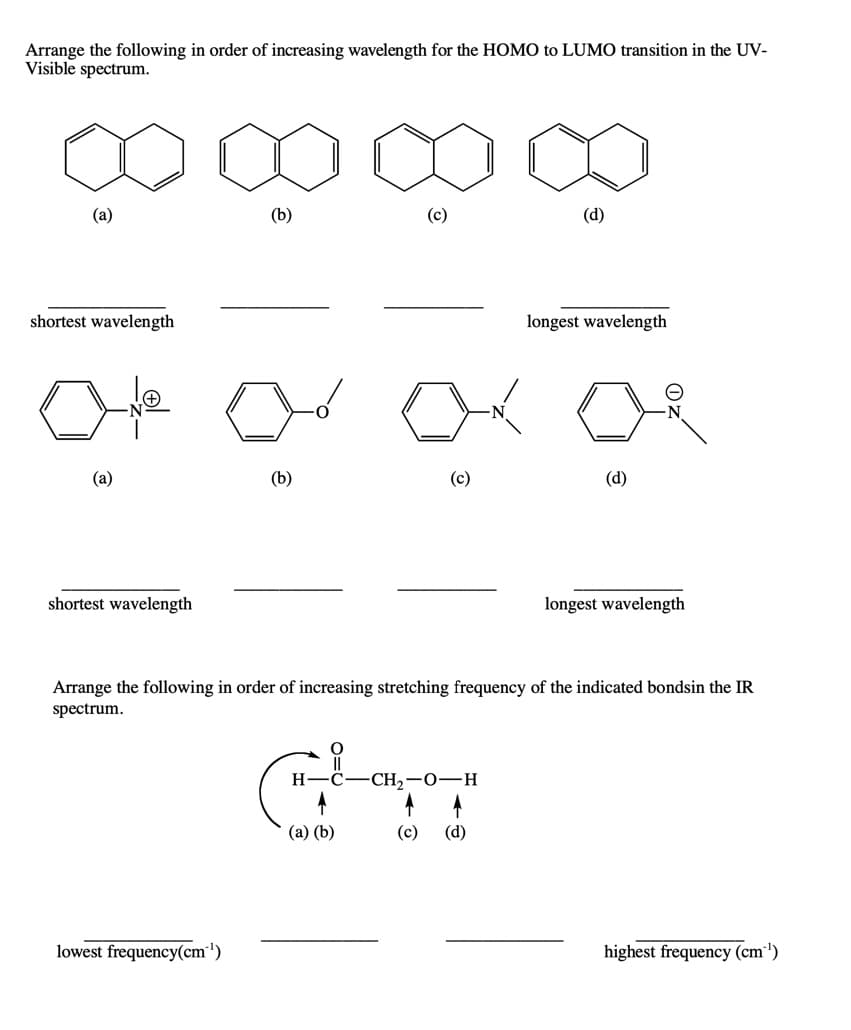
Arrange the following in order of increasing wavelength for the HOMO to LUMO transition in the UV- Visible spectrum. (a) (b) (c) (d) shortest wavelength longest wavelength (a) (b) (c) (d) shortest wavelength longest wavelength Arrange the following in order of increasing stretching frequency of the indicated bondsin the IR spectrum. H C- -CH2-0–H (a) (b) (c) (d) lowest frequency(cm') highest frequency (cm')
Electronic Transitions and Spectroscopy
The term “electronic” connotes electron, and the term “transition” implies transformation. In a molecule, the electrons move from a lower to a higher energy state due to excitation. The two energy states, the ground state and the excited state are the lowest and the highest energy states, respectively. An energy change is observed with this transition, which depicts the various data related to the molecule.
Photoelectron Spectroscopy
Photoelectron spectroscopy (PES) is a part of experimental chemistry. It is a technique used in laboratories that involves projecting intense beams of radiation on a sample element. In response, the element ejects electrons for which the relative energies are measured.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images









