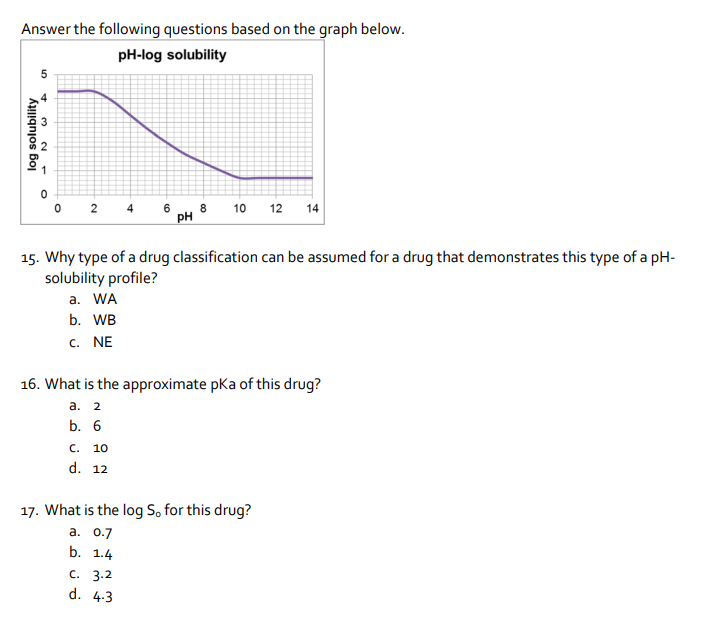
Answer the following questions based on the graph below. pH-log solubility 2 4 6 pH 10 12 14 15. Why type of a drug classification can be assumed for a drug that demonstrates this type of a pH- solubility profile? log solubility
Q: Determine the molar concentration of a Ca(OH)2 solution whose pOH is 3.47 at 25.0 °C. (the answer…
A: To calculate molar concentration of Ca(OH)2 , we would first calculate concentration of OH- using…
Q: Why do we titrate medications
A:
Q: to accurately determine the acidity of a solution how many types of litmus paper must be used?…
A: To identify: The indicator which is not classified as an universal indicator and is used to identify…
Q: Each row of the table below describes an aqueous solution at 25 °C. The second column of the table…
A: Different type of Solutions have different compositions and based on these compositions, the Acidic,…
Q: 3. Volume of NAOH at the equivalence point- 4. Half the volume of NaOH at the equivalence point---…
A: According to question, In question volume of NaOH is given and change in pH when NaOH is added to…
Q: Calculate the solubility of each of the following compounds in moles per liter. Ignore any acid-base…
A:
Q: Starting a solution( 15.0ml 0.200M CoCl2 (times) 6H2O+9.00ml 12.0M HCl) WHICH CHANGED THE COLOR TO…
A: When any reaction at equilibrium is disturbed then Le chatelier principle gives the direction of…
Q: An aqueous solution is prepared by dissolving 16.7 grams of Aluminum hydroxide in 645.0 mL solution.…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: The HNO3 in a 0.50M HNO3 solution is 100% dissociated. what are the molar concentrations of the…
A: Given : Concentration of HNO3 taken = 0.50 M And HNO3 is 100 % dissociated. The dissociation…
Q: acetylsalicylic acid + sodium hydroxide ---> sodium acetylsalicylate + water You have a sodium…
A: Given : Normality of NaOH i.e sodium hydroxide = 0.1231 N And the balanced reaction taking place is,…
Q: 1) Please define the following: Indicator Titration Neutralization Endpoint Conditioning…
A: To define Indicator, Titration, Neutralization, Endpoint, Conditioning, Standardization.
Q: Some chemical compounds are listed in the first column of the table below. Each compound is soluble…
A: The given compounds are ethyl amine, sodium perchlorate, sodium hydroxide, and perchloric acid.…
Q: Some chemical compounds are listed in the first column of the table below. Each compound is soluble…
A:
Q: Directions. Read the paragraph below and perform what is asked. The pH of the average soft drink is…
A: Soft drinks Normally packed in plastic, glass, or aluminum cans.
Q: What is the molarity of a 12.46 mL sulfuric acid solution if 18.22 mL of 0.100 M potassium hydroxide…
A: Stoichiometry is mainly based on the calculation of moles and volumes. These two values are used to…
Q: Some chemical compounds are listed in the first column of the table below. Each compound is soluble…
A: Answer On the basis of dissociation we pridict the ions and basicity…
Q: Some soluble compounds are listed in the table below. Complete the table by filling in the name or…
A: Ionic compounds are those compounds in which the constituent particles are ions held together by…
Q: Each row of the table below describes an aqueous solution at 25 The second column of the table shows…
A: pH = - log[H+] Therefore, more H+ releasing the compound lesser is its pH and hence more acidic the…
Q: It is used to deliver a small quantity of distilled water 2. Reagents whose label of containers are…
A: To write one word for the given sentences:
Q: Keff is defined as: 1- Static acid solubility 2- Weak base ionization constant 3- Constant effective…
A: A multiple choice question based on general chemistry that is to be accomplished.
Q: Calculate the concentration of 45.0 mL of a phosphoric acid solution that neutralized 20.8 mL of…
A: H₃PO₄ + 3NaOH → Na₃PO₄ + 3H₂O Volume of H3PO4 = 45 mL = 0.045 LVolume of NaOH = 20.8 mL = 0.0208 L…
Q: An ion with a positive charge is ...?
A: The Ion with positive charge is called cation
Q: Some soluble compounds are listed in the table below. Complete the table by filling in the name or…
A: Covalent compounds or molecular compounds are formed by sharing of electrons whereas ionic compounds…
Q: Carbon dioxide and water react to form bicarbonate ion and hydronium ion. Hyperventilation (rapid…
A:
Q: Use the graph below to answer the following questions. Provide a short reasoning for your answers.…
A: The question is based on the concept of titrations. we have to answer the questions asked.
Q: If an uknown concentration 25.00 mL HCl consumes 20 mL of 0.123 M NAOH to reach endpoint, calculate…
A: Given :- Volume of HCl solution = 25.00 mL Volume of NaOH solution = 20 mL Molar concentration of…
Q: 2. What does this imply for the size of the equilibrium constant for this reaction? CH;COOH(s)…
A: Given: Test tube 1 - benzoic acid + 2 mL water Test tube 2 - benzoic acid + 2 mL HNO3 (2 M) Test…
Q: 12. Derivatives of barbituric acid are soluble in solutions: A. Ammonia B. Sodium carbonate C.…
A: A question based on solutions that is to be accomplished.
Q: Fill in the necessary information. Choices for chemical test: -Water solubility test -Litmus paper…
A: Lead acetate test is used to identify the compounds containing sulfur in amino acids. Biuret test is…
Q: 1. Solubility Test Observations
A: Solubility tests can suggest the size and limit of a dark compound and the presence of crucial or…
Q: what mass of barium iodide is contained in 250ml of a barium iodide solution that has an iodide ion…
A: Molar mass of barium iodide=391.136 g/molBarium iodide solution that has Iodide ionconcentration=…
Q: Each row or the table below describes an aqueous solution at 25 °C. The second column of the table…
A: When neutral salt is added then no effect on pH. If base is added the pH increases
Q: Describe the blood hydrogen carbonate buffer system. What happens when blood acidity rises? Show the…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: A 0.1-L unbuffered solution needs the pH adjusted from 6.8 to 2. How many microliters of a 6 molar…
A:
Q: Some chemical compounds are listed in the first column of the table below. Each compound is soluble…
A: C2H5NH2 :-weak base HC2H3O2 :-weak acid
Q: NH2 NH3 + HCI A в
A: We have to determine the compound A and B is soluble in water or not
Q: The following items are potential problems someone might encounter while performing a titration. For…
A: Forgetting to rinse the burette with NaOH solution causes considerable effect on calculated…
Q: Four solutions of an acid dissolved in water are sketched below, as if under a microscope so…
A:
Q: 1.Volume of NaOH at end point VI= ml Calculation of pH ISHOW CALCULATION FROM THE GRAPH TATTACH…
A: Since you have asked a question with multiple subparts, we will answer only first 3 subparts for…
Q: 1.0L of 1.0M hydrochloric acid was spilled and needs to neutralized. How many grams of the solid…
A: The neutralisation reaction taking place is given as Hence we can see that 2 moles of HCl can be…
Q: In mixing two similar solutions but of different concentrations, the resulting solution has a…
A: Two solutions of similar solution with different concentrations are mixed.
Q: Each row of the table below describes an aqueous solution at 25 °C. The second column of the table…
A: A - Neutral and Basic on adding HCl pH gets lower as concentration oh H+ ions get Increase B-…
Q: Each row of the table below describes an aqueous solution at 25 °C. The second column of the table…
A:
Q: A 150 mL solution of NaOH is titrated with HI. Phenol red is the indicator, which is red at high pH…
A: Answer: For the pH scale, high pH indicates the basic solution and low pH indicates the acidic…
Q: Determine the molar concentration of each ion present in the solutions that result from each of the…
A: As per our guidelines we can only solve first three sub-parts. Please resubmit the other questions…
Q: A student is presented with the following data: pH meter reading 10.5 Concentration of H+ ions low…
A: Given details, The pH of the solution = 10.5, The hydrogen ion concentration = low in the solution,…
Q: Graph your data by plotting the pH (y-axis) versus the amount of citric acid solution added…
A:
Q: Some chemical compounds are listed in the first column of the table below. Each compound is soluble…
A: Ionic compounds : compounds formed by complete transfer of electrons and ions are involved in bond…
Q: The HCl in a 0.10M HCl is 100% dissociated. What are the molar concentrations of HCl, , and in the…
A: Given information: Concentration of HCl = 0.10 M Also, it is given that the solution is 100 %…
Step by step
Solved in 4 steps with 2 images

- The hydroxide ion has the formula OH−OH−. The solubility-product constants for three generic hydroxides are given here. Generic hydroxide KspKspK_sp XOHXOH 1.30×10−81.30×10−8 Y(OH)2Y(OH)2 3.40×10−103.40×10−10 Z(OH)3Z(OH)3 6.00×10−156.00×10−15 Use these values to answer the following questions. 1. The removal of an ion is sometimes considered to be complete when its concentration drops to 1.00×10−6 1.00×10−6 MM. What concentration of hydroxide would cause Y2+Y2+ to "completely" precipitate from a solution?An aqueous solution of hypochlorous acid is used a disinfectant in many sanitizing sprays and swimming pool cleaners. Use the dissociation constant of hypochlorous, HOCl. HOCl ---> H+ + OCl- For 3.0x10-3 M solution of hypochlorous acid, calculate molar concentrations of all three species and the percent dissociation of the acid.Is Ag2SO is added to water ,the salt will be?. Soulable or insoulable? Explain why?
- Q6 chapter 15 titration: multimedia activities Solve Asap pleaseCompute for the titratable acidity of vinegar sample if 6 ml of 1.0 N NaOH was used to titrate 25 ml of sample. Can this sample be used commercially if the standard is 4% titratable acidity?How will Le Chatlier’s Principle be used to? Cause an insoluble salt to become more soluble
- A) Write the Ksp expression for the sparingly soluble compound lead phosphate, Pb3(PO4)2. Ksp = If either the numerator or denominator is 1, please enter 1. b.) Write the Ksp expression for the sparingly soluble compound silver sulfate, Ag2SO4. Ksp = If either the numerator or denominator is 1, please enter 1.For PbCl2, Ksp = 0.0000137 What is the molar solubility of PbCl2.For a solution containing 0.050 M sodium butanoate (the sodium salt of butanoic acid, butanoic acid can be found in appendix G). Calculate the fraction of association A)0.011%B)1.84%C)5.25%D)3.09%E)0.008% Do not give handwriting solution.
- Solid silver acetate is slowly added to 50.0 mL of a 0.376 M ammonium chromate solution until the concentration of silver ion is 0.0386 M. The percent of chromate ion remaining in solution is _________ %. Solid magnesium chromate is slowly added to 150 mL of a potassium fluoride solution until the concentration of magnesium ion is 0.0269 M. The maximum amount of fluoride remaining in solution is ___________ M.What are the K1 and K2 (dissociation constants)? Given... Molarity NaOH: .3, Concentration of unknown: 31 g/L # mL NaOH to reach 1st inflection point: 25 (pH 4) # mL NaOH to reach 2nd inflection point: 50 (pH 11) pH @ 1st 1/2 equivalence point: 2.6Write the Ksp expression for the sparingly soluble compound silver sulfide, Ag2S. Ksp = If either the numerator or denominator is 1, please enter 1. --- Write the Ksp expression for the sparingly soluble compound silver iodide, AgI. If either the numerator or denominator is 1, please enter 1. Ksp =

