Q: 9. The following is the balanced equation for the combustion of butanol in oxygen: CAH9OH@ + 6 O2 →…
A: The balanced combustion reaction for butanal is given below - C4H9OH + 6O2 ---> 4CO4 + 5H2O…
Q: Q1/ How many molar masses are contained in 1- 5.76 g of KI ?( Atomic mass K=39, l=127) 2-4.00 liter…
A:
Q: 2C2H6 + 7O2 = 4CO2 + 6H2O If we instead had the combustion of propane, C3H8, how many grams of water…
A: Combustion means burning of the substance in presence of air to produce water and carbon dioxide.…
Q: over 2 billion pounds per year. Acrylonitrile, the building block for polyacrylonitrile fibers and a…
A:
Q: professor Scimemi has accepted you as a Master’s student and you are involved in a project that…
A: You need to make 2.5 mM KCl. The formula for weighing is as follows: Amount of KCl (g) = [Molarity…
Q: Fingernail–polish remover is primarily acetone (C3H6O).How many acetone molecules are in a bottle of…
A: Given data contains, Volume of acetone is 325 mL. Density of acetone is 0.788 g/cm3.
Q: by adding water to calcium carbide (CaC2). CaC2(s) + 2H,O(1) → C2H2(g) + Ca(OH),(aq) How many grams…
A: Introduction : Acetylene is produced by the reaction of calcium carbide with water, it is made from…
Q: NH3(aq) + CO2(aq) → CH,N20(aq) + H20(1) n an industrial synthesis of urea, a chemist combines 32.1…
A: Given that, mass of ammonia, and mass of Co2. And limiting reagent is asked.
Q: How many equivalents of PO43 are there in 6.00 mol of Mg3(PO4)2 ?
A:
Q: Ibuprofen, the anti-inflammatory drug in Advil, has the formula C13H18O2 What is the empirical…
A: Ibuprofen is an anti-inflammatory drug
Q: What is the number of molecules in 319g of Li2SO4? (Take NA = 6 x1023)
A: The question is based on mole concept. We have to determine number of molecules present in the…
Q: How many molecules of ascorbic acid (vitamin C: C6H8O6)are in a 50 mg tablet? (need molar mass)
A: Given data contains, Mass of tablet is 50 mg. The molar mass of ascorbic acid (C6H8O6) is calculated…
Q: How many liters of ethylene glycol antifreeze (C;H;O2) would you add to your car radiator containing…
A: Given data: Volume of water = 15.0 L Density of ethylene glycol = 1.1 g/mL
Q: The green color of leaves is due to thepresence of chlorophyll, C 55 H 72 O 5 N 4 Mg. A fresh…
A: Chlorophyll is a green pigment that absorbs sunlight and helps plants absorbs energy and utilize…
Q: Compound "X" has a density of 1.71 g/L at 755 mm Hg and 25 °C. What is the molar mass of compound…
A: Density = 1.71 g/L Pressure = 755 mm Hg = 0.993 atm ( 1 atm = 760 mmHg) Temperature = 25 °C =…
Q: For the balanced chemical reaction 4Al + 3O2 → 2Al2O3 how many grams of Al2O3 can be made by…
A:
Q: Prevacid is used to treat gastroesophageal reflux disease (GERD).Thechemical formula of Prevacid is…
A: The question is based on mole concept. we have to calculate the mass of fluorine atoms of carbon.…
Q: a) Calculate the molar mass of sucrose, C12H22O11.(b) How many moles of sucrose are present in:…
A: a) Molecular formula of sucrose is C12H22O11. Molar mass of carbon is 12.0107 g.mol-1. Molar mass of…
Q: Ethyne (C2H2), also known as acetylene, can be synthesized by reacting calcium carbide (CaC2) with…
A: The given reaction is : The mass of CaC2 = 56.0 g The mass of H2O = 75.0 g The molar mass of CaC2…
Q: 1.) What is the molecular formula of a compound with the empirical formula CH and a formula mass of…
A: 1 since , Empirical formula = CH therefore , Empirical formula mass = 12+1=13 amusince ,…
Q: www-awn.aleks.com Significant Figures Counter Sign out Sign out My Courses-Blackboard...…
A: Number of moles of phosphoric acid is calculated as given below,
Q: Equivalent weight of Al(OH)3 is а. 26 g O b. 27 g С. 78 д O d. 39 g
A:
Q: What number should be placed in the blank in order for this equation to follow the Law of…
A:
Q: What is the molecular formula of a compound with the empirical formula CH2 and a formula mass of…
A:
Q: C6H6(1) + O2(g) → H20(g) + CO2(g)
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: The chemical formula for propane is: CH;CH,CH3 Calculate the molar mass of propane. Round your…
A: Molar mass is the sum of atomic masses of each atom present in a molecule. Atomic mass of C= 12…
Q: Ethyne (C2H2), also known as acetylene, can be synthesized by reacting calcium carbide (CaC2) with…
A:
Q: The formula for lactose is C12H22O11, and the monohydrate is C12H22O11H2O. What is the percent by…
A:
Q: During an experiment you extract 112 mg of caffeine from a 16g sample of coffee. What is the mass…
A: A) Given: The mass of caffeine in coffee = 112 mg = 0.112 g ( Since 1 mg = 0.001 g) The mass of…
Q: The chemical formula for octane is: CH; (CH,),CH; 9. Calculate the molar mass of octane. Round your…
A: Octane is saturated hydrocarbon compound which is a component of gasoline i.e pertrol. It is…
Q: The molecular formula of aspirin is C9H8O4. How many aspirin molecules are present in one…
A:
Q: A chemist needs 1.28 g of sodium hypochlorite, NaOCl(s), for an experiment, but she only has sodium…
A: The mass of a compound can be determined by the molar mass of compound and the respective molar mass…
Q: The chemical formula for dimethyl ether is: CH;OCH3 Calculate the molar mass of dimethyl ether.…
A: The chemical formula of dimethyl ether states that one diethyl ether molecule contains two carbon…
Q: A lawn fertilizer is rated as 4.40% nitrogen, meaning 4.40 g of N in 100g of fertilizer. The…
A: Answer: This is a question based on stoichiometric calculation where moles of nitrogen should be…
Q: A student finds that they have 0.2545 g of NH4Cl, 1.1502 g of NaCl, and 1.0181 g of SiO2. Calculate…
A: Given mass of NH4Cl = 0.2545g mass of NaCl = 1.1502 g mass of SiO2 = 1.0181 g Initial mass if…
Q: C6H1206 + NH3 +1.5 O2 →C;H9NO4+ CO2 + 3H20 (glucose) (glutamic acid) a) If 18 grams of glutamic acid…
A: First of all convert the mass of given compound and then convert it to the mole by using formula :…
Q: A major component of honey is fructose (C6H12O6) with a molar mass of 180.16 g/mol. A tablespoon…
A:
Q: The chemical formula for t-butanol is: (CH;),COH Calculate the molar mass of t-butanol. Round your…
A: Molar mass is the sum of the atomic masses of all the atoms present in a molecule. Atomic mass of C=…
Q: True or False? One mole of molecular O2 = 6.02 x 1023 O2 molecules. Mass of O2 in 100g of CO2 =…
A:
Q: Aspirin can be prepared from salicylic acid (C-H,O3 ), which has a molar mass of 138.12 g/mol, and…
A:
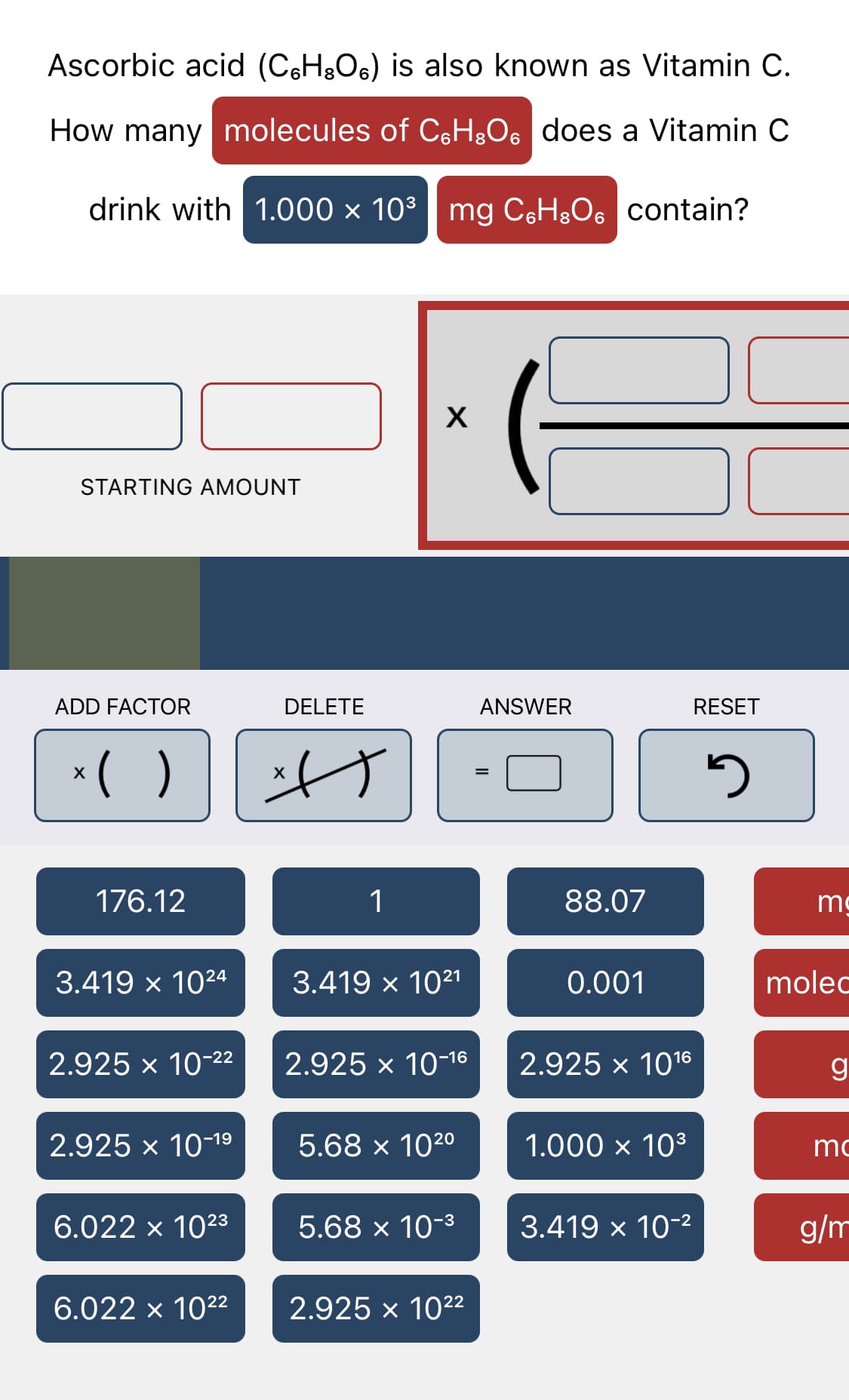
Q: Ascorbic acid (CsH&Os) is also known as Vitamin C. What quantity in molecules of CH&Os does a…
A: we know, moles is defined as the mass divided by the molar mass => moles = mass / molar mass…
Q: A fictional cubed-shaped bacterium, Bacterius cubis, occupies a volume of 2.8 femtoliters. This…
A:
Q: 13) If a 25 kg LPG tank is 30% butane (C4H10) , how many molecules of butane are contained in the…
A:
Q: Aspirin can be prepared from salicylic acid (C7H6O3 ), which has a molar mass of 138.12 g/mol, and…
A:
Q: Ascorbic acid (C&H$O6) is also known as Vitamin C. What quantity in molecules of C6H&O6 does a…
A:
Q: If 7.700000000000000 × 10²⁵ molecules of CO₂ are produced in a combustion reaction, how many liters…
A:
Q: Convert 3.104g CoCl•6H2O to moles
A: Given: Mass of CoCl.6H2O=3.104 g The molar mass of CoCl.6H2O can be calculated by using the atomic…
Q: A high-performance heater that burns propane, C3H8(g), is adjusted so that 100.0 g of O2(g) enters…
A: The number of moles of oxygen (O2) present (nO) in 100.0 g of oxygen gas is calculated using…
Q: What is the mass of a drop of methanol containing 2.90x1022 CH3OH molecules
A:
Q: C3H8 + 5O2 --> 3CO2 + 4H2O How many…
A: To calculate grams of oxygen required, we would use dimensional analysis method . For this we would…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Oceanic uptake of carbon dioxide is thus described:CO2 (g) + H2O ⇔ H2CO3, K = [H2CO3]/PCO2 = 3 x 10-2 M atm-1 H2CO3 ⇔ HCO3- + H+, K = [HCO3-][H+]/[H2CO3] = 9 x 10-7 moles/LHCO3- ⇔ CO32 - + H+, K = [CO32 -][H+]/[HCO3-] = 7 x 10-10 moles/LCharge balance equation:[H+] = [OH-] + [HCO3-] + 2[CO32 ] If the CO2 concentration in the atmosphere is 300 ppm, what is the pH of the ocean?Chemistry In an analysis of the content of carbohydrate present in a glycoprotein, the following results were found: 12.6, 11.9, 13.0, 12.7 and 12.5 g of carbohydrate per 100 g of protein. Taking into account that σ is unknown, the confidence interval for the average value at a 90% confidence level of the carbohydrate content is: Select one:to. 12.5 ± 0.4b. 12.5 ± 0.2c. 12.5 ± 0.5d. 12.5 ± 0.3If a bag of fertilizer were labeled as containing 35% K2O, a. What is the analysis when expressed as %K? b. Assume the bag is labeled as 150% P, calculate the percentage P2O5 in the bag.
- Consider the following data concerning the equation: H2O2 + 3I– + 2H+ → I3– + 2H2O [H2O2] [I–] [H+] rate I 0.100 M 5.00 × 10–4 M 1.00 × 10–2 M 0.137 M/sec II. 0.100 M 1.00 × 10–3 M 1.00 × 10–2 M 0.268 M/sec III. 0.200 M 1.00 × 10–3 M 1.00 × 10–2 M 0.542 M/sec IV. 0.400 M 1.00 × 10–3 M 2.00 × 10–2 M 1.084 M/sec The rate law for this reaction is a. rate = k[H2O2][I–][H+] b. rate = k[H2O2][H+] c. rate = k[H2O2]2[I–]2[H+]2 d. None of these e. rate = k[H2O2][I–] f. rate = k[I–][H+]Consider the following data concerning the equation: H2O2 + 3I– + 2H+ → I3– + 2H2O [H2O2] [I–] [H+] rate I 0.100 M 5.00 × 10–4 M 1.00 × 10–2 M 0.137 M/sec II. 0.100 M 1.00 × 10–3 M 1.00 × 10–2 M 0.268 M/sec III. 0.200 M 1.00 × 10–3 M 1.00 × 10–2 M 0.542 M/sec IV. 0.400 M 1.00 × 10–3 M 2.00 × 10–2 M 1.084 M/sec The rate law for this reaction is a. rate = k[H2O2]2[I–]2[H+]2b. rate = k[I–]c. None of thesed. rate = k[I–][H+]e. rate = k[H2O2][I–][H+]f. rate = k[H2O2][H+]Consider the following data concerning the equation: H2O2 + 3I– + 2H+ → I3– + 2H2O [H2O2][I–][H+]rate I 0.100 M 5.00 × 10–4 M 1.00 × 10–2 M 0.137 M/sec II. 0.100 M 1.00 × 10–3 M 1.00 × 10–2 M 0.268 M/sec III. 0.200 M 1.00 × 10–3 M 1.00 × 10–2 M 0.542 M/sec IV. 0.400 M 1.00 × 10–3 M 2.00 × 10–2 M 1.084 M/sec a. The rate law for this reaction is a. rate = k[H2O2][I–][H+] b. rate = k[H2O2]2[I–]2[H+]2 c. rate = k[I–][H+] d. rate = k[H2O2][H+] e. rate = k[H2O2][I–]
- Acetaminophen, a popular drug taken as pain reliever and fever reducer, is produced together with acetic acid from the reaction of 3.05 g 4-aminophenol and 4.1 ml of acetic anhydride. Acetaminophen was extracted at 60% yield. Density of acetic anhydride at 20 C, 1.08 g/ml. Calculate the actual no. of grams of acetaminophen produced. [Determine L.R., E.R.]You have a lot of paperwork laying around. A ream of stacked paper is 500 sheets and is 2.0 inches thick. Let's say you had a mole of paper. (1 mole of paper = 6.022 x 1023 sheets of paper) and it was all stacked up in 10 stacks of equal height, each containing a tenth of a mole of sheets. The ten stacks sit one on top of the other. How high are they? (Hint: find the height of a stack of containing 0.1 mole of paper. USE DIMENSIONAL ANALYSIS!) Here are are some conversions you might need: 2.54 cm = 1 in; around 9.46 x 1015 meters / light yr and 3.26 light yr / parsec.2b) What is the primary consideration?
- Nagyung wanted to determine the heat of dissolution for potassium chlorate (KClO3, MW: 122.55 g/mol) in water. He measured 10 grams of KClO3 and dissolved it in a calorimeter containing 250 g of water. The temperature drop was 3.5 K. What is qH2O? WHat is qrxn? WhaT IS THE change of dissolution of reaction? is the reaction exothermic or endothermic?At 25°C and 1 atm, 0.500 mol/kg solution of NaCl is water has VNaCI = 18.63 cm³/mol and VH2O = 18.062 cm³/mol. Find the volume of a solution prepared by dissolving 0.5000 mol of NaCl in 1000.0 g of water.Chemistry 5 grams of cereal was crushed and dissolved in 25 mL water and placed in a dialysis bag. This sample was dialysed over a week in a beaker which contains 500 mL of water. 5 mL of the dialysate (an aliquote from the 500 mL) was separated to determine the glucose concentration. The glucose concentration was determined as 5 mM. Calculate the amount (% w/w) of glucose present in the cereal. Hint: The molecular weight of glucose is 180.



