Q: 1) Draw the structure of the alkene product for the E2 elimination reaction shown below. 1)a 2)b…
A: In E2 elimination the groups must be anti to leave. The base will attack hydrogen and the bond will…
Q: A reaction proceeds with a 13:3 ratio of enantiomers. Convert this ratio to the percent enantiomeric…
A: % enantiomeric excess is given by the difference of percent major enantiomer and percent minor…
Q: Draw the curved arrow(s) to depict the formation of the keto form of an enolate ion via a strong…
A: The hydrogen carbonyl (adjacent to C=O bond) are highly acidic and due to which it is readily lost…
Q: 25) The intermediate carbocation in SN1 has tetrahedral geometry A) True B) False
A:
Q: How many substitution product/s is/are formed when metabromo anisole is treated with ammonia? A.…
A: when metabromo anisole is treated with ammonia reaction proceed through benzyne mechanism.
Q: Draw the structure of the product of the reaction between the compound shown below and H,S04. CH3…
A: Here, the given compound is reacting with H2SO4 , we have to find out the product formed in this…
Q: Select the starting material for the transformation below. MCPBA CH3 CH3 CH2 C Oc
A: Given,
Q: 3.) Consider the hydrogenation reaction below, which replaces all r bonds with carbon-to-hydrogen o…
A: The given reaction is shown below: For C=10, the number of hydrogen in saturated molecule is…
Q: For the given ee values, calculate the percentage of each enantiomer present.a. 90% eeb. 99% eec.…
A: Constitutional isomers can be defined as the molecules which have the same molecular formula and…
Q: Draw the organic product(s) of the following reaction. NaOC2H5 / C2H5OH CH;CH2-CEC-H You do not have…
A: The reaction is as follows:
Q: 6. Draw all of the stereoisomers that you would form in the reaction of CHCI3 and KOTBU given the…
A:
Q: Br b. H3C the squiggly line means both E and Z alkenes form. You may draw either in your mechanism…
A:
Q: 5. Consider the following reaction: H-CI (ii) (i) intermediate product (a) Use curved arrows to…
A:
Q: 1. Write the correck IUPAC names For Hhe Folloui ng organic compounds: Br CH3 H3C H. Assign E en or…
A:
Q: CH3 MCPBA H3C COH, H* Ph. CH3
A:
Q: Draw a structural formula for the product of the reaction shown. OCH3 • Use the wedge/hash bond…
A: A reaction between diene and dienophile, whose product is to be determined.
Q: Question 8 Rank the following carbocations from most stable to least stable: II II O III >I> II OI>…
A: Given that, Three carbocations have been given,we have to find out order of stability?
Q: 1,2 addition of Br2 to an alkene results in a _______________. A. alcohol B. vicinyl dihalide C.…
A: 1,2 addition of Br2 to an alkene results in a ?
Q: 9. Cubane and its derivatives have been considered for use as explosives and as materials for energy…
A: To calculate the strain energy of cubane and describe the structural feature leading to this much…
Q: Rank the following groups in order of decreasing priority. −NH2, −CH2NH2, −CH3, −CH2NHCH3
A: Priority is given to elements on the basis of atomic numbers. If the atomic number is more, its…
Q: 4. Br 5.
A: Give the IUPAC name of the following structures--
Q: 5. (a) Answer the following questions based on the reaction scheme below. Jawab soalan berikut…
A:
Q: 5. Consider the following reaction: H-I (ii) (i) intermediate product (a) Use curved arrows to…
A: We have to solve the reaction and thwn answer the questions. Let us do this step by step.
Q: Ignoring stereochemistry, what is the major organic product of the reaction of 2,4-dimethylpentane,…
A: The substitution of one of the hydrogen of the alkane group with a halogen atom is called…
Q: Part 5a. Rotamers. Answers the following questions about the C-C bond indicated below 3-methylhexane…
A: Here we have to determine the energy cost for a CH3/CH2CH3 gauche interaction of 3- methylhexane…
Q: b) Use curved arrows to explain the formation of the product. Comment on whether the reaction will…
A: Given:
Q: Certain carbon–carbon double bonds can also be stereogenic centers. explain how this can occur, and…
A: The three additional stereogenic centers in discodermolide has to be located
Q: 1. Write the correct IUPAC names for the following organic compounds: a) Br CH3 H3C (Assign E or Z…
A: The IUPAC name of the given compounds are as follows:
Q: 7. Which of the following is more stable? 8. Name the compound according to IUPAC nomenclature. (Use…
A: When the high priority bulky groups are on the opposite side of the double bond, the alkene is…
Q: Ignoring stereochemistry, what is the major organic product of the reaction of 2,4-dimethylpentane,…
A: Chlorination reaction are proceeds via radical mechanism, in presence of light.
Q: 10. Draw a tetrahedral representation of (R)-pentan-2-ol in the box below.
A: A tetrahedral molecule has a bond angle of 109.5°. Configuration R will be assigned to the molecule…
Q: d) Draw the structures of A and B in the following reactions. N-CH3 i) CH3I (Excess) i) CH31…
A: Given that : We have to draw the structure of compound A and compound B in the following reaction :
Q: (1)Draw a structural formula for the more stable carbocation intermediate formed in the reaction…
A:
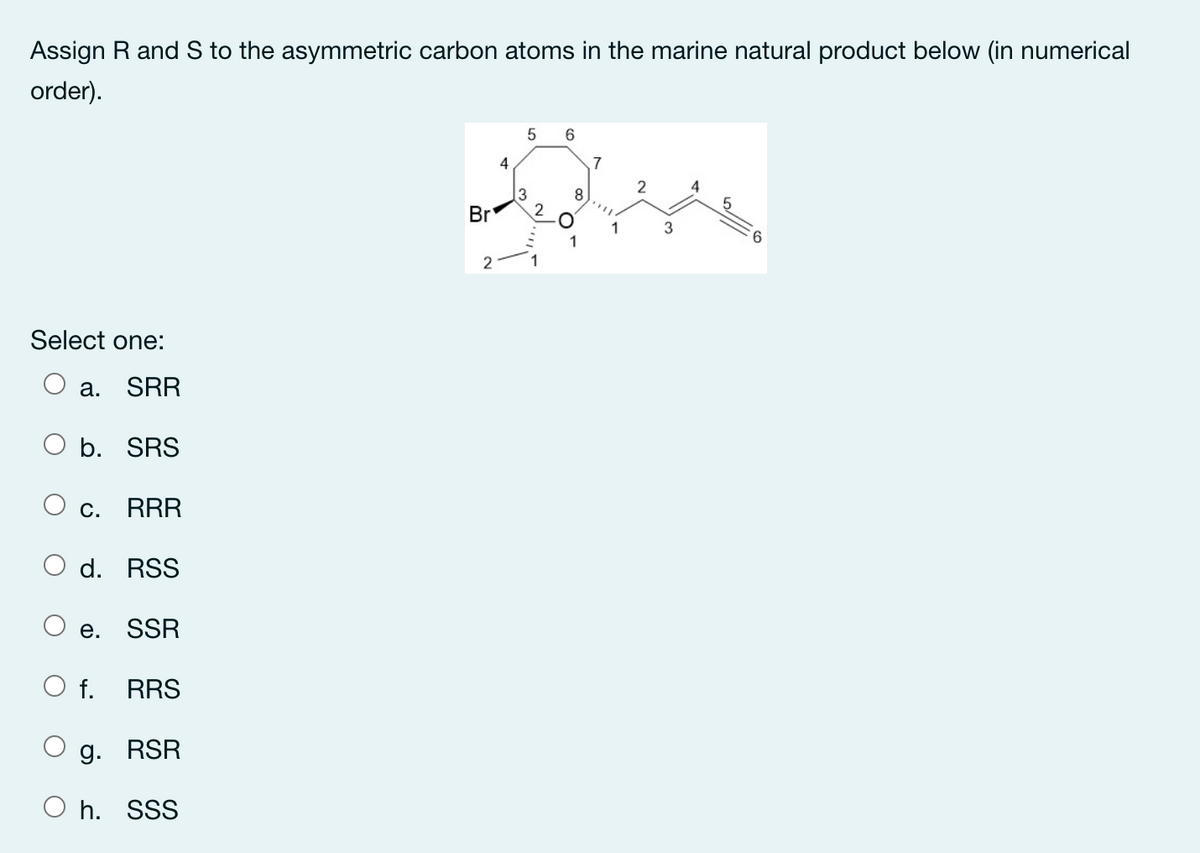
Q: Assign R and S to the asymmetric carbon product below (in numerical order). 5 6 3 Br 2.
A: According to the question, we need to determine the configuration of symmetric carbon atoms in the…
Q: Include structure(s) for the product(s) in the following reaction and upload a jpg image of your…
A:
Q: 7. Draw the structure of these molecules clearly depicting R/S configuration (wedge/dash notation).…
A: If the three groups attached to the chiral center are ordered clockwise from highest to lowest…
Q: Draw the structural formula of the product of the reaction shown below. Na OCH₂CH3 + CH3CH₂OH • You…
A: Three membered rings formed by two carbon and one oxygen atom is known as epoxide ring. The…
Q: NBS, (light) hv, CH2CI2 NO 2 CH3CI, AlCl3 1. CH3CH2CI, AlCl3 2. Br2, FeBr3
A:
Q: 4.28 Name each alkene and specify its configuration using the E,Z system. (See Examples 4.3, 4.4) Br…
A:
Q: 23) In the addition reaction pictured below, on what carbon would you expect the -SOs to be added?…
A: Benzene is electron rich species and it show easily electrophilic aromatic substitution reaction. In…
Q: 2) How many nodes, other than the node coincident with the molecular plane, are present in the 3 of…
A: Annulene: Annulenes are monocyclic hydrocarbons that contain the maximum number conjugated double…
Q: 4. Write the correct IUPAC names for the following orga compounds: b) Br CH3 H3C (Assign E or Z…
A: The systematic naming of organic compound is given by IUPAC. The naming of organic compound is done…
Q: Ignoring stereochemistry, what is the major organic product of the reaction of 2,4- dimethylpentane,…
A: This is photolytic chlorination reaction of alkane. This reaction follows radical mechanism and…
Q: 5. Rank the following compounds by their reactivity with CI- (1 = least reactive, 3 = most reactive)
A: Rank Reactivity against Cl-
Q: predict the major product of the reaction sequence below
A:
Q: Q2 Complete and balance the following reactions: a. CH3- CH=CH-CH3 + Br2 pg ofo courbonige b. + Cl2
A:
Q: b. Ciclohexanona + NH3 H2, Ni ciclohexilamina
A: b. Carbonyl compounds on reaction with ammonia form imines. Imines on reduction H2 /Ni form…
Q: Draw the product of the following compounds with the given image. 1. A. BH3 B. H2O2, OH- 2. A. O3 B.…
A:
Q: 1. CH3CH2CI, AlCl3 2. Br2, FeBr3 Br2, H20 H*, H20
A:
Q: Order each bond from having the strongest IMF (on top) to having the weakest IMF (on bottom)_ t 1.…
A: C-F molecule is polar in nature having a considerable difference in the electronegativity of C and F…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- up an example (not appearing in this ChemActivity) of a pair of molecules that are a)constitutional isomers, b) conformers. c) configurational stereoisomers.What is the value in kJ/mol, of the lowest energy trnasition above? a.) 545 b.) 444 c.) 320 d.) 266Anti-Markovnokov’s rule is followed in the presence of? a. Ni b. CCl4 c. H2O2
- Consider the following aromatic compounds a para-substituted compound?, the most reactive towards EAS? an ortho-substituted compound? does not react with RX, FeX3?Which group in the following pair is assigned the higher priority in R,Snomenclature −CH2NH2, −NHCH3Use the Hammond postulate to explain why (CH3)2C = CH2 reacts faster than CH3CH = CH2 in electrophilic addition of HX.
- Which of the following is most soluble in basic medium? a. cyclopropane b. 1,3-cyclobutadiene c. 1,3-cyclopentadiene d. benzeneCompound X is insoluble in cold KMnO4, Br2 in CCl4, and conc. H2SO4. Compound X is most likely: a. an alkane b. none of these c. an alkene d. an alcohol e. an alkyl halide Indicate which of the ff. statements regarding nucleophilicity is incorrect. F- is more nucleophilic, hence, more reactive towards methyl iodide than Cl-. Second row elements are more nucleophilic than first row elements of comparable basicity. The rate of SN2 reaction may be markedly affected by the nucleophilicity of the attacking atom. Nucleophilicity is the affinity of a nucleophile to an electrophilic carbon Which of the following alkynes can be deprotonated by NaNH2 in liquid NH3? 3-Methylhex-2-yne Pent-2-yne 3-Methylbutyne none of these Hex-3-yneWhat is the structure based on the HNMR?
- Explain briefly and clearly the following concepts, taking as reference the molecule of n-butane and the corresponding drawings or illustrations. See pages 149-152 of the book Organic Chemistry, sixth edition (J. G. Smith). 4. What is steric hindrance in a conformation? Then draw a picture to illustrate the concept? 5. What is the torsional stress of a conformation? Then draw a picture to illustrate the concept? 6. Describe 1,3-diaxial interaction and illustrate with a specific example.please provide the machanisms of 1a, 1e, 1fIn 1935, J. Bredt, a German chemist, proposed that a bicycloalkene could not have a double bond at a bridgehead carbon unless one of the rings contains at least eight carbons. This is known as Bredt’s rule. Explain why there cannot be a double bond at this position.



