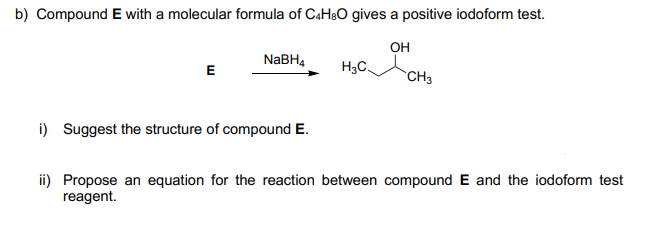
b) Compound E with a molecular formula of CAH&O gives a positive iodoform test. он H3C. NABH4 E CH3 i) Suggest the structure of compound E. ii) Propose an equation for the reaction between compound E and the iodoform test reagent.
Q: 6.)Calculate the pH of a 0.400M solution of phosphoric acid, H3PO4.
A:
Q: The solid rocket propellant for the Space Shuttle (and Tomahawk Missiles) contain ammonium perchlora...
A:
Q: Consider the following data on some weak acids and weak bases: acid base K. name formula name formul...
A:
Q: A mixture of 0.02993 mol of O, 0.02165 mal of NOCI, 0.02691 mol of NO, and 0.01037 mol of C is place...
A: Answer: First of all, with the help of values of initial moles of gases and equilibrium moles of chl...
Q: 2 reactions involved in the complete oxidation of cobaltous ion *
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: Determine if the statements are true. The larger the surface area, the higher the dissolution. ...
A: True or false: As per our guideline we have to answer first three questions only:
Q: Problem 9 1-Propose a reasonable biosynthesis of Bisabolyl cation from Farnesylpyrophosphate
A: According to Q&A guidelines of Bartleby, we are supposed to answer only the first question out o...
Q: 2. What is the volume of 0.35 mol of oxygen gas at STP?
A:
Q: Give biosynthesis of thujone from gerany|pyrophosphate
A: Thujone is a monoterpene with a ketone group and it is found in several plants including Salvia offi...
Q: Indicate what specific electron will have the following quantum number indicated below: 8. 4, 3, +1,...
A:
Q: 1. An organic compound was found to contain 54.5% C, 9.2% H, and 36.3% O. a. What is the empirical f...
A: Solution: From the elemental composition of any compound we can calculate, the empirical formula. W...
Q: Indicate whether the salts below would form solutions that are acidic, basic, or neutral in pH. KCIO...
A:
Q: In the mid-1930s a substance was isolated from a fungus that is a parasite of ryes and other grasses...
A: Given that, lysergic acid diethylamide (LCD, C20H25N3O). It is a weak base, having Kb = 7.6×10-7. Th...
Q: At 600°C, carbon monoxide gas and hydrogen gas reacts with each other to form methane gas and water ...
A: Given T = 600
Q: a. Br CH,ONa Br2 CH,CO,H Light NaOH/H20 0H HO.
A: The correct answer is given below
Q: Mass of NaOCl in commercial bleach titrated. ?
A: Moles help to calculate mass
Q: For a 0.0491 M aqueous solution of cyanic acid, HCNO (K, = 3.5*104), determine the following. What a...
A: Weak acids are the weak electrolytes that dissociate in aqueous medium to form hydronium ions. The d...
Q: A solution is prepared by dissolving 27.03 grams of FeCl,* 6H;O, and diluting to give a final volume...
A: The molarity (M) of a solution is the ratio between the number of moles (n) of solute to the solutio...
Q: 2. A compound contains 43.38 percent Na, 11.33 percent C, and 45.29 percent oxygen. What is the empi...
A: Here, I am providing you the solution of only question 2 as you have asked for it. Given, Mass % of ...
Q: 3) Design a reasonable synthesis for the following transformation. = trort + B.
A: Here we have synthesize the given polymer which is a polyester obtained from starting material 1,4-d...
Q: a) What is the advantage of very small particle sizes in LSC columns? (b) With reference to the Va...
A: a) Small particle sizes are used in the LSC columns.
Q: Calculate the pI value of GE- dipeptide The pKa values of the terminal amino and carboxyl groups...
A: Calculate the pI value of GE- dipeptide ?
Q: (a) What are colloids? How are they classified? (b) Discuss the origin of charge on the colloidal pa...
A: (a) Answer - Colloids - A colloid is a mixture in which one substance consisting of microscopically ...
Q: 70 65 Vapor 60 55 50 2.4 45 40 Liquid n diagram hexane 1.0 atm 35.1 30 0.0 0.2 0.4 1.0 0.6 Mole frac...
A: Soln
Q: e K,
A:
Q: mass
A:
Q: Solid carbon dioxide is also called dry ice. Dry ice is placed in a sealed container. Some CO2 subli...
A: The equilibrium reaction taking place is given as, => CO2 (s) ⇔ CO2 (g) Given: Volume of the cont...
Q: Give biosynthesis of Carvone from gerany|pyrophosphate in clear handwritten!......
A: Carvone is a naturally occurring ketone found in the essential oils of caraway, dill, and spearmint ...
Q: You have a solution which is 45.6% by mass isopropyl alcohol (MM = 60.11 g/mol) in water. The densit...
A:
Q: 2/Identify each substance as Acid base Conjugate acid Conjugate base
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a subst...
Q: It is desired to make a 0.1M solution by adding 500grams of copper sulfate pentahydrate crystals. Wh...
A: Molarity is a concentration term for a solution. The molarity of a given solution is defined as the ...
Q: Which graph corresponds to this reaction? NH3(g) + HCI(g) 5 NH,CI(s) D AG AG AG AG +
A: In this question we are given a chemical reaction and we have to choose the correct graph between Gi...
Q: what is the expected mass of the products (5-amino-2-chlorophenol and 4-chloro-3-hydrixyacetanilide)...
A: Amount of 2-chloro-5-nitrophenol added = 2.75 g Molar mass of 2-chloro-5-nitrophenol ...
Q: Directions: Give the charge of the atoms. 1. 2. + + ++ ++ 3. + + + + ++
A: The number of protons and electrons in ions are given as,
Q: Help me find the % deviation : #1: 7.77 #2 : 7.78 #3: 7.74 i know the average is 7.76 ...
A: Not
Q: Classify each of the following compounds as ionic (I), molecular (M), binary acid (BA), or oxyacid (...
A: This question is related to inorganic compounds and such compounds are classified into ionic, molecu...
Q: 90 gm glucose dissolve in 500 g water. Molality of the solution is Select one: a. 4 b. 1.5 c. 1 d. 2
A: Number of moles is calculated by dividing the given mass with molecular mass.
Q: An aqueous solution with 15 g sulfuric acid is dissolved to make 0.2 F solution. The specific gravit...
A: We are authorised to solve only first three subparts of the question. Please post rest of the subpar...
Q: Distinguish three (3) differences between ionic and covalent bonding with an example of it.
A: Ionic bonding and covalent bonding has many differences.
Q: Consider the following reaction CH3CH2CO,CH2CH3 (g) H20 (g) CH;CH,CO>H (g) CH;CH2OH (g) Kp = 0.236 a...
A:
Q: Solving an enthalpy of neutralization problem. (see Chemistry 2e, Example 5.5): when 47.35 g of wate...
A: Given: Mass of water = 47.35 g. Initial temperature of water = 22.6 oC And final temperature of wate...
Q: QUESTION 7 Solid Sodium Chloride is usualy separated out of a mixture by: O Dissolving it in water U...
A: ANSWER : Option (a) Solid sodium chloride is usually separated out of a mixture by "Dissolving it in...
Q: Exercise 18 Find the density of F2 gas at 20.0°C and 188 Torr. [Answer: 0.39 g dm]
A: Given 13) P1 = 90 kpa , V1 = 20 dm3 , T1 = 30°C = 303 K P2 = 250 kpa , V1 = ? , T1 = 20°C = 293 K...
Q: What is the percent ionization of a 0.0289 M aqueous solution of cyanic acid, HCNO (K, = 3.5E-4)? i ...
A: The ionisation of weak acid are very low. The percentage of ionisation can be calculated as follows
Q: Why would 1-iodo-2-nitrobenzene be difficult to synthesize using EAS? (EAS= aromatic electrophilic s...
A:
Q: 1. Name the three classifications of compound microscope. 2. Differentiate magnification and resolut...
A:
Q: Formula of the unknown iron fluoride hydrate
A: Mass of hydrate = (30.896 - 29.986) g = 0.91 g Mass of anhydrous salt = (30.456-29.986) g = 0.47 g M...
Q: Differentiate the structure of a crystalline solid and amorphous solid. Give example of their struct...
A: The answer is as follows:
Q: 6. The chloride of an unknown element, X, is a liquid at 20 °C. This chloride reacts with water, giv...
A: A question based on periodic trends and properties that is to be accomplished.
Q: Aqueous sulfuric acid (H,SO,) will react with solid sodium hydroxide (NaOH) to produce aqueous sodiu...
A:
Step by step
Solved in 2 steps with 1 images

- Cadaverine (1,5-diaminopentane) and putrescine (1,4-diaminobutane) are two compounds that are formed by bacterialaction and are responsible for the odor of rotting flesh. Drawtheir structures. Suggest a series of reactions to synthesize pu-trescine from 1,2-dibromoethane and any inorganic reagentsTwo moles of organic compound ‘A’ on treatment with a strong base gives two compounds ‘B’ and ‘C’. Compound ‘B’ on dehydrogenation with Cu gives ‘A’ while acidification of ‘C’ yields carboxylic acid ‘D’ with molecular formula of CH2O2. Identify the compounds A, B, C and D and write all chemical reactions involved.Compound A is an alcohol that undergoes oxidation to produce compound B.Compound B is a ketone that gives positive triiodomethane reaction. Compound B isthen reacted with phenyl magnesium bromide, C6H5MgBr in the presence of aqueousacid to form compound C. Compound C has the molecular formula of C9H12O. Deducethe structure for compound A, B and C. PLEASE PROVIDE CLEAR DRAWINGS AND EXPLANATIONS
- Grignard reagent is a versatile tool in synthetic organic chemistry. Using bromocyclopentane as a starting material, show how a Grignard reagent, X, is synthesized. Reaction of X with water produces compound Y while treatment in carbon dioxide followed by hydrolysis forms compound Z. 3-methyl-2butanone reacts with X and hydrolyses to yield compound AA. Draw the structural formulae of compounds Y, Z and AA and write the chemical equations respectively.Compound A has the molecular formula C14H25Br and was obtained by reaction of sodium acetylide (HC≡CNa) )with 1,12-dibromododecane. On treatment of compound A with sodium amide, it was converted to compound B (C14H24). Ozonolysis of compound B gave the diacid HO2C(CH2)12CO2H. Catalytic hydrogenation of compound B over Lindlar palladium gave compound C (C14H26), while hydrogenation over platinum gave compound D (C14H28). Sodium-ammonia reduction of compound B gave compound E (C14H26). Both C and E yielded O═CH(CH2)12CH═O on ozonolysis. Assign structures to compound A through E so as to be consistent with the observed transformations.(a) Write the structures of main products when aniline reacts with the following reagents :(i) Br2 water (ii) HCI (iii) (CH3CO)2O/pyridine(b) Arrange the following in the increasing order of their boiling point :C2H5NH2, C2H5OH, (CH3)3N(c) Give a simple chemical test to distinguish between the following pair of compounds : (CH3)2NH and (CH3)3N
- The sex attractant of the housefly has the formula C23H46. When treated with warm potassium permanganate, this pheromone gives two products: CH3(CH2)12COOH and CH3(CH2)7COOH. Suggest a structure for this sex attractant. Explainwhich part of the structure is uncertain4. An organic was subjected to sodium fusion and the resulting Lassaigne’s extract boiled with FeSO4 and acidified with concentrated H2SO4, yielding a Prussian blue color. Give the structure of one organic compound which fits this description?Show how to convert carboxylic acids to other functional groups, and devisemultistep syntheses using carboxylic acids as starting materials and intermediates.Explain how acid chlorides are used as activated derivatives of carboxylic acids
- Give the expected organic product when phenylacetic acid, PhCH2COOH, is treated with reagent Q.)LiAlH4 followed by H2OCompound A is a branched-chain alcohol that undergoes oxidation to produce compound B. Compound B is a ketone that gives positive triiodomethane reaction. Compound B is then reacted with phenyl magnesium bromide, C6H5MgBr in the presence of aqueous acid to form compound C. Compound C has the molecular formula of C11H16O (i) Deduce the structure for compound A, B and C. (ii) State the observation when compound C is added with acidified potassium dichromate(VI).An organic was subjected to sodium fusion and the resulting Lassaigne’s extract boiled with FeSO4 and acidified with concentrated H2SO4, yielding a Prussian blue color. Give an example of a compound fits this description?

