Q: Balance the following chemical equation and determine the mass in grams of FeO(s) needed to produce…
A: The reaction given is, => FeO (s) + C (s) → Fe (s) + CO2 (g) Given: Mass of Fe to be produced =…
Q: Chemically and physically describe the combustion of methane CH4 (g) + 2 O2 (g) → CO2 (g) + 2 H20…
A: Combustion of Methane : Combustion of Methane is a chemical reaction in which methane gas burns,…
Q: Give an example of Interhalogen Compounds?
A: Compounds containing two distinct forms of halogens (fluorine, chloride, bromide, iodine or…
Q: The reaction of lithium with sulfur is similar to that of sodium with sulfur. Write the balanced…
A: Write the balanced chemical equation for the reaction of lithium with sulfur. Include phase symbols.
Q: In our bodies, sugar is broken down with oxygen to produce water and carbon dioxide. Write a…
A: Given Reactant = Glucose and Oxygen Complete oxidation reaction = ?
Q: Balance the following chemical equation (if necessary): H:O(1) → H:(g) + O:(g)
A: The given reaction is : H2O (l) → H2 (g) + O2 (g) This reaction is unbalanced. The Oxygen atoms are…
Q: Ariana plans to react a sample of magnesium carbonate (MgCO3) with hydrochloric acid (HCl) to…
A: Ariana plans to react a sample of magnesium carbonate (MgCO3) with hydrochloric acid (HCl) to…
Q: Balance the following equation and determine how many moles of aluminum oxide can be produced from…
A:
Q: balance this chemical reaction __________NaHCO3 → ________Na2CO3 +__________ H2O +…
A: Given chemical reaction is, __________NaHCO3 → ________Na2CO3 +__________ H2O + ______________CO2
Q: What mass of silver oxide, Ag2O, is required to produce 25.0 g of silver sulfadiazine, AgC10H9N4SO2,…
A: Mass of silver sulfadiazine = 25 g Molar mass of silver sulfadiazine = 357.14 g/mol Moles of silver…
Q: Write a balanced chemical equation for the incomplete combustion of methane to produce CO(g) as the…
A: The incomplete combustion of methane in the presence of limited amount of oxygen gas leads to the…
Q: Acetylene gas, C2H21g2, is used in welding. (a) Write a balanced equation for the combustion of…
A: The unbalanced chemical equation for the reaction of acetylene with air is shown in equation (1).
Q: Liquid water reacts with solid sodium to produce aqueous sodium hydroxide (NaOH) and hydrogen gas.…
A:
Q: What mass of methane is required to produce 1.130 kg of carbon dioxide during combustion? (Assume…
A:
Q: Write a chemical equation for the combustionof each of the following substances. If a…
A: Combustion, also known as combustion, is a high-temperature exothermic redox chemical reaction that…
Q: Write a balanced chemical equation for the hypothetical reaction of liquid dichloromethane, CH2CI2,…
A: Given that : We have to write a balanced chemical equation of liquid dichloromethane, CH2Cl2, with…
Q: The combustion of solid glucose (CH12O6) CSH12 O6 (s) 1
A: Combustion :- Burning a substance in excess of air or oxygen gas is called as combustion . In the…
Q: How many grams of oxygen are produced when 582 grams of water decomposes? 2H2O (l) --> 2H2 (g) + O2…
A:
Q: Write a balanced equation for the reaction of ethane (C 2H 6) with O 2 to form CO 2 and H 2O
A: A balanced chemical equation is defined as the equation where equal numbers of total individual…
Q: The oxidation state of chlorine in CIO is O A) -5. O B) +5. O C) +7. D) 0.
A: Oxidation state is the total number of electrons that an atom either gains or loses in order to form…
Q: Is the following chemical equation balanced? Does it follow the Law of Conservation of Mass? 2Fe…
A: In a balanced chemical reaction all the atoms of each element is same in reactants side with that of…
Q: Write chemical equations for each of the following chemical and physical processes: (a) Reaction of…
A:
Q: What is the balanced oxidation and reduction equations for the reactions taking place in a Fe and Cu…
A: Since iron have more negative value of reduction potential than the reduction potential of copper so…
Q: What element Is oxidized in the followlng chemical reaction? NIO2 + Cd + 2H20 NI(OH)2+ Cd(OH)2
A:
Q: what is a balanced equation for the reaction of nonane,C9H20 , with oxygen O2?
A: The reaction of nonane with oxygen is a combustion reaction. In combustion reaction, a hydrocarbon…
Q: Write the chemical equation of the thermite reaction, in which iron(III) oxide is reduced by…
A:
Q: The reaction of pentane, C5H12, with oxygen, O2, gives carbon dioxide and water. (a) Write a…
A: Combustion of pentane is occurring.
Q: Which chemical species gets oxidized and which gets reduced in the overall chemical equation: 2Zn(s)…
A: You have given the chemical reaction- 2Zn(s) + O2(g) => 2 ZnO(s)
Q: Given the following balanced chemical equation, calculate the theoretical yield of carbon dioxide…
A:
Q: Write a balanced equation for the combustion of isooctane, C8H18, a component of gasoline.
A: The balanced equation for the combustion of isooctane has to be given.
Q: H2S+NAOH NazS+H2O
A:
Q: Write the chemical formula for manganese(IV) chromate
A: Given, a chemical name of compound manganese(IV) chromate.we are asked to write the chemical formlua…
Q: Balance the following equation, and then calculate how many moles of oxygen are needed to produce…
A: Al (s) + O2 (g) -----------> Al2O3 (s) Balanced reaction :- 4 Al (s) + 3 O2 (g) --------> 2…
Q: What hydrocarbon is needed to complete and balance the following reaction? spark 5 O2 3 CO2 4 H20 A…
A: Since you have posted 2 completely unrelated questions, we are entitled to answer the first only.
Q: basic solution, molecular chlorine, Cl 2 , reacts with hydroxide ions, OH −
A: Cl2+OH- ⇌ ClO3 - + Cl- +H20 Self oxidation -reduction reaction
Q: Which of the following chemical formula is for a cyclic compound that contains two double bonds? O…
A: Degree of unsaturation :- It indicates how many double bonds or triple bonds or rings are present in…
Q: Give the chemical formula for (a) carbonic acid, (b) sodiumcyanide, (c) potassium hydrogen…
A: The composition of a chemical compound can be presented in a very precise was with the use of…
Q: Write chemical equations for each of the following chemical and physical processes: - Combustion of…
A: (i) Combustion of CH3OH 2CH3OH(l) + 3O2 (g) --->2CO2 (g) + 4 H2O (l) (ii) Thermal decomposition…
Q: Write a balanced chemical equation for the acid-base reaction of oxalic acid, H2C2O4, with sodium…
A:
Q: belo 1.) NaH 2.) Br HO,
A: Stereochemistry studies the spatial arrangement of atoms. SN2 reaction is the nucleophilic…
Q: What mass of methane is required to produce 1.130 kg of carbon dioxide during combustion? (Assume…
A: The balanced equation will be:- CH4(g) + 2O2(g) → CO2(g) + 2H2O(g) Molar mass of CO2 = 44.01g/mol…
Q: Write and balance the Chemical Equation: HC2H3O2 + CaCO3→ calcium acetate + H2O + carbon dioxide
A:
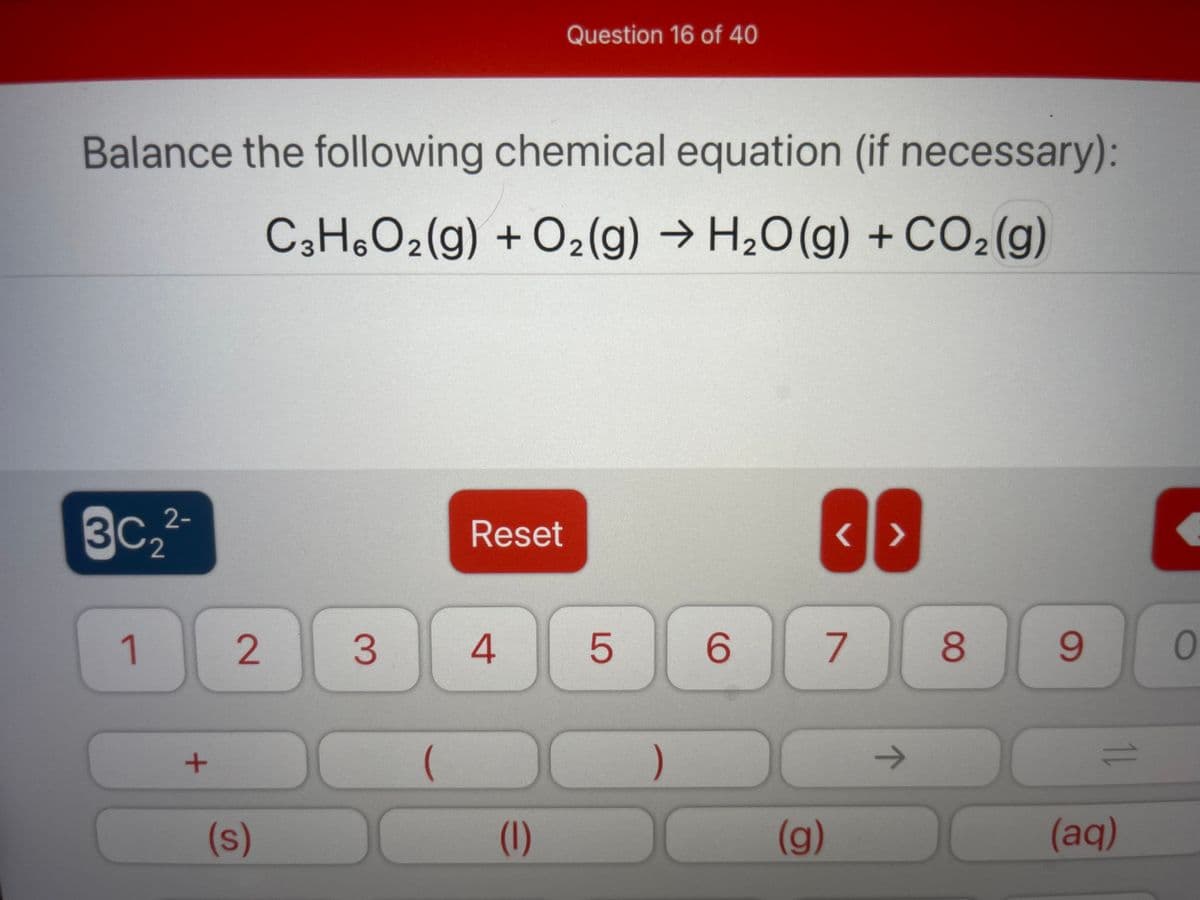
Q: Balance the following equation by filling the gaps with appropriate numbers. CSHB 02 H2O
A: In balanced reaction number of each atom on reactant side is equal to number of each atom on product…
Q: If the heat of combustion for a specific compound is −1260.0 kJ/mol−1260.0 kJ/mol and its molar mass…
A:
Q: Give the chemical formula for the compound Aluminum fluoride Iron(III) sulfıde Zinc nitrate
A: "Since there are multiple sub-parts in this question and it is not mentioned that which one has to…
Q: How many grams of Al2O3 can be produced from 9.5 g Al?
A: Given, Mass of Al = 9.5g. The reaction of Al2O3 formation from Al is given as follows:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- How much iron oxide (Fe2O3) is needed to make a 500 mg fuel rich mixture of aluminum (Al) and iron oxide that has an equivalence ration of 1.3? What is the actual F/O ratio for this mixture? Please provide only typed answer solution no handwritten solution needed allowedMr. Naresh works in a multinational company. He is stressed due to his hectic schedule. Mr. Amit, his friend, comes to know that he has started taking sleeping pills without consulting the doctor. Mr. Amit requests ‘ Naresh to stop this practice and takes him to a Yoga centre. With regular Yoga sessions, Mr. Naresh is now a happy and relaxed man. ‘ After reading the above passage, answer the following questions: (i) Write the values shown by Mr. Amit. (ii) Which class of drugs is used in sleeping pills? (iii) Why is it not advisable to take sleeping pills without consultation with the doctor?QUESTION 4 Calculate ΔG°rxn for the reaction below at 25.0°C. 2H2O2 (ℓ) → 2H2O (ℓ) + O2 (g) Report your answer in kJ/mol to two decimal places. ΔG°f (kJ/mol) H2O2 (ℓ) -120.35 H2O (ℓ) -237.13
- Calculate the following values. Enter your answers in scientific notation. (a) What is the Kb of the benzoate ion, C6H5COO−? Kb = × 10 (b) What is the Ka of the 2−hydroxyethylammonium ion, HOCH2CH2NH3+? ( pKb of HOCH2CH2NH2 = 4.49 ) Ka = × 10Give answer to all parts? Initial Temp of HCl: 22.96 C Final Temp of HCl: 28.30 C Moles of HCl: .05 moles Moles of NaOH: .05 moles Cs water: 4.184 J/g C Accepted delta Hrxn: -55.8 kJ/mol 1. what is the qwater. pls include formula. 2. calculate delta Hrxnn using the moles, temp and cswater 3. calculate delta Hrxn % errorIf the empirical formula of a compound is found to be CH2, which of the following value is a plausible molecular mass for the compound? The answer is 70.15. How did you get that?
- 13. A sample of 15.00 g of a hydrated sodium sulfate salt contains 7.05 g of water. The value of the "x" in the hydrated salt is: NaSO4 . xH2O Answers: a)7 b)10 c)1 d)3 e)5is the A:B ratio just a place holder for the unknown compound can you use other variables in place of "A and B" also would H2O work as another formula instead of Na2O3. Syngas is a mixture of carbon monoxide, carbon dioxide, and water vapor that is used for making fertilizer, methanol, and other products. It is produced by heating fossil fuels under low-oxygen conditions to promote incomplete combustion. Write an equation for the complete combustion of the gasoline component cyclopentane, C5H10 (l) and the incomplete combustion of cyclopentane with 20% of the carbon appearing as CO(g).
- So for the question below, I have the answer worked out from class but I don't understand certain steps. For example, why does KiCi+KaCa / KaCa = 4.50 become KiCi/KaCa + KaCa/KaCa = 4.50 ? Where does the second KaCa come from? Next, you cancel values and get 1 + Ka,i(Ci/Ca) = 4.50 -- in this step where does the one come from? Glutathione is an important antioxidant found in plants and animals. You are measuring glutathione in blood, but the analysis is complicated by many potential interferents including ascorbic acid. In one of your studies, you analyze a solution of 10.0 ppb glutathione and 1.5 ppb ascorbic acid, the signal was 4.50 times greater than the signal you measured for sample containing only 10.0 ppb glutathione. What is the selectivity coefficient for this analysis?Give (in order) the correct coefficients to balance the following equation:NH3+ O2→ NO + H2O 4, 4, 4, 6 2, 3, 2, 3 1, 1, 1, 1.5 4, 5, 4, 6 2, 2, 1, 3Calculate deltaH1 Please provide 3 sig figs, units are KJ/mol, please only give number value in answer. Mg(s) + 1/2O2(g) --> MgO(s) deltaH1 = ? Mg(s) + 2H+(aq) --> Mg2+(aq) + H2(g) deltaH2 = +10.1 kJ/mol MgO(s) + 2H+(aq) --> Mg2+(aq) + H2O(l) deltaH3 = -5.00 kJ/mol 1/2O2(g) + H2(g) --> H2O(l) deltaH4 = -286.0 KJ/mol

