Answer the following questions based in the scheme below: Determine the principle involved in the following conversion, either chemoselective, regioselective, or stereoselective. (iv) Write the synthetic steps, and provide all suitable reagents. OH obo H₂N H₂N
Answer the following questions based in the scheme below: Determine the principle involved in the following conversion, either chemoselective, regioselective, or stereoselective. (iv) Write the synthetic steps, and provide all suitable reagents. OH obo H₂N H₂N
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter20: Dienes, Conjugated Systems, And Pericyclic Reactions
Section: Chapter Questions
Problem 20.38P
Related questions
Question
Please solve this
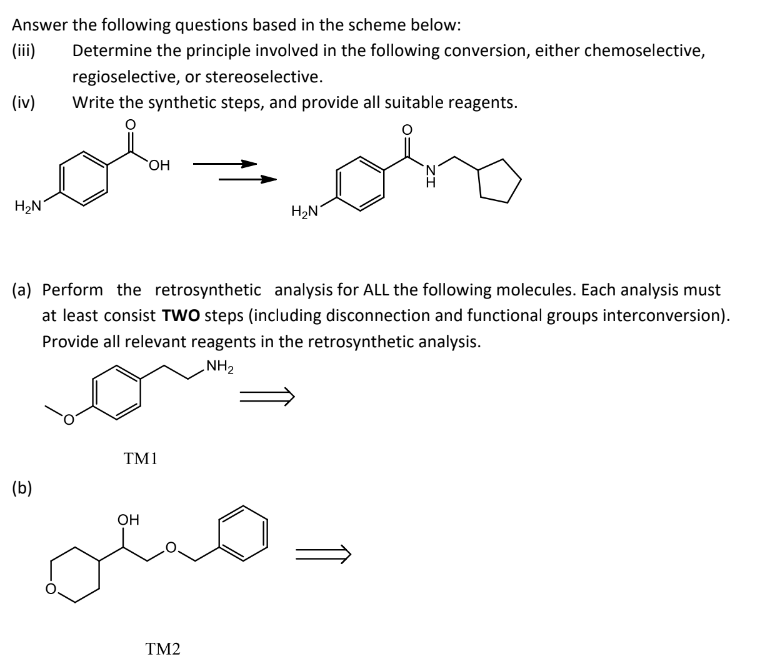
Transcribed Image Text:Answer the following questions based in the scheme below:
Determine the principle involved in the following conversion, either chemoselective,
regioselective, or stereoselective.
(iv)
Write the synthetic steps, and provide all suitable reagents.
OH
H₂N
H₂N
(a) Perform the retrosynthetic analysis for ALL the following molecules. Each analysis must
at least consist TWO steps (including disconnection and functional groups interconversion).
Provide all relevant reagents in the retrosynthetic analysis.
NH₂
TM1
(b)
OH
TM2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning