:
Q: 2,3-Dimethylbutane and 2,2-dimethylbutane have the same molecular ion in the mass spectrum, but only…
A: a.
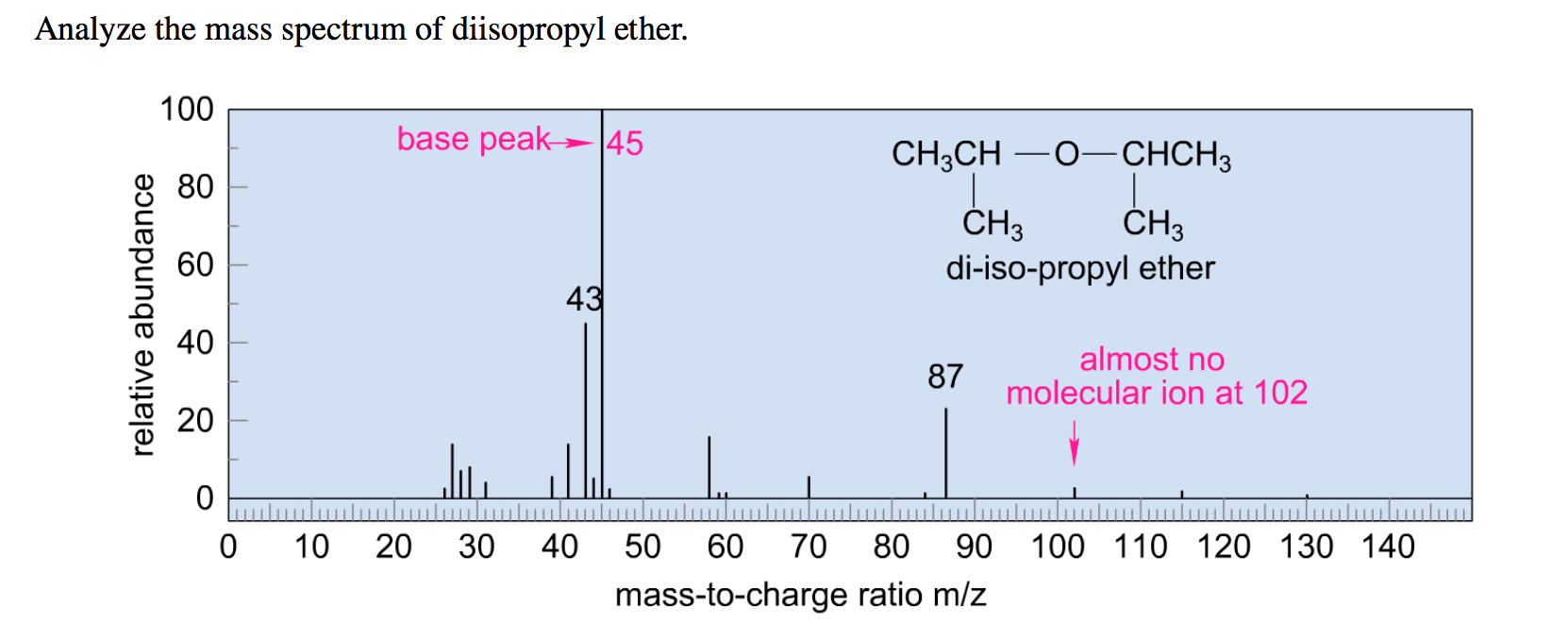
Identify the fragment giving rise to the peak at m/z 43.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- What does cross peak X tell you?What is the mass spectrometry of this compound ?Mass spectrometry is often used to both identify and quantify compounds. What is different between mass spectra with changing concentrations of a compound? How would you design a method (i.e. what would you need) to both identify and quantify a compound using mass spectrometry?

