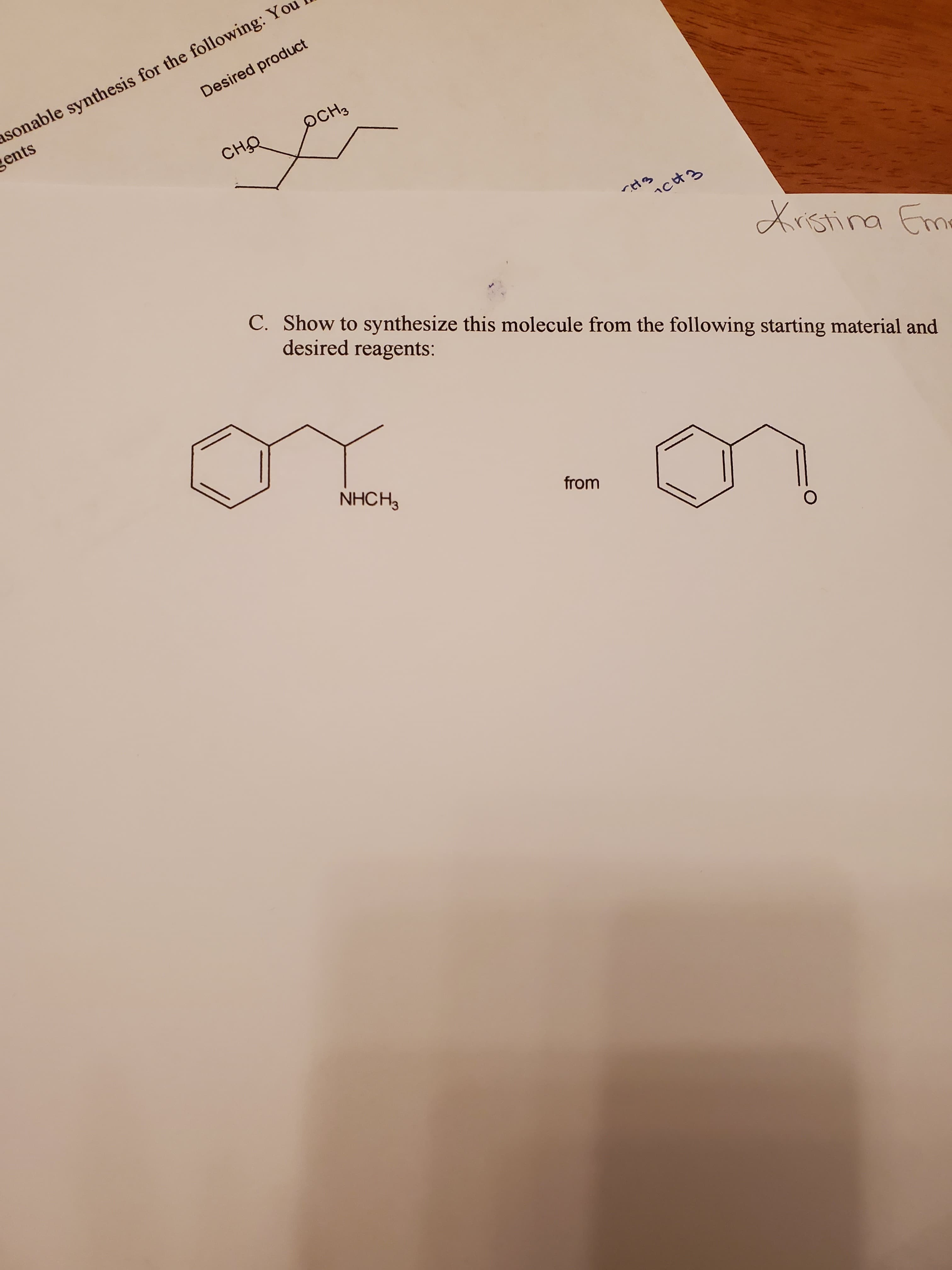
asonable synthesis for the following: Yo gents Desired product оснз сно icH3 Xrstina Eme C. Show to synthesize this molecule from the following starting material and desired reagents: or NHCH3 from
Q: MeO. ZON A
A: Given reaction is electro cyclisation reaction.
Q: The most common method for the synthesis of unsymmetrical ethers is the Williamson synthesis, a…
A: Williamson's synthesis is a method for synthesis of formation of ether by SN2 reaction of alkyl…
Q: For the following reaction provide the expected major organic product(s). Include all stereoisomers…
A: When an unsymmetrical reagent is added to an unsymmetrical alkene then the negative part of the…
Q: Explain the steps involve in the below organic reaction in detail a. ONe HN
A: A zwitterion or dipolar ion is a molecule which contains an equal amount of positive and negative…
Q: Reaction of with CH3CH₂COOH forms CH3CH2CH2OH OLIAIH4 HCI KMnO4 O CH3NH2 NaBH4 as a product.
A: -> Reactant is CH3CH2COOH . -> Product is CH3CH2CH2OH .
Q: The most common method for the synthesis of unsymmetrical ethers is the Williamson synthesis, a…
A: Williamson synthesis is used to prepare ether using alcohol and alkyl halide. First, alcohol is…
Q: Show how you would synthesize the following compounds, storting with benzene or Or toluene ond any…
A: Welcome to bartleby !
Q: HCI ÇI
A:
Q: Please write the corresponding name only of the products (A, B, C, D and E) Compound Ais an…
A: The structure of 3,4-dimethyl-3-hexanol is as follows:
Q: H3CSH 2 singlets of integration =3 2 overlapping triplets of integration =2 TPPM -5 -6 15 14 13 12…
A: The α,β-unsaturated ketones and aldehyde undergo two types of nucleophilic addition. The one is…
Q: How to retrosynthesize this compound. Give steps in the foward direction also, with reagents. NH
A:
Q: Synthesize the following yntheses storling from the istorting compounds. using necessory organic od…
A:
Q: identity the reagents necessary to achienve the trandormation below. You will need to une at ntane…
A:
Q: Which of the following is/are the expected product/s of the reaction of 2-pentanone (CH3COCH2CH2CH3)…
A: In a haloform reaction, there must be CH3CO- group.
Q: Explain the steps involve in the below organic reaction in detail ONa H3N
A:
Q: GivE REASONABLE SiNTHE SES OF THE INDICATED PeoouCTS FROM THE GivEn STARTING MATERIALS, Snow…
A: The above formation can be done with aldol condensation reaction followed by the Catalytic reduction…
Q: mechanism for the of hydrolysis (1-bromo of Propose a reaction methylpropyl)cyclohexane. Including…
A: Stereochemistry is branch of chemistry in which we deal with arrangement of atoms in three…
Q: (b) Provide the retrosynthesis along with forward reaction for the the following compounds as…
A: Retrosynthesis is the imaginary breaking of bonds of the molecule in such away that the molecule is…
Q: 2, Solvolysis is referring to substitution reactions Whee The solvent 's study in res kigaked the…
A: The path followed during the solvolysis of compound 1 and compound 2 is needed to be discussed along…
Q: Use the EAA or DEM synthesis and show how you would prepare each of
A:
Q: (c) During Corey's synthesis of allomone he utilised the ketone shown below. Outline the…
A: The α,β-unsaturated carbonyl compound is identified by the presence of the double bond in the…
Q: Synthesize the following compounds with any starting materials you'd like (within reason!)!
A: We have to synthesize of 1-phenyl-1-butyne from suitable reactants and reagents.
Q: Provide He Skelera) Structucs of He tuo reactants necessary te preducts under to produce each of He…
A: Given reaction is example of Diels-Alder reaction. We can find the reactants of this reaction by…
Q: Oraw the necessary base and electrophile to prepare the following alkenes as maj USION
A: Alkene are unsaturated hydrocarbon having carbon-carbon double bond. They have general formula…
Q: H2O, H+
A: Since you have posted a multiple question so i have first one for you. If you need any specific…
Q: CH, CH, 1. BH, THE 2. NaOH, H0, H,O, он H,C CH2 Alkenes can be hydrated via the addition of borane…
A: It follows the hydroboation- oxidation reaction it follows the two step hydration reaction that…
Q: the 1,3 - Cyc lo pentadiene, using finorgaric compound , încluding 2) Based, on or ganic chernicols.…
A:
Q: The following presented reaction scheme lead to the synthesis of which major product from-benzene,…
A:
Q: Outline how one might achie ve the foltowing transE Showing reagents Synthetic scheme. - show…
A:
Q: Provide only one plausible retro synthetic scheme for the molecule below. In addition, provide a…
A:
Q: Taking into account anti periplanar geometry, predict the major E2product formed from attached…
A: E2 elimination reaction takes place when the leaving group is anti-periplanar to the acidic proton.…
Q: How many elimination product(s) is /are possible in the reaction of compound…
A: Here, we have to find the number of elimination products that are possible in the reaction of…
Q: natwe of the Substit uen ts (orthe / pau us meta). Note yon should have one synthe tic Talke…
A: The products given are,
Q: Synthesis: Synthesize the following compounds using the starting material shown a reagents you may…
A: The given reaction is the conversion of the alkene to syn- diol. 1 methylcyclohexene is converted to…
Q: Reaction of with CH3CH₂COOH forms CH3CH2CH2OH as a product. O NaBH4 O HCI O LiAlH4 O CH3NH2 O KMnO4
A: Given, Reaction of ...............with CH3CH2COOH forms CH3CH2CH2OH as a product. Fill the blank ?…
Q: Which reagent(s) sequence(s) is/are optimum for preparing butanoic acid from 1-propanol? i. K2Cr2O7…
A:
Q: Synthesize Novocaine from benzene and any other reagents of four carbons or fewer
A: INTRODUCTION: The structure of Novocaine is :
Q: How can you synthesize the attached compound from starting materialscontaining no more than four…
A: The attached compound from starting materials containing no more than four carbons has to be…
Q: The hydroboration–oxidation of internal alkynes produces ketones.(a) When hydroboration–oxidation is…
A: Hydroboration-Oxidation reaction:- This reaction happens in unsaturated bonds like alkene and…
Q: (B1) The following Compound can undergo Signifiant Kefo-enol + possesses enhan ced enot form.…
A:
Q: Taking into account anti periplanar geometry, predict the major E2product formed from attached…
A: The compounds in which the leaving group and the adjacent hydrogen atom are anti-periplanar to each…
Q: Provide the major organic product of the following reaction. NBS, Heat ROOR
A: NBS is the reagent used for providing bromine in low concentration. It is used for allylic…
Q: the major organic product of the following reaction. Be sure to indicate proper -mistry and…
A: here we are required to find the product of the reaction
Q: De most common method for the synthesis of unsymmetrical ethers is the Williamson synthesis, a…
A: The complete synthesis of 2-propoxypropane from propene is show below, The acid catalyzed hydration…
Q: (b) Provide forward reaction) compounds provided : (i) (ii) NC suitable retrosynthesis for the CN as…
A:
Q: when tne fonow soluolysis in nmethano Fue prdus are obtamedincludina' sten eolsomers oniytwo ae…
A: In this mechanism bromine ion is good leaving group and methoxy ion is the reagent. first…
Q: + COOH HOOD. A
A: Here we have to write product formed , it's stereochemistry and it's name in the following Diel's…
Q: Which of the following bicyclic ring systems can be prepared by an intermolecular Robinson…
A: The bicyclic ring systems which can be prepared by an intermolecular Robinson annulation has to be…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- predict the major products of the following reactions. İf it possible, write all steroisomersSynthesis: draw/provide the likely organic products generated. With attention to regiospecificity and stereochemistry indicating ALL likely products( major,minor and trace)OChem help... Complete the following reaction sequence: Indicate regiochemical/stereochemical details as relevant. Provide the stepwise mechanism for the following... (see image)
- Provide the products, intermediates, and/or reagents forthe following reactions:Ochem.... Complete the following reaction sequence: indicate regiochemical/stereochemical details as relevant. Also, provide the structures for the major organic products formed in each step... (see attached image)give reactions and intima mechanism of following reactions-
- provide the major profucts ( wrote "no reaxtion "if you think so) for the following reactions with correct stereochemistry. Please explain step by step with explanationSuggest a detailed mechanism for the reaction below. Represent the product in the most stable conformation and also its stereochemistry.Give the MAJOR PRODUCT/S of the following reactions. Include stereochemistry asappropriate.
- please provide the missing organic materials starting materials, reagents/reactions, conditions or organic products in the reaction below. kindly provide and show ther eaction mechanism as wellOne possible way of determining the identity of an alkene, is to let itundergo an oxidative cleavage reaction in the presence of hot basicpotassium permanganate. You are given two containers said to containdifferent alkenes. Container A is marked as cis / trans‐2‐butene andcontainer B as 2‐methyl‐1‐butene. Explain by referring to the formation ofproducts, how you would verify the identity of the alkenes.Give the major product of the follow epoxidations. Give the structure of all reagants abbreviated.

