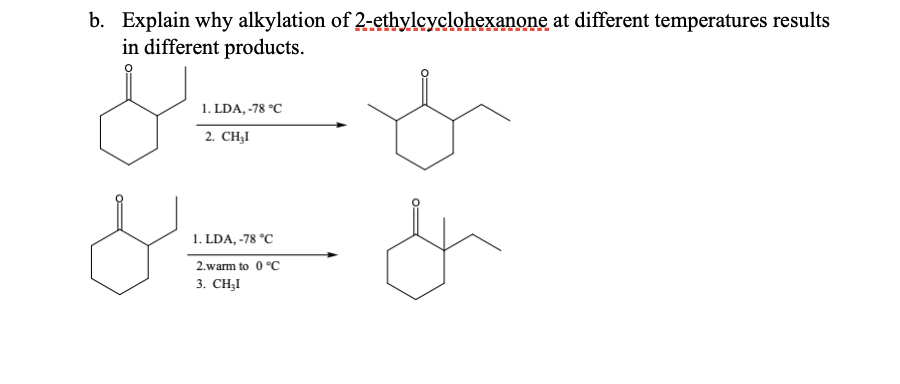
b. Explain why alkylation of 2-ethylcyclohexanone at different temperatures results in different products. 1. LDA, -78 °C 2. CHI 1. LDA, -78 °C 2.warm to 0°C 3. CH₂I
Q: PROBLEM How might you carry out the following transformation? More than one 20-12 step is needed.…
A: The objective of the question is to understand the chemical transformation process of converting a…
Q: I completed an experiment "Iron contenr of fruit and vegetable through complex ion formation" How…
A: The number of moles of a substance is the amount of a substance in which there are 6.022×1023…
Q: 23. H
A: Wheb ethanol reacts with isobutyric anhydride, it forms ethyl isobutyrate and isobutyric acid as…
Q: Calculate theorical yield and % yield of the Synthesis of 2,5-Dimethyl-1-phenylpyrrole using aniline…
A: To calculate the theoretical yield and % yield of the synthesis of 2,5-Dimethyl-1-phenylpyrrole…
Q: None
A:
Q: The enthalpy of vaporization of Substance X is 18.0- and its normal boiling point is -93. °C.…
A: Step 1: Convert the initial and final temperatures from °C to Kelvin.Step 2: The relation between…
Q: Write the Ksp expression for the sparingly soluble compound silver sulfide, Ag2S.Ksp = BLANK/BLANK…
A: The objective of the question is to write the Ksp expression for the sparingly soluble compound…
Q: 1. Predict the elimination products of the following reactions. When two alkenes are possible,…
A: The objective of the question is to predict the elimination products of the given reactions and to…
Q: Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any…
A:
Q: The active ingredient of bleach such as Clorox is sodium hypochlorite (NaClO). Its conjugate acid,…
A: Given:The word pH stands for the potency of hydrogen and it is used to measure the concentration of…
Q: None
A:
Q: What would be a reasonable guess for the wavelength of maximum absorbance for FD&C Orange 2?…
A: The objective of the question is to determine the wavelength of maximum absorbance for FD&C…
Q: The Solubility Product Constant for barium chromate is 2.0 x 10^-10 . The maximum amount of barium…
A: The objective of this question is to find the maximum amount of barium chromate that will dissolve…
Q: Decrease volume Add Heat Left Left END Left Remove HCl(aq) Right Right Left Right Decrease pressure…
A:
Q: None
A: Buffers play a crucial role in maintaining the pH of a solution, and understanding their operation…
Q: Potassium metal reacts with water to form potassium hydroxide and hydrogen gas according to the…
A: The objective of the question is to calculate the pressure of hydrogen gas in the container. The…
Q: Sn2 and N (starting concentration 10 mol/L) should be separated by fractionated precipitation using…
A: Step 1: To solve this problem, we can use the solubility product constants to determine the…
Q: Question 5. (20 points) Consider the following reaction. + PhLi (2 eq.) then water a) Propose a…
A: The objective of the question is to understand the reaction mechanism involving PhLi…
Q: None
A: Step 1:This is a subsitution reaction. Br is a good leaving group. We also have a primary alkyl…
Q: What is the name of this structure?
A: Step 1: Step 2: Step 3: Step 4:
Q: Draw a stepwise mechanism for the following reaction, a key step in the synthesis of ticlopidine, a…
A: The objective of the question is to find the stepwise mechanism of the following reaction of…
Q: The following results were obtained for an enzyme-catalyzed reaction Substrate concentration (mmol…
A: The objective of this question is to calculate the Michaelis-Menten constant (Km) and the maximum…
Q: PROBLEM Acid-catalyzed hydrolysis of a nitrile to give a carboxylic acid occurs by 20-25 initial…
A:
Q: Calculate the pH of solutions of the following weak acids. This one is bit tricky. Think carefully…
A: Calculate the pH of a solution of a weak acid, using the dissociation of acid and equilibrium…
Q: What volume in liters, L, of a 0.850 M solution can Jorge prepare from 0.350 L of a 3.00 M solution?…
A: The objective of this question is to find out the volume of a 0.850 M solution that can be prepared…
Q: Which gas will diffuse the FASTEST under the same conditions? F2 CH2Cl2 COC12 Ar
A: The objective of the question is to determine which among the given gases - F2, CH2Cl2, COC12, Ar -…
Q: he titration of 20.0-mL sample of 0.105 M Succinic Acid (Ka= 6.16 X10^-5) with 0.125 M NaOH. The…
A:
Q: Fill in the boxes. One molecule per box. If the molecule is chiral, be sure to specify if it is…
A:
Q: Show reaekson and don't use hend raiting and step by step solutions
A:
Q: A 54.0 L container holds 6.27 moles of oxygen gas, O2, and 4.19 moles of neon gas, Ne, when the…
A: The objective of this question is to calculate the pressure inside a cylinder containing oxygen and…
Q: 21. For a reaction, what generally happens if the temperature is increased? A) A decrease in k…
A:
Q: How do you solve the 1s and 2pz hydrogenic wavefunctions for HF?
A: To solve the 1s and 2pz hydrogenic wavefunctions for hydrogen fluoride (HF), we can use the general…
Q: None
A: Step 1: Step 2: Step 3: Step 4:
Q: A solution is prepared at 25 °C that is initially 0.41 M in chloroacetic acid (HCH2CICO2), a weak…
A:
Q: Draw the structure for O2 and then complete the following sentences regarding its structure. This…
A: Lewis Structure of O2 The Lewis structure for O2 shows two oxygen atoms connected by a double bond.…
Q: None
A: Part 2: Explanation:Step 1: Identification of Functional Group:The presence of "CO₂H" suggests the…
Q: None
A:
Q: Draw the most likely conjugate base resulting from this acid-base reaction. Include all lone pairs.…
A: Step 1: When a proton is removed from hexanenitrile, it becomes its conjugate base (also known as…
Q: Need full mechanism
A: The objective of the question is to provide the mechanism.
Q: None
A:
Q: The Solubility Product Constant for barium phosphate is 1.3 x 10^-29 . The maximum amount of barium…
A: Given:Ksp of Ba3(PO4)2 = 1.3 × 10⁻29Find:Solubility of Ba3(PO4)2 in presence of common…
Q: Sample weighing 7.52-g was dissolved in enough water and diluted to 250.0-mL. 100.0-mL of the…
A: The objective of the question is to determine the purity of the sample used in the given…
Q: None
A: The stepwise approach is indicated in the question's answer.
Q: 11. Supply both sets of missing reagents and the intermediate. (6 pts)
A: Step 1:Friedel-Crafts Acylation: Benzene can be acylated using an acyl chloride (such as propanoyl…
Q: A certain half-reaction has a standard reduction potential Ered = -0.08 V. An engineer proposes…
A:
Q: Draw the first round of beta -oxidation for lauroyl-CoA
A: Approach to solving the question: Detailed explanation:The first round of beta-oxidation for…
Q: The vapor pressures of CCl4 (A) and C₂HC 3 (B) between T = 350 and 360 K, can be determined…
A: The objective of the question is to calculate-The mole fraction of in Liquid PhaseThe mole fraction…
Q: Name the following compound then draw the compund in wedge and dash structure form
A:
Q: None
A: Approach to solving the question:1. Calculate the moles of NaH2PO4 and Na2HPO4.2. Determine the…
Q: Be sure to answer all parts. In a first-order decomposition reaction, 50.0% of a compound decomposes…
A: Given,50.0% of a compound decomposed in 18.5 minutes.Thus, half-life (t1/2) = 18.5 min
Step by step
Solved in 2 steps with 1 images

- Explain why pentane-2,4-dione forms two different alkylation products (Aor B) when the number of equivalents of base is increased from one totwo.How does each of the tripling [RX] changes affect the rate of an E2 reaction?For problem 8.17, all of the reactions will be SN2. For each reaction, identify and evaluate each nucleophile (strong? weak? Strong or weak as a base?) Also, evaluate each solvent as polar protic or polar aprotic. I recommend drawing the structure of each solvent.
- The following reaction takes place several times faster than the reaction of 2-chlorobutane with HO-: a. Explain the enhanced reaction rate.b. Explain why the OH group in the product is not bonded to the carbon that was bonded to the Cl group in the reactant.Use the appropriate reagent used in the reaction. (d),(e) please!!organic chemistry 37) The most reactive towards nucleophilic acyl substitution is:
- For the reaction scheme below: (a) Suggest a reagent X that could be used to accomplish the first step. (b) Draw the curly arrow mechanism for the second step in the scheme.This is Wittig Rxn: Whatever mechanism you choose to draw is fine since you can leave the base as B: Draw the arrow pushing mechanism using the compounds below – Constant: 4-nitrobenzyl benzaldehydebase 1: triethylaminebase 2: NaOHbase 3: K2CO3ylide: Acetonyltriphenylphosphonium chloride1.Draw the product, please shows step by step in details 2.Explain if (CH3)2CH in this reaction can attach in ortho positon in Friedel Craft Alkylation?? why or why not???

