(AsH,) in acidic aqueous solution. Be sure to add physical Write a balanced half-reaction for the reduction of aqueous arsenic acid (H,AsO,) to gaseous arsine state symbols where appropriate. Don't Know Submit 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Cente Lenovo
(AsH,) in acidic aqueous solution. Be sure to add physical Write a balanced half-reaction for the reduction of aqueous arsenic acid (H,AsO,) to gaseous arsine state symbols where appropriate. Don't Know Submit 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use Privacy Cente Lenovo
Chapter22: Bulk Electrolysis: Electrogravimetry And Coulometry
Section: Chapter Questions
Problem 22.30QAP
Related questions
Question
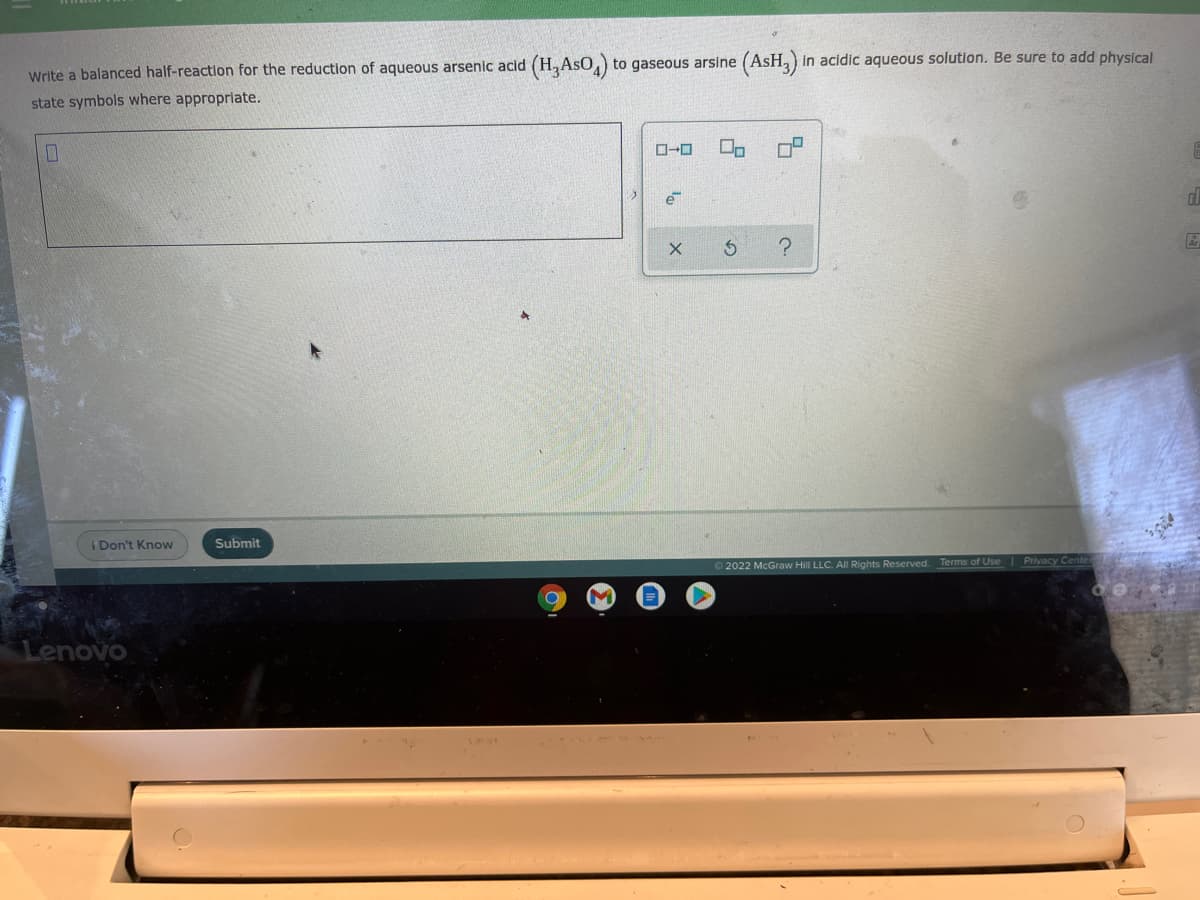
Transcribed Image Text:to gaseous arsine (AsH,) in acidic aqueous solution. Be sure to add physical
Write a balanced half-reaction for the reduction of aqueous arsenic acid (H, Aso,)
state symbols where appropriate.
i Don't Know
Submit
Terms of Use
Privacy Cente
0 2022 McGraw Hill LLC. All Rights Reserved.
Lenovo
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,