Arrange the following compounds in decreasing order of reactivity towards Nal in acetone: I. Methyl bromide II. Isopropyl bromide III. Ethyl bromide IV. t-butyl bromide A IV, I, II, I (B) IV, II, III, I I, II, II, IV D) I, II, III, IV
Q: When a dilute solution of acetaldehyde in D2O containing NaOD is shaken, explain why the methyl…
A: The given reaction is called as deuterium exchange enolization. Whenever a carbonyl compound is…
Q: 7. Compound EE, C4H3O shows a positive result (formation of yellow precipitate) when reacted with…
A: Answer:- this question is answered by using the simple concept of chemical reactions of organic…
Q: (a) How will you carry out the following conversions?(i) Acetylene to Acetic acid (ii) Toluene to…
A: In organic chemistry conversions play an important role to convert one functional group into another…
Q: Which of the following is expected to result in the following compound ? EņCuli CH;CH,MgBr OCH; 1)…
A:
Q: 7. Or the compounds B. D.
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: Identify the structure of the oxygen-containing compound with m/z= 74 (M+ ), 56 and 31 from the ones…
A: The correct structure is B i.e. 1-butanol. The molecular ion peak M+ is at m/z = 74 indicates that…
Q: b) 1. EtMgBr 2. H20
A: When Grignard reagent reacts with ester in presence of water then formation of alcohol takes place .…
Q: 3 CH3I NaOH solution NH2 [7] (11) (i) AGOH (replaces I with "OH) (ii) 180 °C [8] (14) (12) (13) 95%…
A:
Q: In synthesizing primary (1⁰) amines by alkylation of NH3, explain how we can prevent the formation…
A: Given statement is : In synthesizing primary (1⁰) amines by alkylation of NH3, explain how we can…
Q: MCQ 124: Hydrogen bromide have comparatively more resistant to decomposition than A. hydrogen iodide…
A:
Q: XS HI + H,0 IV II III O II O IV
A: In this question we have to tell the starting material of the given reaction and also its mechanism.…
Q: Please find attachment.
A: “Hey, since there are multiple questions posted, we will answer first question. If you want any…
Q: Organotin compounds play a significant role in diverse industrial applications. They have been used…
A:
Q: Alkene 14.1 was reacted with Br, as shown in Figure 14. A 1:1 ratio of products 14.2 and 14.3 was…
A: Bromonium cation is an three membered ring of the bromine where bromine is positively charged. And…
Q: Rank and explain your ranking of the following chlorides of group 15 in the order of increasing iv)…
A: Ionic compounds have positively charged cation and negatively charged anion due to which a dipole…
Q: M5 Explain the different stages of reaction, reaction mechanisms, conditions of reactions, (for the…
A: M5)A reaction takes place in different stages for example lets take a second order reaction it…
Q: (i) Provide the product for the reaction below. (ii) How would you know if the reaction was an SN1…
A:
Q: Arrange the following compounds in decreasing order of reactivity towards Nal in acetone: I. Methyl…
A:
Q: Heating the oxetane below to 560°C gives two products A and B (Scheme 2) A + CI Oxetane Scheme 2 The…
A:
Q: Choose the best reagents from the list provided below for carrying out the following conversions. 2.…
A:
Q: Q1/Do as required: 1-The role of H2SO4 in Preparation of Nitrobenzene. Explain 2- Preparation of…
A: Nitration of Benzene is carried forward in presence of Sulphuric acid and Nitric acid .
Q: (i) Provide the product for the reaction below. (ii) How would you know if the reaction was an SN1…
A:
Q: II (a) I= a, B-diketone, II = a, ß-diester, III = B-keto nitrile (b) I = B-diketone, II = B-diester,…
A: In this question, we will Identify the class of all three Compounds. You can see details Solution…
Q: The crocodile, which can remain under water without breathing for up to 1 h, drowns its…
A: When crocodile spends a lot pf time in underwater the concentration level of CO2 in its body…
Q: Provide a plausible arrow pushing mechanism for the reaction below. 1. ABuLi ŅME2 2. MgCl HO,C. 3.…
A:
Q: b) Complete the following reactions. i. `NH2 „NH2 SOCI, i. iii. LIAIH4
A: In organic reaction, the reactants are organic compounds, the reaction mechanism depends upon the…
Q: The nitration of furan leads preferentially to the formation of 2-nitrofuran rather than the 3-nitro…
A: The electrophile NO2⊕ can attack either at C3 or at C2 carbon of the furan ring. The electrophilic…
Q: 10. Metal-Alkyne complexes (COCL(PPH3),-III(CF3)) - Reactivity compared to olefins (Ni(C,H)3) -…
A:
Q: Give the major organic product for the reaction paying particular attention to regioselectivity. "D…
A: See major products below.
Q: CATION ELIMINATION TESTS ADDED REAGENT(S) OBSERVATIONS NaOH Dark brown ppt Excess NaOH Undissolved…
A: According to conditions given above , the possible cation should be Fe3+.
Q: Compound A has the formula CioH16 On catalytic hydrogenation over palladium, it reacts with only one…
A: Ozonolysis is an organic reaction shown by unsaturated alkenes and alkynes. The carbon-carbon…
Q: Question 4
A: Nucleophiles are those species which act as electron rich. These species offer electrons during any…
Q: 1. Phenylamine (CoHsNH2) is a brown liquid. 1 It is insoluble in water but soluble in dilute acids 2…
A: we have to determine correct statement about phenylamine.
Q: 4. The anisole derivative below undergoes a selective cross coupling reaction on only one of the two…
A:
Q: Provide the structure of the reactants necessary to prepare the following compound using the Wittig…
A: The structure of the reactant needed to prepare the given product using wittig reaction = ?
Q: NO2 но F II II IV
A: ->If electron donating group attached to the benzene ring then it increases the electron density…
Q: 6. Complete the following reaction scheme (а) hv Br2 (b) hv NBS ÇCL4, (a solvent) (c) H2 Pd/C
A:
Q: The reaction between Pl3, PSCl3 and Zn powder gives P3ls as one of the products. The sołution state…
A: This Question is based upon P31 NMR.
Q: Arrange the following in increasing reactivity to ethanolic AGNO3 L1-bromopentane II. 2-bromopentane…
A:
Q: 1) The ¹H NMR spectra of carvone and the epoxidation product(s) are extremely complicated, so we've…
A: 1H-NMR spectroscopy is used for identification of structure of the unknown compound. With help of…
Q: Op H,SO, NHCCH, + HNO, 0,N- -NHCH, + H,O CH,CO,H nitric acid (16 N) acetanilide p-nitroacetanilide…
A: The reaction taking place is given as,
Q: For the reaction given below, predict which product will be the major one and by what mechanism it…
A: In given reaction - Sovlent used is DMF which is aprotic and non polar. In reactant —OTs is very…
Q: Propose a reaction mechanism for P4S3 + I2 isomerization
A:
Q: When a dilute solution of acetaldehyde in D2O containing NaOD is shaken, explain why the methyl…
A: When a dilute solution of acetaldehyde in D2O containing NaOD is shaken, the methyl hydrogens are…
Q: 16. Furan undergoes ring opening upon treatment with sulfuric acid; it reacts almost explosively…
A: We have been asked to study the difference in reactivity between furan and furoic acid.
Q: Diels Alder Reaction of Tetraphenylcyclopentadienone Purpose of the Experiment: To successfully…
A: A question based on organic chemistry, which is to be accomplished.
Q: Arrange the following in decreasing reactivity to Nal in acetone 1. 1-bromopentane II.…
A: Arrange the following in decreasing reactivity to Nal in acetone I. 1-bromo pentane II. 2-bromo…
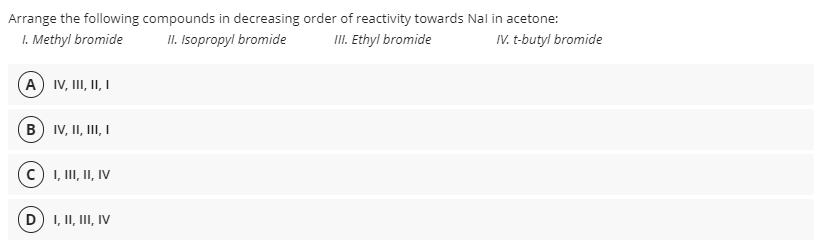

Step by step
Solved in 2 steps

- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4AI(C2H5)3 →3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d = 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3): d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257 L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides. One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(CzHs)3). 3SnCl4 + 4Al(C2H5)3 → 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.160 L of SnCl4 (d= 2.226 g/mL) was treated with 0.346 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). What is the theoretical yield in this experiment (mass of tetraethylstannane, Sn(C2H5)4)? If 0.257L of tetraethylstannane (d= 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?What happens when (write reactions involved) iv and v
- Organotin compounds play a significant role in diverse industrial applications. They have been used as plastic stabilizers and as pesticides or fungicides.One method used to prepare simple tetraalkylstannanes is the controlled direct reaction of liquid tin(IV) chloride with highly reactive trialkylaluminum compounds, such as liquid triethylaluminum (Al(C2H5)3). 3SnCl4 + 4Al(C2H5)3 3Sn(C2H5)4 + 4AlCl3 In one experiment, 0.230 L of SnCl4 (d = 2.226 g/mL) was treated with 0.396 L of triethylaluminum (Al(C2H5)3); d = 0.835 g/mL). If 0.335 L of tetraethylstannane (d = 1.187 g/mL) were actually isolated in this experiment, what was the percent yield?Describe how the product is purified. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°. The…When 37 is solvolyzed in trifluoroacetic acid approximately equimolar amounts of 38 and39 are obtained. Only 4 percent 40 is formed. Is this evidence for or against bridging byhydrogen?
- Provide details about the reaction workup. 4,4'-DIBROMOBIPHENYL [Biphenyl, 4,4'-dibromo-] Submitted by Robert E. Buckles and Norris G. Wheeler1. Checked by R. S. Schreiber, Wm. Bradley Reid, Jr., and Robert W. Jackson. 1. Procedure In a 15-cm. evaporating dish is placed 15.4 g. (0.10 mole) of finely powdered biphenyl (Note 1). The dish is set on a porcelain rack in a 30-cm. desiccator with a 10-cm. evaporating dish under the rack containing 39 g. (12 ml., 0.24 mole) of bromine. The desiccator is closed, but a very small opening is provided for the escape of hydrogen bromide (Note 2). The biphenyl is left in contact with the bromine vapor for 8 hours (or overnight). The orange solid is then removed from the desiccator and allowed to stand in the air under a hood for at least 4 hours (Note 3). At this point, the product weighs about 30 g. and has a melting point in the neighborhood of 152°. The crude 4,4'-dibromobiphenyl is dissolved in 75 ml. of benzene, filtered, and cooled to 15°.…In an experiment, triphenylmethanol is prepared using the Grignard reaction. Reaction of bromobenzene with magnesium in ether produces phenylmagnesium bromide. This Grignard reagent then reacts with methyl benzoate to produce the corresponding alkoxide. Reaction of the alkoxide with aqueous acid then produces the alcohol. Give a plausible, three dimensional structure for the complex RMgBr-2(C2H5)2O. How do you think the ether molecules are bonded to the Grignard reagent?Explain why eugenol readily dissolves in dichloromethane but salts don't
- Provide the necessary product(s) (i), arrows (ii)and charges (iii)..771g of acetominophen reacts with 1.0 eqivalent of 1.5M KOH and 1.2 equivalents of pure liquid n-butyl bromide in ethanol solvent to give .901g of the willimason ether product a) calculate the volume of the base and the volume of the SN2 electrophile needed for the reaction needed for the reaction using the equivalents given above b) caclulate the percent yield of the reactionWhat happens when benzaldehyde reacts with sodium bisulfite? What happens when acetone reacts with sodium bisulfite? Explain and include a reference video or study.

