Q: Rotate 180 degrees around the C3-C4 bond. Redraw the molecule with the left side of the molecule…
A: Given,
Q: A single bond, compared to a double bond, A) is longer. B) is stronger. C) has a higher bond…
A: A covalent bonding between two atoms can be either a single bond, double bond or a triple bond. A…
Q: Whyπ bonding restricts rotation around double bonds
A: In double bonds two types of bonds are present namely sigma and pi-bonds. π bonds are formed by…
Q: H .C-C=C-C H C- H
A:
Q: Which of these structures shows how the pictured molecule will look after it is flipped over…
A:
Q: Draw the molecule by placing atoms on canvas and connecting them with bonds H2CCCl2
A: Answer:- This question is answered by using the simple concept of writing the Lewis structure which…
Q: Draw the line-angle structure for each of the following molecules and then name them.
A: Draw the line-angle structure for each of the following molecules and then name them.
Q: The cyclohexane chair has the headrest to the right and the footrest to the left. Draw a cyclohexane…
A:
Q: Q1// Draw the position isomer for the chemical formula ? A. C;H,Br В. С.Ни
A:
Q: Which molecules show an appropriate number of bonds around each carbon atom?
A: A question about carbon, which is to be accomplished.
Q: How can draw 3D representation with bond and molecular dipoles? CH4 ClO4^- PF3
A: The dipole moment lies along the bond and points from the electropositive atom to the…
Q: Circle any/all of the following that exhibit dipole-dipole attraction between molecules? Please…
A: Welcome to bartleby ! Introduction : We have to check which molecule contain dipole
Q: (a) Which dipole pair shown below has the lower energy? (b) If you double the distance between the…
A: When same charges comes to each other then they repel each other and energy will increase. When…
Q: Answer the following question about acetonitrile (CH3C≡N:). Question: In what type of orbital does…
A: Lone pair is defined as the pair of electrons that is present in an orbital corresponding to atom or…
Q: d. What is the dihedral angle for the Br-C-C plane and the tBu-C-C plane at position F in the energy…
A:
Q: When you know the overlapping orbitals in the cross, it can be known A- Geometric shape Angle values…
A: In this question, we will see that the overlapping orbitals in the crossed what is meant. You can…
Q: Which of the following pairs of electrons have the highest repulsion?
A: a
Q: Draw dot and cross diagram for A, B, C, D Draw the shape of A, B, C, D and propose values for bond…
A: Lewis dot structure - It is the structural representation of the molecule which is shown by the…
Q: Benzene is especially stable due to... the electrons of the double bonds are delocalized O its…
A: The delocalization of electrons provide extra stability to the compounds.
Q: Which of the following best exhibits hydrogen bonding? Explain your answer. H2Se CH. HC1 NH3
A: Different type of compounds/Molecules have different type of chemical forces and intermolecular…
Q: which of these five atoms will bond with the fewest number of hydrogen atoms, and which will bond…
A: Every atom wish to achieve nearest noble gas configuration and for this, it either loses electrons…
Q: Q2) arrangement the following compound according to increase the Boiling point with reason?…
A: Boiling point is the temperature at which any liquid vaporizes is called boiling point of that…
Q: 10 draw the condensed and line bond structure of the compound
A:
Q: π
A: In first Compound CO : it's have triple bond so this Compound contain one sigma and two pi bond.…
Q: For parts A, B, and C label each carbon in the strucutres with the correct hybridization.
A:
Q: Can the highlighted labeled bond rotate 180° O yes without breaking any bonds in the molecule? O no…
A: Symmetry operation: It is a structural action that takes place in molecule so, that when any plane…
Q: Draw all possible isomers having molecular formula C4H8 that contain one π bond.
A: Isomers refers to the molecules or ions having same identical molecular formulas but differ only on…
Q: Draw a skeletal (ine-bond) structure of octane. Select to Draw
A: The solution of above question is-
Q: The antianxiety drug Diazepam (Valium) is shown. a) Label the type of orbital that each of the…
A:
Q: ent bond that always rotates freel
A: Double bond have both sigma and pi bond and it have less covalent bond character than ionic bond…
Q: Classify the folloiwng chairal molecules as R or S а)
A:
Q: Draw two five carbon rings that share an atom.
A: When two rings share only one carbon atom then such compounds are known as spirocyclic compounds.
Q: A cumulene has double bonds directly connected to each other. Identify all the atoms that must lie…
A: The structure given is an allene which always has two double bonds. sp hybridization: This type of…
Q: Describe why a carbon atom (C) is capable of forming fourcovalent bonds.
A: Covalent bond: It is defined as the chemical bond in which atoms share electron pairs with each…
Q: Explain why Circle the molecule below with the lower boiling point NH2
A:
Q: Arrange the following in order of increasing boiling point: RbF, CO2, CH3OH, CH3Br. Explain your…
A: From given Factors which affect boiling point is discussed and each compound is associated with that…
Q: Identify the most polar bond in each molecule. Q) HSCH2CH2OH
A: In modern periodic table element are arranged in the increasing order of their atomic number. S…
Q: common name for the skeletal (line-bond) structure shown here.
A:
Q: Please draw molecule directions are in photo
A: The molecular formula of the unknown is C6H14O. The double bond equivalence of the compound can be…
Q: How many (non H) axial positions are shown in the following molecule
A: In this question, you will find-out the total number of axial position without axial hydrogen (…
Q: Which molecule would be more polar, CH3F or CH3Cl? And why?
A:
Q: What is the F-B-F bond angle in BF3?
A: VSEPR theory:The structure of a molecule can be determined by using the VSEPR theory. According to…
Q: What is the dipole direction in H-D?
A: Answer - Dipole direction - The electric dipole moment, a vector, is directed along the line from…
Q: what is the geometry at the carbon for the picture?
A: Hybridization is defined as the phenomenon of orbital mixing to form new hybrid orbitals that are…
Q: Answer the following question about octocrylene (see Attachment), a common sunscreen component.…
A: The Lewis structure does not give any idea about molecular geometry but it purposed the distribution…
Q: İl classes of compouds in
A:
Q: Rank the indicated bonds from shortest to longest in length. a Shortest
A: There are different types of bond , Single bond , double bond and triple bond.
Q: what is the geometry around the double bonds ? from left to right
A: What is the geometry around the double bonds from left to right ?
Q: Srmine the number of unshared pairs at each designated atom Be sure your answers are consistent with…
A: In compound 1: atom a is carbon having 4 valence electrons which is shared with hydrogen and sulphur…
Q: 4. Draw dipoles for each of the polar bonds in the molecules below: a. b. Но Но
A: When there is a Diffarence in electronegativity of atoms ,then the bond formed by those atoms are…
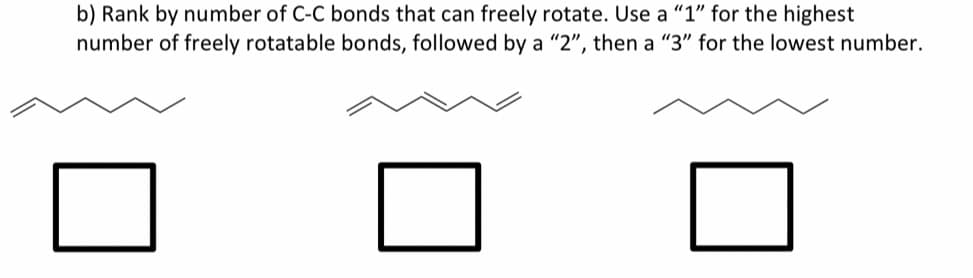
Step by step
Solved in 2 steps with 1 images

- Please refer to the Newman projections shown below to when answering questions (a) – (f)Draw an exact 3-D representation of the molecule from the model. please answer all question step by step. Answer follow image: Please must mention: valency electron, molecular geometry handwritten answerDraw the potential energy of the molecule circled at the top. Draw the other 3 Newman projections based on the one already drawn by rotating the back carbon clockwise. Explain your answers
- Why ester C=O has higher stretching frequency than amide?Complete this graph of relative potential energy vs. rotation of the C1C2 bond of ethane. Thepoint drawn for you indicates the P.E. at 0° (eclipsed).Draw an exact 3-D representation of the molecule from the model. Please must mention: valency electron, molecular geometry
- Which of the attached is (are) possible Newman projections for 2-methylpentane?Use flat representation of rings, not chair in the drawing. Determine the most and least stable. Consider the most stable chair for each of these isomers, and then draw the most stable and least stable isomer based on a comparison of the best chair for each one.Then draw the most stable and least stable Newman projection conformation from the C4-C5 bond in the molecule above

