Q: Which parts of an element do not vary in number?a. electronsb. neutronsc. protonsd. All of these…
A: Electrons are negatively charged. Protons are positively charged. Neutrons are neutral.
Q: Generally YAC contains how many elements?a) 1b) 2c) 3d) 4
A: YAC stands for yeast artificial chromosomes and is widely used in the field of genetic engineering.…
Q: The mass number for an isotope is 14. This isotope could be: a carbon atom with 8 neutrons an oxygen…
A: Mass number of an atom is the sum of the number of protons and neutrons in the nucleus of the atom.…
Q: The atomic number of sulfur is 16, Sulfur combines with hydrogen by covalent bonding to form a…
A: Every element has smallest unit of matter called atom that retain the properties of an element.…
Q: Rank the bonds in order of increasing strength - Van Der Waals, Ionic , H-bonds, Ion-dipole, London…
A: Melting and boiling points are measures of strength of the attractive forces between individual…
Q: 1. SEP Compare Data Of the metals shown in the figure, which has the highest melting point? Which…
A: Of the metals given in the figure, molybdenum (Mo) has the highest melting temperature i.e. 2617°C
Q: Which element is the world's lightest metal and increasingly a vital component of batteries used in…
A: Any metals heaviness and lightness depends upon the density of that metal. Density is defined as…
Q: Define the Primary Bonding in materials with examples and drawings.
A: Primary bonding is generally defined as a donation and also defined as mutual sharing of valence…
Q: What is the basic units of matter and the defining structure of elements? a. Cell b. Atom c.…
A: Matter- It is any substance that takes up space, has volume and has mass. It exists in various…
Q: _____________contribute hydrogen ions (H+) to a solution.a. Basesb. Isotopesc. Acidsd. Compounds
A: Answer: Introduction: According to the Lowry-Bronsted definition, an acid is a substance which is a…
Q: 1.Match the following terms with their correct definitions: matter, atom, molecule, element, and…
A: Elements are the simplest complete chemical substances. A compound consists of two or more types of…
Q: The percentages of naturally occurring elements making up the human body (see Table 2.1) are similar…
A: Introduction: The requirement for trace elements might actually be viewed as a synapomorphy.
Q: Which element is the world's lightest metal and increasingly a vital component of batteries used in…
A: Metals are solid opaque elements that has good conductivity of heat and electricity. Lightness of…
Q: Determine the mass in grams of 6.42 × 10²¹ atoms of carbon. (The mass of one mole of carbon is 12.01…
A: Mole (mol): the amount of material having 6.02214 × 1023 particles. This number is known as the…
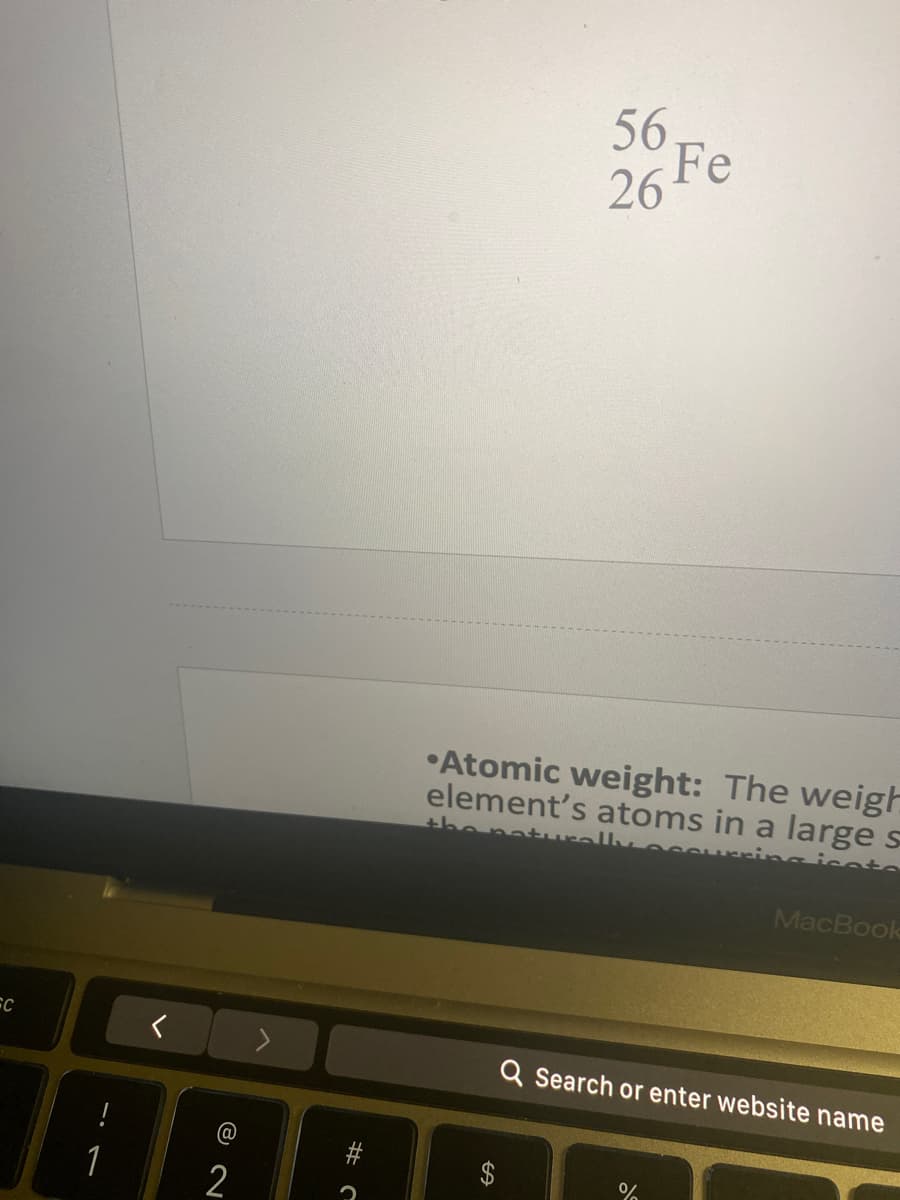
Q: 7. Give the symbol of and the number of protons in one atom of: Lithium Iron Охygen Krypton Bromine…
A: Symbols and number of protons in one atom are as below : Lithium - Li - 3 Iron - Fe - 26 Oxygen - O…
Q: State whether the bonding in each compound is likely to be covalent and short reason to prove it i)…
A: An atom is the basic and fundamental unit of matter. An atom consists of electron and the nucleus in…
Q: Relate atomic structure to the periodic table of the elements
A: Atom is smallest unit of matter which forms element Atomic structure can be defined as structure of…
Q: Chemical Formula Elements and Number of Atoms Model 1. H, 2. 0, CH, 5. NaOH 3. 4.
A: The Atom is the smallest unit of a matter that form an element. The atom contains electrons and…
Q: Describe the characteristics of transition metals.
A: Elements that have d orbit partially filled are known as Transition elements. These metals come…
Q: Please explain compounds and chemical bonds, ionic, covalent, and hydrogen bonds
A: The questions ask about the compounds, chemical bonds, ionic bonds, covalent bonds, hydrogen bonds.
Q: Explain 3 properties of water?
A: Water is a universal solvent
Q: Describe the electrical charge, mass, and location of the threemajor subatomic particles in an atom?
A: The Atom had been believed to be the smallest undivided particle possible in any element. Later on…
Q: 16) An ion with six protons, seven neutrons, and a charge of 2+ has an atomic number of A) four B)…
A: Physical as well as chemical properties of an element depend upon its atomic structure, the number…
Q: The name given to the atomic particle found outside the nucleus of an atom isa. proton.b. neutron.c.…
A: The smallest particle of matter is called an atom. It is the very basic structural unit of any…
Q: Can you please answer all of the sub questions please I really appreciate it
A: Atom is the smallest unit of matter composed of protons, electrons, and neutrons. It has two…
Q: Do a web search to identify each of the following elements>isotopes and indicate the number of…
A: Atoms are the small particles that contain three main types of subatomic particles.
Q: 10. Identify the following as either chemical change (C) or physical change (P) baking a pie melting…
A: A chemical change is a process during which one or more ingredient (reactant) chemically reacts to…
Q: Atomic number equals the number of 8 or Охудen Atomic mass equals the number of 15.999 8. 30 3 Li…
A: An atom refers to the smallest unit of matter. It comprises protons, neutrons, and electrons.…
Q: 9. Draw the Electron Structure for Rb & O 10. Draw the Electron Structure for Sr & F Formula Unit:…
A:
Q: Predict polarity of molecules using electronegativity and molecular geometry (VsePr).
A: VSEPR theory is valence shell electron pair repulsion theory. It is mainly used to predict the…
Q: In the periodic table, the elements are arrangedaccording to ________ .a. size c. mass numberb.…
A: A periodic table is a tabular table. It displays all the chemical elements in this table. Over time…
Q: Complete the following table Symbol # protons # electrons # neutrons Atomic Mass Number Number 6°CO…
A: The atomic number is considered as the number of protons exhibit in the nucleus. These protons…
Q: Look up the valence electron configuration, covalent atomic radius, effective nuclear charge, first…
A: The valence shell electronic configuration of the p-block elements is ns2np1–6, and they are found…
Q: Sort the subatomic particles according to their masses. Drag each subatomic particle into the…
A: Neutron is a subatomic particle that has no charge and has a mass slightly greater than the proton.…
Q: Q1// Draw the position isomer for the chemical formula ? A. C,H,Br B. CHu
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: A solution of calcium phosphate in water contain 235 gms of CP per L at 30 deg c. The density of the…
A: Percentage composition is calculated as the percentage by mass of individual elements contributed to…
Q: ELEMENTS MACROMOLECULE FUNCTION MONOMER POLYMER EXAMPLE(S) CONTAINED CARBOHYDRATES LIPIDS
A: Carbohydrates supply energy to the body, which is one of their key purposes. Before reaching the…
Q: What is formed when an atom loses or gains an electron? O a. ion O b. bond O C. molecule O d. a new…
A: Molecules of every substances are made up of small particles. These are called atoms. Bohr's model…
Q: 5. (a) How many covalent bonds does each of the following type of atom typically form? (i) H (iii) C…
A: INTRODUCTION Covalent bond Whenever there is an equal share of electrons between the atoms, covalent…
Q: Element Characteristics Easy to bend and shape • Good conductor of heat • Good conductor of…
A: The metals are substances that has high conductivity and high electricity.
Q: Calculate the plane density of (110) plane at BCC sbructure Crystal : 2
A: For this (110) plane there is one atom at each of the four cube corners through which it passes,…
Q: Choose the correct answer 1-An important result of Rutherford’s work was to establish that... *…
A: The gold-foil experiment revealed that the atom is made up of a thin, large, positively charged…
Q: I. Research for structure (submit on short bond paper, handwritten only)
A: 1. Phenolphthalein, (C20H14O4), organic compound of the phthalein family commonly used as an…
Q: Diagram and describe the atomic structure of biologically important elements.
A: Most of the elements in living matter have relatively low atomic numbers. The four most abundant…
Q: A nitrogen atom has 7 protons, and the most commonisotope of nitrogen has 7 neutrons. A radioactive…
A: Isotopes are the atoms of same element having same atomic number but different mass numbers. For…
Q: OCCURS IN WATER WITI 1. cobalt 2. strontium 3. selenium 4. vanadium
A: Osteoarthritis, also known as degenerative joint disease (DJD), is one of the most common type of…
Q: The smallest unit of an element that still retains thedistinctive behavior of that element is an…
A: Given: Choose the correct answer: The smallest unit of an element that still retains the distinctive…
Step by step
Solved in 2 steps

- 0.47 absorbance/ 4713.6 mol/L =In scientific notation please & 0.57 abosorbance/4713.6 mol/L=What do you call to the unit amount of a substance used for measuring large quantities of very small entities such as atoms, molecules or other specified particle like counting molecules of medicine in 0.20g confusion?For Be-10, find the: a.) mase defect (in grams) b.) binding energy in kilojoules per mole. mass proton= 1.00728 amu; mass neutron= 1.00867 amu; mass Be-10 = 10.013534679 amu
- Q.1. A solution of calcium phosphate in water contain 235 gms of CP per L at 30 deg c. The density of the substance at this temperature is 1.143 gm/cc. Calculate , composition in wt%, mole%,atomic % and volume%of water. Density of water 0.999 gm/cc.Chlorine is an element with the symbol Cl. Draw and label atomic structure of chlorine indicating the number and locations of protons, neutrons and electrons in one atom of chlorine.Determine the mass in grams of 2.10 × 10²¹ atoms of copper. (The mass of one mole of copper is 63.55 g.)
- Define the mole, and calculate the molar mass of a compound from the molecular formula.When 0.5 mol of a certain ionic substance is dissolved in 1.0 kg of water, the freezing point of the resulting solution is -2.8 °C (270.35 K), How many ions does the substance give when it dissolves?The atomic number on an element tells us the number of
- An ion has 15 protons, 16 neutrons, and 18 electrons. The symbol for the ion is 31P3+. 31P3-. 31Ar3+. 34S+. 34S-.The smallest unit of an element that still retains thedistinctive behavior of that element is an ________.a. electronb. atomc. elemental particled. isotopeElemental analysis of a compound with molar mass 342.3 g/mol gives the following mass percent composition: C 42.11%, H 6.48%, O 51.41%. Find the molecular formula of the compound. Enter your answer in the space below using the following format: if the molecular formula of a compound containing elements X, Y, and Z is X2YZ3 enter your answer as X2YZ3.

