As shown in the figure, a container with a moveable piston and containing a monatomic ideal gas in an initial state A undergoes an isovolumetric, then an isothermal, and finally an isobaric process to complete the cycle. P (atm) B V (L) When the gas is in the initial state, the volume is 6.00 L, the pressure is 2.00 atm, and the temperature is 200 K. The gas pressure of 5 times the initial value (state B). The gas is then allowed to expand isothermally to some new volume (state C). Finally it is compressed isobarically to its initial state. (Due to the nature of this problem, do not use rounded intermediate values in your calculations-including answers submitted in WebAssign.) first warmed at constant volume to a (a) Find the number of moles of the gas. .73 moles (b) Find the temperature of the gas at state B (in K). 1000 (c) Find the temperature of the gas at state C (in K). 1000 K (d) Find the volume of the gas at state C (in L). (e) Determine values (in kJ) for Q, W, and AEint for the process A → B. kJ W = kJ ДЕ, int kJ
As shown in the figure, a container with a moveable piston and containing a monatomic ideal gas in an initial state A undergoes an isovolumetric, then an isothermal, and finally an isobaric process to complete the cycle. P (atm) B V (L) When the gas is in the initial state, the volume is 6.00 L, the pressure is 2.00 atm, and the temperature is 200 K. The gas pressure of 5 times the initial value (state B). The gas is then allowed to expand isothermally to some new volume (state C). Finally it is compressed isobarically to its initial state. (Due to the nature of this problem, do not use rounded intermediate values in your calculations-including answers submitted in WebAssign.) first warmed at constant volume to a (a) Find the number of moles of the gas. .73 moles (b) Find the temperature of the gas at state B (in K). 1000 (c) Find the temperature of the gas at state C (in K). 1000 K (d) Find the volume of the gas at state C (in L). (e) Determine values (in kJ) for Q, W, and AEint for the process A → B. kJ W = kJ ДЕ, int kJ
Chapter2: The Kinetic Theory Of Gases
Section: Chapter Questions
Problem 43P: The product of the pressure and volume of a sample of hydrogen gas at 0.00 is 80.0 J. (a) How many...
Related questions
Question
I need help on d-f
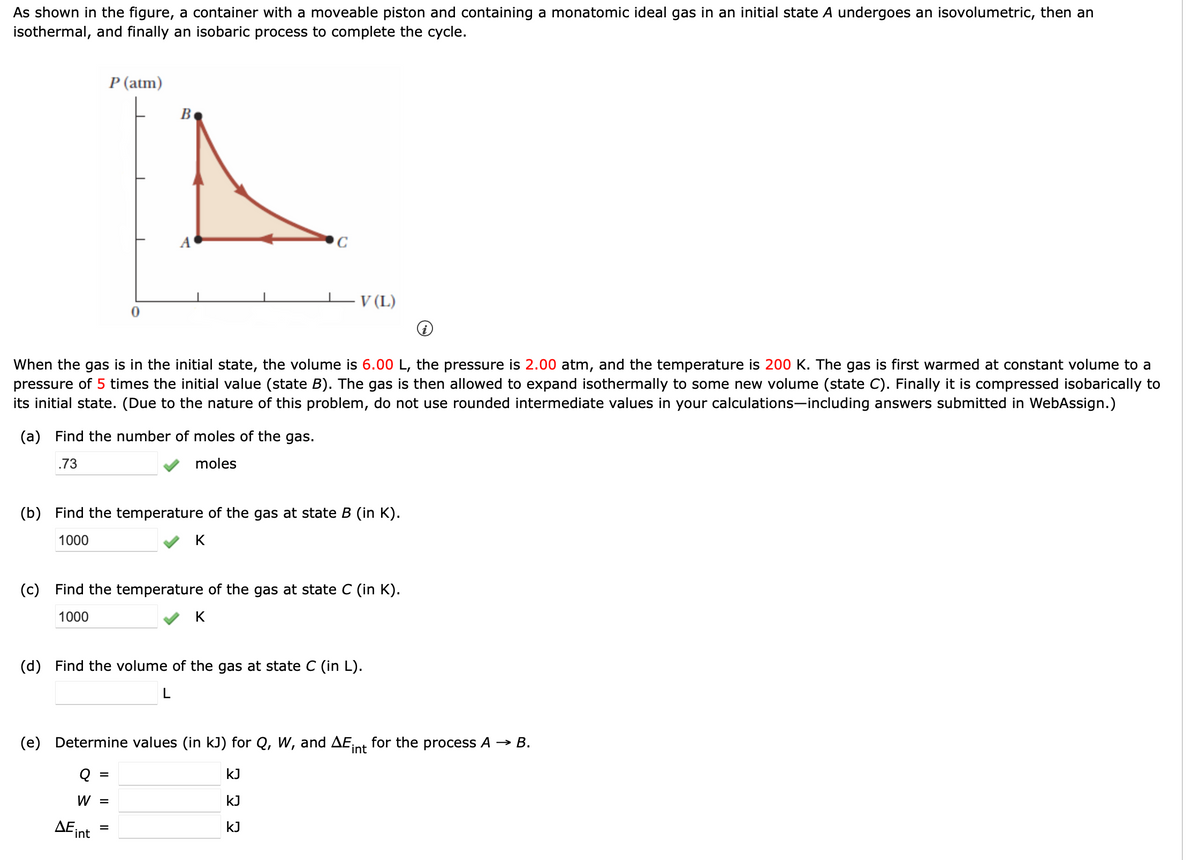
Transcribed Image Text:As shown in the figure, a container with a moveable piston and containing a monatomic ideal gas in an initial state A undergoes an isovolumetric, then an
isothermal, and finally an isobaric process to complete the cycle.
P (atm)
В
A
C
V (L)
When the gas is in the initial state, the volume is 6.00 L, the pressure is 2.00 atm, and the temperature is 200 K. The gas is first warmed at constant volume to a
pressure of 5 times the initial value (state B). The gas is then allowed to expand isothermally to some new volume (state C). Finally it is compressed isobarically to
its initial state. (Due to the nature of this problem, do not use rounded intermediate values in your calculations-including answers submitted in WebAssign.)
(a) Find the number of moles of the gas.
.73
moles
(b) Find the temperature of the gas at state B (in K).
1000
K
(c) Find the temperature of the gas at state C (in K).
1000
K
(d) Find the volume of the gas at state C (in L).
(e) Determine values (in kJ) for Q, W, and AEint for the process A → B.
Q
kJ
%3D
W =
kJ
ΔΕ,
kJ
int

Transcribed Image Text:(d) Find the volume of the gas at state C (in L).
(e) Determine values (in kJ) for Q, W, and AE for the process A → B.
int
kJ
W =
kJ
ΔΕ.
int
kJ
%3D
(f) Determine values (in kJ) for Q, W, and AEnt for the
ess B → C.
Q =
kJ
W =
kJ
ДЕ.
int
kJ
(g) Determine values (in kJ) for Q, W, and AEint
for the process C → A.
Q =
kJ
W =
kJ
AE:
int
kJ
%D
(h) Determine values (in kJ) for Q, W, and AEnt for the complete cycle A → B → C → A.
kJ
W =
kJ
ΔΕ.
Fint
kJ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

