Q: DHA is a fatty acid derived from fish oil and an abundant fatty acid in vertebrate brains.…
A: One molecule of DHA undergoes ozonolysis to obtain following products.
Q: What is the degree of unsaturation for C6H8? Draw two possible structures.
A:
Q: Cyclopropane and cyclobutane have similar strain energy despite the fact that the C—C—C bond angles…
A: INTRODUCTION: Structures of cyclopropane and cyclobutane are shown below:
Q: Explain the process of Calculating Degrees of Unsaturation ?
A: Unsaturation in the compound is the measure of the double bonds or the rings present in the…
Q: How to tell if a cluster is coordinatively saturated or unsaturated? For a example the structure of…
A: Meaning of coordinatively saturated means compound have single bond and unsaturated coordinatively…
Q: Knowing that cyclohexane and hexene are isomers, suggest 2 methods allowing their separation.
A: Cyclohexane on heating will break open and behave like open chain saturated compound. It will give…
Q: Assuming standard valencies (C = 4, H = 1, N = 3, 0 = 2, and X = 1), a compound with molecular…
A:
Q: The potential energy of a CH3 group in ethane as it is rotated around the C-C bond can be written V=…
A: For fully eclipsed conformation, Angle = 1800 V= 12V0(1 +cos(1800) =0
Q: Consider 1-bromopropane, CH3CH2CH2Br. (a) Draw Newman projections for the conformations in which…
A: Newman projection: These are one of the ways to explain stereochemistry of alkanes. The…
Q: Draw and name the six isomeric cyclopentane of molecular formula C7H14. These will include four…
A: The isomers are as shown below: There are 4 constitutional isomers of cyclopentane with the same…
Q: Perform a conformational analysis of 2-methylbutane, (CH3)2CHCH2CH3, looking down the C2-C3 bond.…
A: The different structures obtained by the rotation of C3 about the C2- C3 bond by 600 are shown…
Q: Identify the s-trans conformation of (3Z,5Z)–4,5-dimethyldeca–3,5–diene.
A: E,Z nomenclature is given to double bond in which each olefinic (double bond) has different groups.…
Q: Calculate the index of hydrogen deficiency for 1-hexene, C6H12, and account for this deficiency by…
A: Formula to calculate the index of hydrogen deficiency (also known as the degree of unsaturation):…
Q: What is the degree of unsaturation of C8H10ClNO? a. 4 b. 5 c. 6 d. 7
A:
Q: Explain conformational analysis of cyclohexane.
A: The molecular formula of cyclohexane is C6H12. The planar structure of cyclohexane has bond angle of…
Q: Describe and explain the stereochemistry of 2-bromo-1-chloropropane molecule
A: for an optically molecule, pair of enantiomers exists.
Q: Measure the C-C-C and C-C-H bond angles in the energy-minimized models of cis and trans isomers of…
A: The structures of cis and trans isomers of cyclooctene are as follows:
Q: 1. Calculate the degrees of unsaturation for the molecular formula C6H12O and draw two possible…
A: degree of unsaturation is equal to the total number of π-bonds and rings present in an organic…
Q: Calculate the index of hydrogen deficiency of cyclohexene, C6H10, and account for this deficiency by…
A: Multiple and rings reduces the hydrogen count by 2 and it is called degree of unsaturation or index…
Q: Draw a molecular orbital picture for the LUMO of 1,3-Butadiene
A:
Q: Write structural formulas for toluene (C6H5CH3) and for benzoic acid (C6H5CO2H) (a) as resonance…
A:
Q: Brainberene, a compound isolated from an organic chemist’s brain, has the molecular formula C16 H26.…
A: The molecular formula of Brainberene is C16 H26. When Brainberene is subjected to catalytic…
Q: how many possible consitutional isomers are there for C2H4Cl2?
A: Isomer are pair of compound which have same molecular formula but different structural formula…
Q: What is the saturation pressure of benzene at 700°C?
A:
Q: Indole smells terrible in high concentrations but has a pleasant floral-like odor when highly…
A: Huckel's rule for aromaticity: The compound must be cyclic planar geometry with (4n+2) pi electrons.…
Q: b) Would the conformational energy of the 1,3- and 1,4 - isomers of dimethylcyclohexanes be similar…
A:
Q: Treatment of cyclohexene with C6H5CHI2 and Zn(Cu) forms two stereoisomers of molecular formula…
A: The given reaction is an example of a Simon-smith reaction in which the reaction of an alkene with…
Q: What is the IR stretching frequencies of C-C stretches of alkenes and alkynes? What are their…
A:
Q: :Regarding 1,3-butadiene When substituted by two phenyl groups the cis- conformation wavelengths…
A: Given molecule is: 1,3-butadiene. How the substitution on 1,3-butadiene effect its absorption in…
Q: Does a unit of unsaturation of 5 signify anything?
A: The degree of unsaturation or hydrogen deficiency can be mathematically expressed as follows: Degree…
Q: Calculate the number of degrees of unsaturation in a compound ofmolecular formula C4H6, and propose…
A: Unsaturated compounds are those which contain double bonds or triple bonds. Unsaturated compounds…
Q: (b) Discuss the effect of ring strain on the internal C-O stretching in cyclic alkenes: ke tones
A: The main use of IR spectroscopy is in organic and inorganic chemistry. It is used by chemists to…
Q: Measure the C-C-C and C-C-H bond angles in the energy-minimized model of cyclohexene and compare…
A: In the energy-minimized model of cyclohexene, for C-C-C and C-C-H bonds, the possible bond angles…
Q: 3. use the molecular formula C4H8O2 to illustrate the following isomers: a) (+)-gauche and…
A: The chemical formula of the unknown compound provides information about the nature of the elements…
Q: Build the molecular models of cis-1,2-dichlorocyclopentane and trans-1,2-dichlorocyclopentane. Then…
A: Cis-1,2-dichlorocyclopantane and trans-1,2-dicyclopantane are enantiomers or superimposed mirror…
Q: An unknown compound of molecular formula C5H10O2, gives the HNMR data shown below. Which of the…
A:
Q: Show that the structural formula of vitamin A can be divided into four isoprene units bonded…
A: Isoprene is a common organic compound with the formula CH2=C(CH3 )−CH=CH2. Isoprene is an…
Q: Are the C¬C bonds cyclopropane weaker than those in cyclohexane?
A: To understand the bond strength in cycloalkane and to compare them in case of cyclopentane and…
Q: Calculate the units of unsaturation of C6H7NO. And draw out the
A: We have to Calculate the units of unsaturation of C6H7NO
Q: How could you distinguish between cyclohexane and 2-hexene C6H12 using UV-vis spectrum?
A: Note: cyclohexane having only sigma electrons. Therefore σ → σ* are takes place around 135 nm.…
Q: Determine the densities of the xylene isomers (o, p, and m) at 40°C.
A:
Q: What is the index of hydrogen deficiency of Methylcyclohexene? O 2
A: Option B is Correct Answer.
Q: AROMATIC HYDROCARBONS Properties MULTIPLE RING SYSTEMS White volatile crystals with Moth repelants…
A: COMPOUND-NAPTHALENE Properties of Naphthalene: The actual properties and substance properties of…
Q: Measure the C=C-C bond angles in the energy-minimized models of the cis and trans isomers of…
A: Central carbon atom (sp2 hybridized) of C=C-C has three electron domains. Hence, according to VSEPR…
Q: what is the difference between cyclobutanol and cylcobutane when observing their ring strain…
A: Given: cyclobutanol and cylcobutane To find: find the difference between cyclobutanol and…
Q: Identify the s-cis conformation of (3Z,5Z)–4,5–dimethyldeca–3,5-diene.
A: Given s-cis conformation = To be determined
Q: Draw the structural formula for at least one bromoalkene with the molecular formula C5H9Br that…
A:
Q: Which of the following in each pair is likely to be moresoluble in hexane, C6H14: (a) CCl4 or CaCl2,…
A: Since hexane is a non polar molecule hence the molecule which will have less polarity will be more…
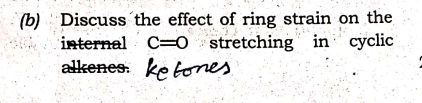
Step by step
Solved in 2 steps with 2 images

- Write structures for products A, B, C, and D showinf stereochemistry. (Hint: B and D are stereoisomers)A compound C4H11N is known from its reactivity andspectroscopic properties to have no hydrogen atomsattached directly to the nitrogen atom. Write all structuralformulas consistent with this informationOn being heated with a solution of sodium ethoxide in ethanol, compound A (C7H15Br) yielded a mixture of two alkenes B and C, each having the molecular formula C7H14. Catalytic hydrogenation of the major isomer B or the minor isomer C gave only 3-ethylpentane. Suggest structures for compounds A, B, and C consistent with these observations.
- Having one mole of carbon suboxide (C2O3) means that you haveExplain the Mechanism - Addition of Dichlorocarbene to an Alkene ?A difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired conguration.a. Label this stereogenic center as R or S.b. A well known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship.c. Suggest a reagent to convert A to the single stereoisomer X.
- What is the degree of unsaturation for compoundC11H7ClO?Compound A, C9H16 reacts with 1 molar equivalent(s) of hydrogen on catalytic hydrogenation. A undergoes reaction with ozone, followed by Zn treatment. What is the structure for A?A difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired configuration. a. Label this stereogenic center as R or S. b. A well known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship. c. Suggest a reagent to convert A to the single stereoisomer X.
- A difficult problem in the synthesis of PGF2α is the introduction of the OH group at C15 in the desired configuration. a. Label this stereogenic center as R or S. b. A well-known synthesis of PGF2α involves reaction of A with Zn(BH4)2, a metal hydride reagent similar in reactivity to NaBH4, to form two isomeric products, B and C. Draw their structures and indicate their stereochemical relationship. d. Suggest a reagent to convert A to the single stereoisomer X.A chiral alkyne A with molecular formula C6H10 is reduced with H2 and Lindlar catalyst to B having the R conguration at its stereogenic center. What are the structures of A and B?The bicyclic alkene P can be prepared by thermal electrocyclic ringclosure from cyclodecadiene Q or by photochemical electrocyclic ringclosure from cyclodecadiene R. Draw the structures of Q and R, andindicate the stereochemistry of the process by which each reactionoccurs.

