4 moles of an ideal monatomic gas undergoes the three process cycle as depicted in the figure below. The cycle is composed of an isobaric, isovolumetric, and isotherma process. P. P. ►V V. V. For the given parameters p,=P,=9-104 Pa and V,=1.1 m³, V,= V=0.55 m3 calculate the following. (Take the universal gas constant as R=8.314 J.mol1.K-1) a) The pressurep. Pa
4 moles of an ideal monatomic gas undergoes the three process cycle as depicted in the figure below. The cycle is composed of an isobaric, isovolumetric, and isotherma process. P. P. ►V V. V. For the given parameters p,=P,=9-104 Pa and V,=1.1 m³, V,= V=0.55 m3 calculate the following. (Take the universal gas constant as R=8.314 J.mol1.K-1) a) The pressurep. Pa
Principles of Physics: A Calculus-Based Text
5th Edition
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Raymond A. Serway, John W. Jewett
Chapter18: Heat Engines, Entropy, And The Second Law Of Thermodynamics
Section: Chapter Questions
Problem 61P
Related questions
Question
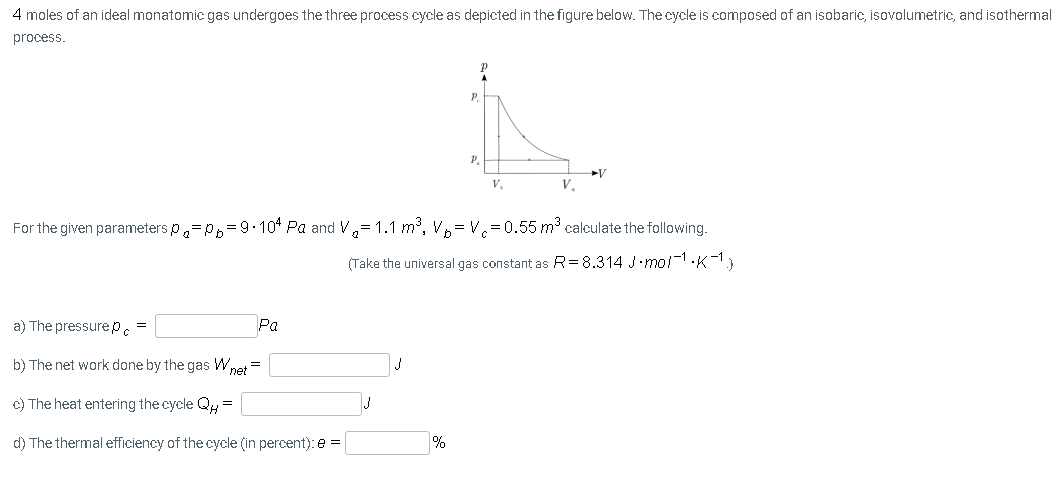
Transcribed Image Text:4 moles of an ideal monatomic gas undergoes the three process cycle as depicted in the figure below. The cycle is composed of an isobaric, isovolumetric, and isothermal
process.
P.
P.
V
V.
For the given parameters p,=p,=9.10* Pa and V,= 1.1 m3, V,= V.=0.55 m3 calculate the following.
(Take the universal gas constant as R= 8.314 J.mol-1.K-1
a) The pressure pc =
Pa
b) The net work done by the gas Wnet =
c) The heat entering the cycle QH =
d) The thermal efficiency of the cycle (in percent): e =
%
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning


Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning

Physics for Scientists and Engineers: Foundations…
Physics
ISBN:
9781133939146
Author:
Katz, Debora M.
Publisher:
Cengage Learning
