Q: Which of the following reactions of benzene requires a Lewis acid? a Bromination b FC…
A: Given statement is : Which of the following reactions of benzene requires a Lewis acid? Options…
Q: Calculate the volume in liters of a 4.30 x 10 -5 mM mercury(II) iodide solution that contains 100.…
A: 4.30 ×10-5 mM mercury(II) iodide solution means , 4.30 ×10-5 mmol = 4.30×10-8 mol mercury(II) iodide…
Q: A buffer is prepared by mixing 206 mL of 0.452 M HCl and 0.500 L of 0.400 M sodium acetate,…
A: Given: Molarity of HCl = 0.452 M Volume of HCl = 206 mL Molarity of sodium acetate = 0.400 M Volume…
Q: Cyclohexene can undergo strong oxidation with potassium permanganate in acidic solutions and yield a…
A: Cyclohexene can oxidize into adipic acid on react with potassium permanganate in acidic solutions
Q: What is the least favorable cation to reduce on the Table of Std. Reduction Potentials? Ca2+ Li+…
A:
Q: Calculate the solubility at 25 °C of Zn (OH), in pure water and in a 0.0100M ZnSO, solution. You'lIl…
A: From alesk data, Ksp of Zn(OH)2=3.0×10-17 Molar mass of Zn(OH)2=99.424 g mol-1
Q: Calculate the pH after addition of 0.00, 5.00, 15.00, 25.00, 40.00, 45.00, 49.00, 50.00, 51.00,…
A: This is the titration with weak acid and strong base .Where HNO2 is weak acid and NaOH is strong…
Q: 1. Me H2N OEt MeOH, H* Et 2. xylenes, heat 1. H3O*, heat 2. NH O. Meo OMe p-TSOH, PhH, reflux 3.…
A: 1. Fischer Indole synthesis. 2. Ketal to ketone. Ketone to imine. Imine reduction. 3. O-Bromination,…
Q: What type of intermolecular force causes the dissolution of RbBr in water? O dispersion forces O…
A:
Q: 3. Suggest experimental means by which the rates of the following reactions could be followed. There…
A: In the question it is given to discuss the experimental means by which the rates of the reaction…
Q: What is the IUPAC name of the following compound? ball & stick v + labels
A:
Q: In the system: C(s) + O2(g) + 2HCI(g) – COCI2(g) + H2O(g) + heat how is it possible to increase the…
A: Because heat is in product side hence if we increases amount of reactants C(s) , O2(g) , HCl(g) then…
Q: Determine the minimum volume of 5.7 M HNO3 required to completely dissolve the sample. Express your…
A: Part E : ) first we would Calculate moles of Cu , then using mole ratio we can calculate moles of…
Q: Which of the following is directly related to reaction rate? A. Concentration B. Pressure C. Time D.…
A: Which of the following is directly related to reaction rate ? A. Concentration B. Pressure C.…
Q: The concentration of CO- in a saturated solution of CuCO, is 1.60x10- mol/L at a certain…
A: * Solubility product is the product of molar concentrations of ion present in solution, when ion…
Q: The third law of thermodynamics is defined as a. the combined mass and energy of the universe…
A: -> at absolute (0K) temperature all the modes of motion stops (no vibration , no translation and…
Q: What happens at the anode in the voltaic cell? Cathode?
A: A voltaic cell is an electrochemical cell that uses a chemical reaction to generate electrical…
Q: The average body temperature of a chicken is 106 degrees Fahrenheit (much warmer than humans!).…
A: Answer: This question is based on ideal gas equation which is shown below: PV=nRTV=nRTP Here:…
Q: What is the sign for each of the following? (Change in) a. S, H, G for the evaporation of a…
A:
Q: A diprotic acid, H, A, has acid dissociation constants of K = 2.32 x 10-4 and K2 = 5.84 x 10-12.…
A:
Q: Use the following data to calculate the value of S° for the reaction given below. Write answer only…
A:
Q: Statins are drugs that have been used to treat high cholesterol by blocking mevalonate synthesis.…
A:
Q: Calculating A chemist prepares a solution of calcium sulfate (CaSO) by measuring out 0.79 g of…
A:
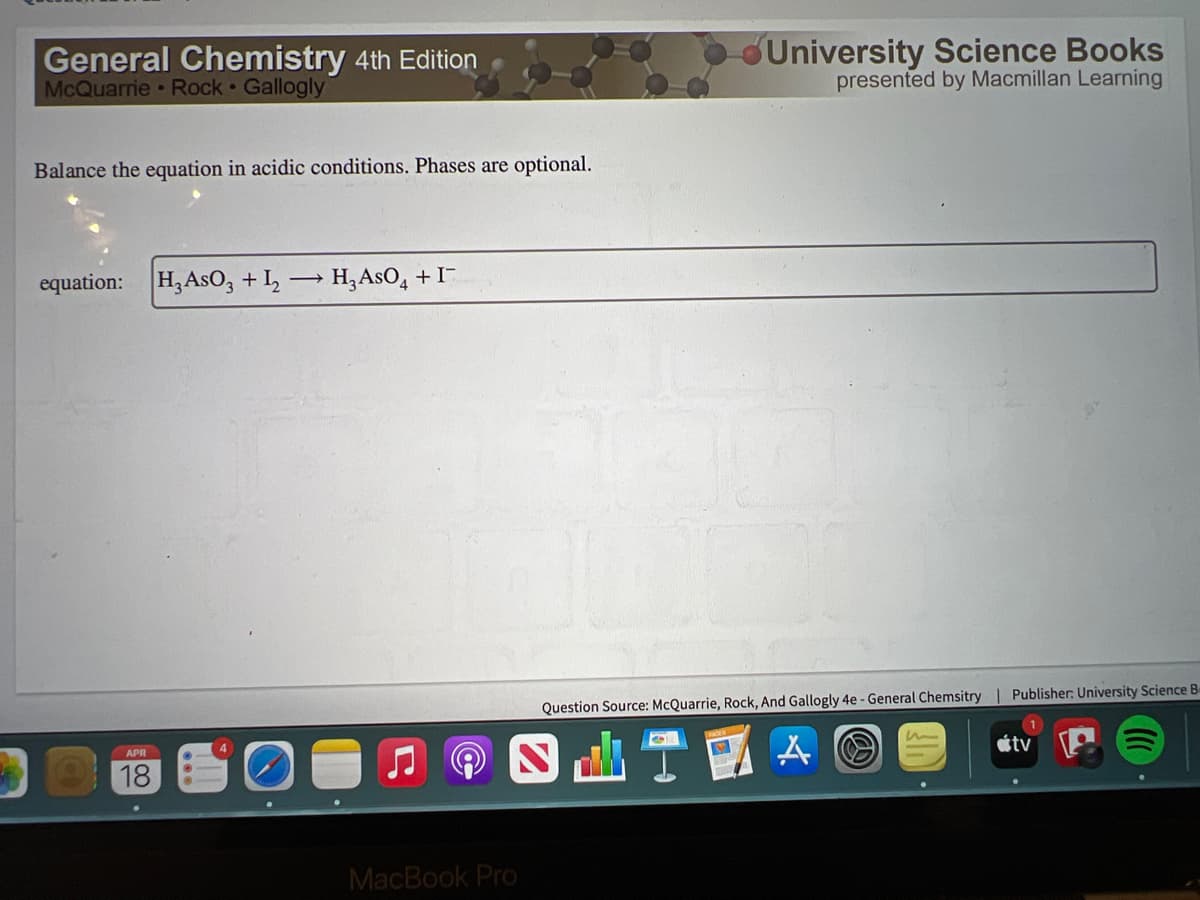
Q: Give a clear handwritten answer...
A: Balanced the given equation--
Q: Using molarity to find solute mass Iculate the volume in liters of a 0.208 mol/L copper(II) sulfate…
A: Given -> Molarity = 0.208 mole/L Weight of CuSO4 = 75.0 gm
Q: In each of the molecules drawn below one chemical bond is colored red. Decide whether this bond is…
A: The polarity of a bond in a molecule can be determined with the help of electronegativity difference…
Q: A 1-butyne can undergo one-time elongation via acetylide formation reaction using sodium amide and a…
A: Note : Terminal alkynes react with base produces the anion and reacts with alkyl halides produces…
Q: Will the following reaction occur spontaneously under standard-state conditions?…
A: Given- 2Al(s)+3Co2+(aq) ->3Co(s)+2Al3+
Q: Describe atomic structure; name and describe the parts of atoms, and their arrangement/location and…
A: The atom is the smallest unit of matter that may be not divided into smallest particle without…
Q: Which solution has the greatest buffer capacity? Which solution can react with the largest amount…
A: Buffer solution: The solution that resists the change in pH is known as a buffer solution. The…
Q: e formula of ionic compounds
A:
Q: make sure reagents are CLEAR write out the mechanism. make sure to include resonance, reagents,…
A: Friedel-Craft alkylation is the importnat reaction for the introduction of alkyl group on the…
Q: Question 11 Given is the mass spectra of n-octane whre peaks are labeled accordingly. Give the…
A: We have to predict the peaks for given questions.
Q: What is the H3O* | and the pH of a benzoic acid-benzoate buffer that consists of 0.13 M C6H5COOH and…
A: Given: Concentration of C6H5COOH = 0.13 M Concentration of C6H5COONa = 0.30 M Ka of C6H5COOH =…
Q: Why does NH3 have a higher boiling point than BH3?To answer this question, draw out the Lewis…
A: The boiling point of ammonia (NH3) is higher than borane (BH3).
Q: Name it とセーBい
A: Naming is Done according To CIP rules in which heavier atom given more priority.
Q: A 3.126 g sample of technetium-95 is sealed in a container. After 44.2 hours, the amount of…
A:
Q: An SN1 reaction is done between 0.25 grams of 2,5- dihydroxy-2,5- dimethylhexane and 2.0mL of 6.0M…
A: Atom economy, E-factor, process mass intensity (PMI) all are important aspects of green chemistry…
Q: Non-ionizing radiation from a variety of sources is used for producing images in humans. Select any…
A: UV, visible light, infrared (IR), microwave (MW), radio frequency (RF), and very low frequency (ELF)…
Q: created Is SO, being created or destroyed by the chemical reaction? destroyed O neither created nor…
A: Given that, the graph of the pressure of the SO2(g) in a reaction vessel during a certain chemical…
Q: A 0.56g sample of gas is contained in 250.0 mL cylinder. It's pressure is 983 mmHg and it's…
A:
Q: Balance the following reactions below using the change in oxidation number method. 1. PbO, + HBr →…
A: Here we have to balance the following chemical equation by change in oxidation number method.
Q: 1a th. Cracemic) Br
A: We have to carry out the given transformations. The required transformations are shown below
Q: Decide whether or not each of the following metals dissolves in 1 M HNO3. For those metals that do…
A: We have to tell what happened when Al is dissolved in 1 M HNO3.
Q: Mass Spectrometry, what high energy particle is responsible is used to bombard molecule so that it…
A: We have to predict the energy particle.
Q: 0.250 mol of N2 is placed into a 0.750 L container. What is the density of the gas? 84.1 g/L 4.67…
A:
Q: H. 1. CHaMgBr 2. Neutralizing work- dn Select to Draw
A:
Q: A chemist titrates 180.0 mL of a 0.4899M potassium hydroxide (KOH) solution with 0.5114M HCl…
A:
Q: A Zn wire and Ag/AgCl reference electrode saturated with KCI (E = 0.197 V) are placed into a…
A: Given data :
Q: Curved arrows are used to illustrate the flow of electrons. Follow the arrows and draw the…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- the free energy for a reaction can be related to the equilibriumconstant through the formula below. K = e (-ΔG° / RT) Therefore if Kc for a reaction is known, Go can be determined, or vice versa. Furthermore, ifyou have the value for Go at two different temperatures, you can calculate H and S throughthe familiar equation for Gibbs energy below, since you have two unknowns but also twoequations. G = H – T S In this lab you will be studying the solubility of borax (Na2B4O5(OH)4*8H2O), a slightly solublesodium salt, at two different temperatures. When solid borax is added to water, theequilibrium below is established. Na2B4O5(OH)4*8H2O (s) <--> 2 Na+ (aq) + B4O5(OH)42- (aq) + 8 H2O(l) If you measure the concentrations for those substances that show up in the reaction quotient,then the Kc for the reaction at that temperature can be calculated. In this lab, theconcentration of borate ion (B4O5(OH)42-) in solution will be measured by titration with standardhydrochloric acid…the free energy for a reaction can be related to the equilibriumconstant through the formula below.K = e (-ΔG° / RT)Therefore if Kc for a reaction is known, Go can be determined, or vice versa. Furthermore, ifyou have the value for Go at two different temperatures, you can calculate H and S throughthe familiar equation for Gibbs energy below, since you have two unknowns but also twoequations.G = H – T SIn this lab you will be studying the solubility of borax (Na2B4O5(OH)4*8H2O), a slightly solublesodium salt, at two different temperatures. When solid borax is added to water, theequilibrium below is established.Na2B4O5(OH)4*8H2O (s) 2 Na+ (aq) + B4O5(OH)42- (aq) + 8 H2O(l)If you measure the concentrations for those substances that show up in the reaction quotient,then the Kc for the reaction at that temperature can be calculated. In this lab, theconcentration of borate ion (B4O5(OH)42-) in solution will be measured by titration with standardhydrochloric acid according to the…the free energy for a reaction can be related to the equilibriumconstant through the formula below.K = e (-ΔG° / RT)Therefore if Kc for a reaction is known, Go can be determined, or vice versa. Furthermore, ifyou have the value for Go at two different temperatures, you can calculate H and S throughthe familiar equation for Gibbs energy below, since you have two unknowns but also twoequations.G = H – T SIn this lab you will be studying the solubility of borax (Na2B4O5(OH)4*8H2O), a slightly solublesodium salt, at two different temperatures. When solid borax is added to water, theequilibrium below is established.Na2B4O5(OH)4*8H2O (s) 2 Na+ (aq) + B4O5(OH)42- (aq) + 8 H2O(l)If you measure the concentrations for those substances that show up in the reaction quotient,then the Kc for the reaction at that temperature can be calculated. In this lab, theconcentration of borate ion (B4O5(OH)42-) in solution will be measured by titration with standardhydrochloric acid according to the…
- the free energy for a reaction can be related to the equilibriumconstant through the formula below.K = e (-ΔG° / RT)Therefore if Kc for a reaction is known, Go can be determined, or vice versa. Furthermore, ifyou have the value for Go at two different temperatures, you can calculate H and S throughthe familiar equation for Gibbs energy below, since you have two unknowns but also twoequations.G = H – T SIn this lab you will be studying the solubility of borax (Na2B4O5(OH)4*8H2O), a slightly solublesodium salt, at two different temperatures. When solid borax is added to water, theequilibrium below is established.Na2B4O5(OH)4*8H2O (s) 2 Na+ (aq) + B4O5(OH)42- (aq) + 8 H2O(l)If you measure the concentrations for those substances that show up in the reaction quotient,then the Kc for the reaction at that temperature can be calculated. In this lab, theconcentration of borate ion (B4O5(OH)42-) in solution will be measured by titration with standardhydrochloric acid according to the…the free energy for a reaction can be related to the equilibriumconstant through the formula below.K = e (-ΔG° / RT)Therefore if Kc for a reaction is known, Go can be determined, or vice versa. Furthermore, ifyou have the value for Go at two different temperatures, you can calculate H and S throughthe familiar equation for Gibbs energy below, since you have two unknowns but also twoequations.G = H – T SIn this lab you will be studying the solubility of borax (Na2B4O5(OH)4*8H2O), a slightly solublesodium salt, at two different temperatures. When solid borax is added to water, theequilibrium below is established.Na2B4O5(OH)4*8H2O (s) 2 Na+ (aq) + B4O5(OH)42- (aq) + 8 H2O(l)If you measure the concentrations for those substances that show up in the reaction quotient,then the Kc for the reaction at that temperature can be calculated. In this lab, theconcentration of borate ion (B4O5(OH)42-) in solution will be measured by titration with standardhydrochloric acid according to the…the free energy for a reaction can be related to the equilibriumconstant through the formula below.K = e (-ΔG° / RT)Therefore if Kc for a reaction is known, Go can be determined, or vice versa. Furthermore, ifyou have the value for Go at two different temperatures, you can calculate H and S throughthe familiar equation for Gibbs energy below, since you have two unknowns but also twoequations.G = H – T SIn this lab you will be studying the solubility of borax (Na2B4O5(OH)4*8H2O), a slightly solublesodium salt, at two different temperatures. When solid borax is added to water, theequilibrium below is established.Na2B4O5(OH)4*8H2O (s) 2 Na+ (aq) + B4O5(OH)42- (aq) + 8 H2O(l)If you measure the concentrations for those substances that show up in the reaction quotient,then the Kc for the reaction at that temperature can be calculated. In this lab, theconcentration of borate ion (B4O5(OH)42-) in solution will be measured by titration with standardhydrochloric acid according to the…
- the free energy for a reaction can be related to the equilibriumconstant through the formula below.K = e (-ΔG° / RT)Therefore if Kc for a reaction is known, Go can be determined, or vice versa. Furthermore, ifyou have the value for Go at two different temperatures, you can calculate H and S throughthe familiar equation for Gibbs energy below, since you have two unknowns but also twoequations.G = H – T SIn this lab you will be studying the solubility of borax (Na2B4O5(OH)4*8H2O), a slightly solublesodium salt, at two different temperatures. When solid borax is added to water, theequilibrium below is established.Na2B4O5(OH)4*8H2O (s) 2 Na+ (aq) + B4O5(OH)42- (aq) + 8 H2O(l)If you measure the concentrations for those substances that show up in the reaction quotient,then the Kc for the reaction at that temperature can be calculated. In this lab, theconcentration of borate ion (B4O5(OH)42-) in solution will be measured by titration with standardhydrochloric acid according to the…the free energy for a reaction can be related to the equilibriumconstant through the formula below.K = e (-ΔG° / RT)Therefore if Kc for a reaction is known, Go can be determined, or vice versa. Furthermore, ifyou have the value for Go at two different temperatures, you can calculate H and S throughthe familiar equation for Gibbs energy below, since you have two unknowns but also twoequations.G = H – T SIn this lab you will be studying the solubility of borax (Na2B4O5(OH)4*8H2O), a slightly solublesodium salt, at two different temperatures. When solid borax is added to water, theequilibrium below is established.Na2B4O5(OH)4*8H2O (s) 2 Na+ (aq) + B4O5(OH)42- (aq) + 8 H2O(l)If you measure the concentrations for those substances that show up in the reaction quotient,then the Kc for the reaction at that temperature can be calculated. In this lab, theconcentration of borate ion (B4O5(OH)42-) in solution will be measured by titration with standardhydrochloric acid according to the…Calculating Thermodynamic Values from an Equilibrium ConstantBackgroundAs discussed in lecture, the free energy for a reaction can be related to the equilibriumconstant through the formula below.K = e (-ΔG° / RT)Therefore if Kc for a reaction is known, Go can be determined, or vice versa. Furthermore, ifyou have the value for Go at two different temperatures, you can calculate H and S throughthe familiar equation for Gibbs energy below, since you have two unknowns but also twoequations.G = H – T SIn this lab you will be studying the solubility of borax (Na2B4O5(OH)4*8H2O), a slightly solublesodium salt, at two different temperatures. When solid borax is added to water, theequilibrium below is established.Na2B4O5(OH)4*8H2O (s) 2 Na+ (aq) + B4O5(OH)42- (aq) + 8 H2O(l)If you measure the concentrations for those substances that show up in the reaction quotient,then the Kc for the reaction at that temperature can be calculated. In this lab, theconcentration of borate ion…
- COMPOUND ∆Hf (kJ/mole) S° (J/K mole) MgCO3 –1111 65.86 BaCO3 –1213 112.1 MgO –601.6 27.0 BaO –548 72.1 CO2 –393.5 213.8 Calculate ∆G° for the reaction of MCO3 ---> MO + CO2 at 25°C for the alkaline earth compounds Mg and BaI. MULTIPLE CHOICE Read and answer each question carefully. Write your answer in the space provided. II. Calculate the AG for the following reactions and predict whether the reaction is spontaneous or not. Use the Gibbs free energy formation provided and this AGrxn= EAGproducts - LAGreactants to be more specific.Using the Debye-Huckel limiting law equation, Please compute ( not qualitatively) the hydroxide ion activity in a 0.050M LiOH solution? Qualitatively , it is 0.05M, I would like to see the calculations please





