Q: For each chemical reaction below state which species is oxidized and which species is reduced. a) Na…
A: Oxidation is defined as the loss of electrons or oxidation may be defined as the chemical process in…
Q: 8H2S + 8Cl2 → S8 +16HCl Given the reaction above identify the A) Reactant reduced answer…
A: This is a redox reaction, 8 S-II - 16 e- → 8 S0 (oxidation) 16 Cl0 + 16 e- → 16 Cl-I…
Q: Hi, Which substance is oxidized in this reaction? 2Zn(s) + O2(g) ---->2ZnO(s) thanks,
A: The substance that is oxidized in this reaction is to be identified-
Q: Determine whether the following is an oxidation or reduction reaction. Just choose one or the other,…
A: This is not an oxidation as well as reduction reaction .For oxidation and reduction there should be…
Q: For each of the following reactions, identify the reactant that is oxidized and the reactant that is…
A: Note : As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost…
Q: 2. Identify the atom being oxidized and the atom being reduced in the following reactions: a) 2 Mg…
A: Oxidation - reduction reaction or redox reaction is defined as the reaction in which oxidation and…
Q: I) Zn + 2HCI --> ZnCl2 H2 II) NaOH + H2S04 -> Na2SO4 + H2O III) H20 --> H* + OH IV) 2H20 --> 2H2 +…
A: The question is based on the principle of redox reactions. we have to identify which of the given…
Q: Write complete balanaced equations. Assign oxidation numbers to all atoms. If the reaction is an…
A: Since, you have asked a question with multiple sub-parts, we will solve the first 3 sub-parts for…
Q: Ca + HBr Oxidation Reduction
A:
Q: Is the following reaction an oxidation, reduction, or neither? HO reduction oxidation neither
A:
Q: Consider the reaction: H2(g) + Cl2(g) → 2HCl(g) Hydrogen is getting ______________. Group of…
A: Increase in oxidation number = Oxidation Decrease in oxidation number = Reduction
Q: Which reaction is not an oxidation-reduction reaction? NADH + ½ O2 + H+ → NAD+ + H2O FAD + H2O →…
A: Oxidation-reduction (or redox reactions) involve addition and/or loss of oxygen and/or hydrogen.
Q: In the following reaction what is the undergoing oxidation? 4KO2(s) + 2CO2(g)--->2K2CO3(s) + 3O2(g)…
A: Applying concept of oxidation and reduction.
Q: What element is reduced in the following chemical reaction? Cu + 2H2SO4 → CUSO4 + SO2 + 2H2O Он O Cu…
A:
Q: In the following reaction, what species is undergoing oxidation (being oxidized)?: Ca + Br2 -->…
A: Oxidation is process which involves the increase in oxidation state of species…
Q: Identify the reactant that gets oxidized in the following reaction. 2 AgNO₃(aq) + CaI₂(aq) → 2…
A: Identify the reactant that gets oxidized in the following reaction. 2 AgNO₃(aq) + CaI₂(aq) → 2…
Q: For each reaction below, identify the atom oxidized, the atom reduced, the oxidizing agent, and the…
A: We have a. Mg + HCl →MgCl2 + H2 b. KMnO4 + KNO2 + H2SO4 → MnSO4 + H2O + KNO3 + K2SO4 c. KClO3 +…
Q: In the reaction below, which species is reduced? Ni(s) + Hg22+ (aq) Ni2+(aq) + 2Hg(1) O a. Hg(1) O…
A:
Q: Identify which reaction is an oxidation - reduction reaction? BaCl2 (aq) + 2KIO3 (aq) →…
A: Oxidation -reduction reaction is that in which oxidation and reduction take place. Oxidation is lose…
Q: Identify the reactant that gets reduced in the following reaction. BiO₃⁻(aq) + 2S₂O₃²⁻(aq) + 6H⁺(aq)…
A: The addition of electrons or decreases in the oxidation state is known as a reduction reaction and…
Q: Indicate if the process is oxidation, reduction, both, or neither Na → e1 + Na*1 S + 2 e1→ s2 -> H-H…
A: Fill in the blanks with suitable process ?
Q: In the following reaction, which is the oxidizing agent? AgNO2+ Cl₂ + 2KOH → AgNO3+2KCI + H₂O O…
A: Answer : Correct option is (b) Cl2 Explainination : oxidizing agent is a…
Q: Which of the following is being reduced? CO2 + H2 → 2CO + H2O OH O C
A:
Q: Determine the number of electrons lost or gained and identify as an oxidation or reduction reaction.…
A: A chemical reaction must be balanced in terms of mass and charge as well. Oxidation is the process…
Q: What is being reduced in this reaction? 2 Na(s) + 2 H20() → 2 N2OH(aq) + H2(g) The hydrogen in H20…
A: Correct option is A the Hydrogen in H2O is being reduced. Oxidation means addition of oxygen or…
Q: Assign oxidation numbers
A: The formal charge on an atom in a compound or an ion is called oxidation number of that atom.
Q: Which of the following transformations is an example of an oxidation? Br A "O., HO "O., C
A: In an oxidation reaction the oxidation state of the element increases. The oxidation state of the…
Q: From the chemical equation, what is being reduced? CuSO4(aq) + Zn(s) --> Cu(s) +ZnSO4 (aq)
A: An element whose positive oxidation number decreases during reaction is said to be reduced.
Q: For the reaction below, which species is being reduced? H26) + Cl2@ → 2 HCl@) O H216) Cl216) O HCle
A: To understand reducing agent ,we have to understand following terms . oxidation = Addition of…
Q: 1. Assign oxidation numbers to the following compounds a) KClO3 b) NO3 c) CH3CHO I attached my…
A:
Q: n the space provided, draw the oxidation and reduction products for the following: сно H OH HO H H…
A: Polyhydroxy aldehydes are the group of the compound that has aldehyde as the main functional group…
Q: In the following reaction, what is the suhstance reduced? Zn(s) + 2 HCI(aq) → ZnCl2 (aq) + H2 (g) OH…
A: If the oxidation no. decreases then this is called reduction.
Q: Which element is oxidized and which is reduced in the following reactions? N2(g) + 3H2(g) -->…
A: Redox reaction: A reaction in which both oxidation and reduction takes place and oxidation states of…
Q: Which element is reduced in the following reaction? Cl2(aq) + 2NaI(aq) →…
A: Since in Cl2 the oxidation state of Cl is 0 And in NaCl we have Cl as Cl- which has oxidation state…
Q: Determine whether the following is an oxidation or reduction reaction (just choose one, not both)…
A: Solution To understand electron-transfer reactions just like the one between metal metal and…
Q: Identify the strongest oxidizing agent (oxidant) Cl2 (E•red = +1.35827 V) Zn (E °red -0.7618 V) Br2…
A:
Q: M Gmail Youlube Maps RO LAB Connec.. Which of the following reactions would NOT be an…
A: A redox reaction is defined as a chemical reaction where a substance undergoes oxidation and another…
Q: Is the following reaction an oxidation, reduction, or neither? CH;CH3 CH;CH,Br oxidation reduction…
A: Carbon is more electronegative than hydrogen. Bromine is more electronegative than carbon.
Q: Is the reactant undergoing oxidation or reduction in each of the following incomplete chemical…
A: A Process is said to be oxidation if it involves: -Addition of oxygen to a substance. -Removal of…
Q: Identify the equations that show the oxidation of neutral atom A and the reduction of neutral atom…
A: Eight options from which oxidation reaction of A and reduction reaction of X is to be identified.
Q: A. Assign oxidation numbers to the following molecules. CH 3 OH C=0 CH エ ○ニ0= C
A: 1. There are 2 atoms of O and an atom of C in carbon dioxide. The oxidation number of O is -2 For C…
Q: Which of the following is an oxidation reaction? Select one: OH 1. LIAIH4. 2. H30* MCPBA OH H2 Pt…
A: The Answer of the question given below,
Q: The following skeletal oxidation-reduction reaction occurs under acidic conditions. Write the…
A:
Q: In the following oxidation-reduction reaction, what is being oxidized? C4H8 + 6 O2 → 4 CO2 + 4 H20 O…
A: Oxidation is a chemical process or reaction in which electrons will be ejected from a species.…
Q: Which of the following substances has been reduced? Ag + H2S Ag2S + H2 O Ag OA92S
A: Reduction :- In terms of oxidation number , reduction can be defined as the decrease in oxidation…
Q: The following skeletal oxidation-reduction reaction occurs under acidic conditions. Write the…
A: In a redox reaction, oxidation and reduction takes place side by side. Oxidation involves loss of…
Q: Which of the following can be oxidized to produce ketone: I. Me⁰ alcohol, II. 1⁰ alcohol, III. 2⁰…
A: Oxidation is the process of loss of electrons or gain of oxygen atom.
Q: In the following reaction, indicate the oxidation numbers for all elements on BOTH sides of the…
A: an species whose oxidation number is increased is said to be oxidized and whose oxidation number is…
Q: Which substance is reduced in the following reaction? NaOH + Li --> LiOH + Na Choose the right…
A:
Q: NEED HELP: Identify which reaction is an oxidation - reduction reaction? BaCl2 (aq) + 2KIO3 (aq)…
A:
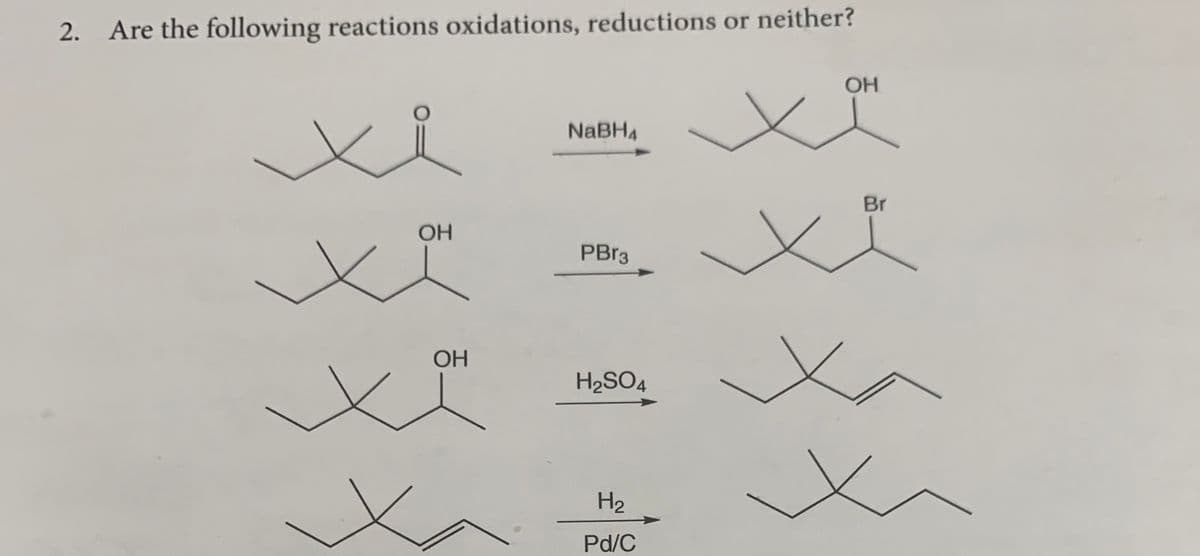
Can i get help with this problem please
Step by step
Solved in 5 steps with 4 images

- Label each of the following changes as an OXIDATION or a REDUCTIONa. H3AsO4 to Asb. O2 to H2Oc. Cr2O72- to CrO42-d. F- to F2e. MnO4- to Mn2+Label each of the following changes as an OXIDATION or a REDUCTIONc. Cr2O72- to CrO42-d. F- to F2e. MnO4- to Mn2+Classify the reaction below as an oxidation, a reduction, or neither.PhCO2H → PhCH2OHA) oxidationB) reductionC) neither
- 7. which of the following is a nonredox reaction? S8(s)+8O2(g)----> 8SO2(g) 2 Li(s) + Mg(NO3)2(aq) ----> 2 LiNO3(aq)+Mgs all the ractions 2PbSO4(s) +heat ---> 2pbSO3(s)+O2(g) H2SO4(aq)+2NH4OH----> @H2O(l) +(NH4)2SO4(aq)________ is reduced in the following reaction: Cr2O72- + 6S2O32- + 14H+ → 2Cr3+ + 3S4O62 + 7H2O O2- S4O62- Cr6+ H+ S2+For the following reactions how do you draw the reaction diagram?
- Which compound is the main product of the reaction in Figure 22? * Compound A Compound B Compound C Compound D None of the above.Determine the reaction type and the organic product(s) for each reaction:1. What is final product? 2. What are the 4 reactions & steps in mechanism (show product and mechanism steps)? 3. It is kinetic or thermodynamic?
- 2H2S(g) + SO2(g) <--> 3S(s) + 2H2O(g) Would this reaction be favored at a high or low temperature?Does the equlibrium favor the reactants or the products in each substitution reaction?Complete the balanced molecular chemicahal equation for the reaction below. If no reaction occurs, write NR after the reaction arrow. RB2CO(aq)+Fe(C2H3)2)2(aq)> Please explain Thanks.



