Another mixture of PClB(g), Clafg), and PCIS(g) is at equilibrium at a different temperature in a different vessel. The system at equilibrium is represented by the following box on the left Then the volume of the vessel is decreased. The box in the middle represents the system before equilibrium is reestablished at the same temperature. Legend -PC, 8. -PCI, Original Equilibrium Immediately After Volume Change New Equilibrium d) In the box above on the right, draw an appropriate number of each type of molecule to represent a possible new equilibrium.
Another mixture of PClB(g), Clafg), and PCIS(g) is at equilibrium at a different temperature in a different vessel. The system at equilibrium is represented by the following box on the left Then the volume of the vessel is decreased. The box in the middle represents the system before equilibrium is reestablished at the same temperature. Legend -PC, 8. -PCI, Original Equilibrium Immediately After Volume Change New Equilibrium d) In the box above on the right, draw an appropriate number of each type of molecule to represent a possible new equilibrium.
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter18: Chemical Equilibrium
Section: Chapter Questions
Problem 54E: For the following system, K=4.86105 at 298K: HClO(aq)+F(aq)ClO(aq)+HF(aq) Assuming that you start...
Related questions
Question
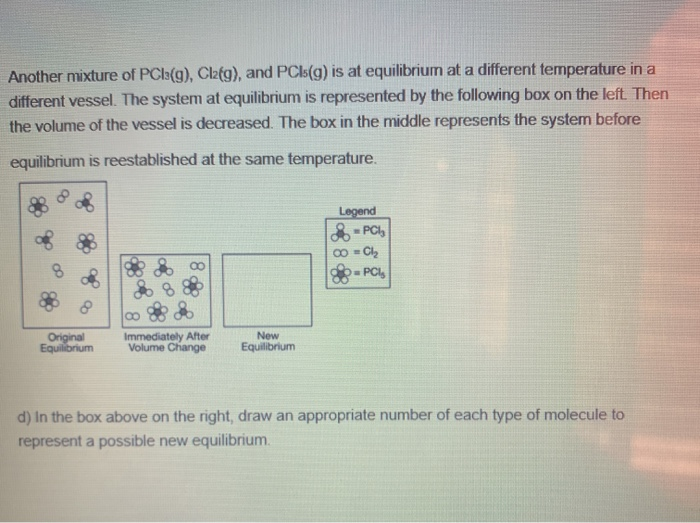
Transcribed Image Text:Another mixture of PCla(g), Clafg), and PCIS(g) is at equilibrium at a different temperature in a
different vessel. The system at equilibrium is represented by the following box on the left Then
the volume of the vessel is decreased. The box in the middle represents the system before
equilibrium is reestablished at the same temperature.
Legend
-PC
0 - Cl,
PC,
Original
Equilibrium
Immediately After
Volume Change
New
Equilibrium
d) In the box above on the right, draw an appropriate number of each type of molecule to
represent a possible new equilibrium.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning