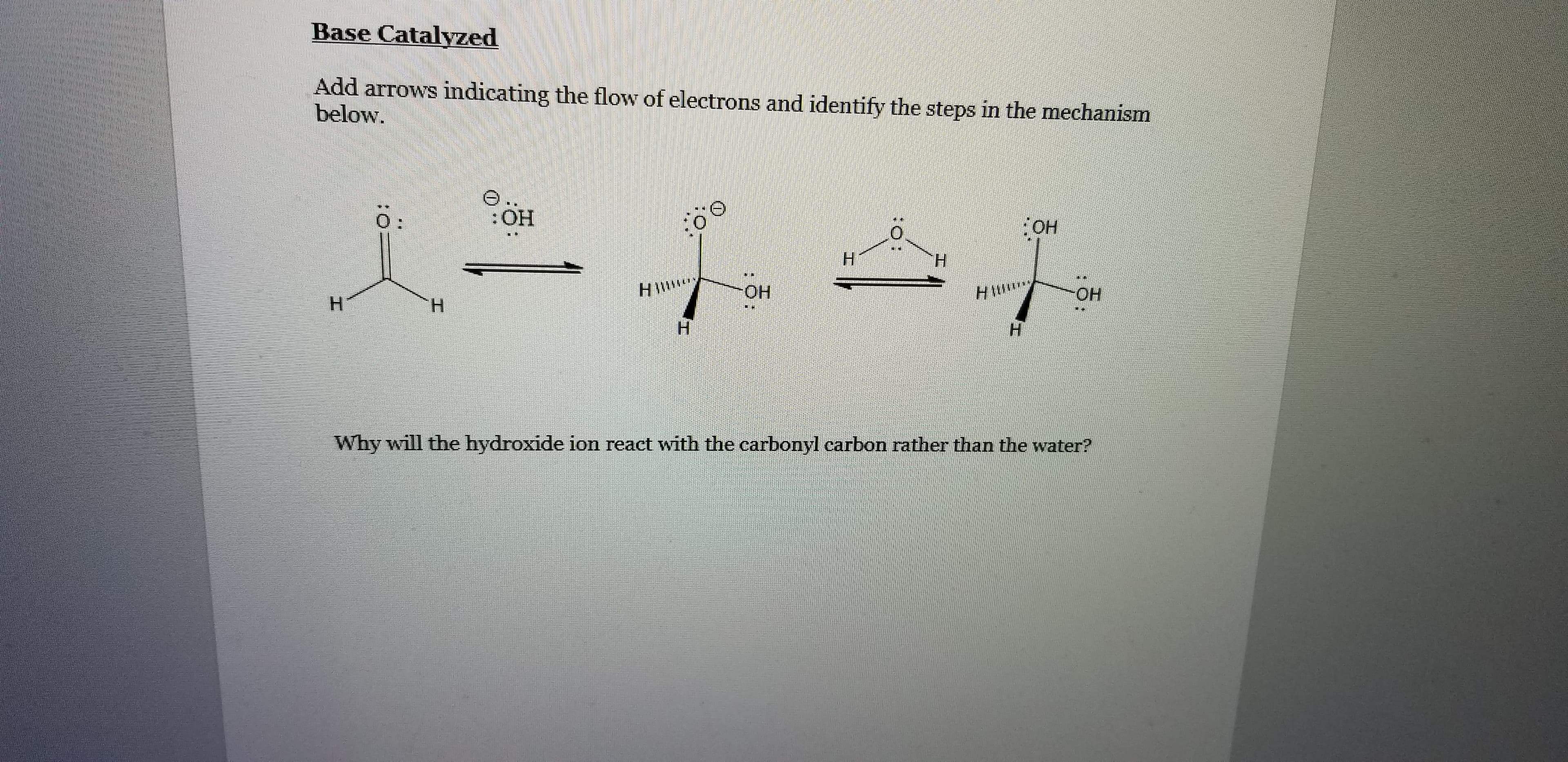
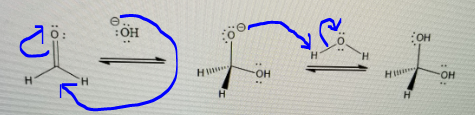
Base Catalyzed Add arrows indicating the flow of electrons and identify the steps in the mechanism below. 0: :HO: H. HIW ОН HIM Он Н Н H. н Why will the hydroxide ion react with the carbonyl carbon rather than the water?
Q: b. Hydrocyanic acid and methylamine (CH;NH;) c. Sulfurous acid (first proton only) and ethylamine…
A: b. When HCN and MeNH2 reacts together then salt MeNH3^+CN^- is formed. Molecular equation : HCN +…
Q: 10. Provide the product resulting from the following reaction. PB 3 HO, Br PB(2 11. Rank the…
A:
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A:
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A: Introduction: According to Bronsted-Lowry theory, acid is a substance that gives an H+ ion or a…
Q: 1. Identify the acids and bases in the following reactions: (al CH3OH + H* - CH3OH;+ (b) CH3OH + NH3…
A: Since here multiple question have been posted so I am going to solve question 2 ,3 and 4. if you…
Q: 15. provide the missing reagents for the following reaction
A: The Grignard reaction: Alkyl, vinyl, or aryl-magnesium halides ( RMgBr) is called as Grignard…
Q: Which carboxylic compound of each pair would you expect to be stronger acid? Indicate (a) or (b) on…
A: In case 1) Me3N+ is an electron withdrawing group. It with draws electron from O-H bond through…
Q: Pls help ASAP. Pls do all 4 i beg
A:
Q: 1) 2) 이 거 + m ○ u Mo + 'H f Ome MeoNa Meol [H+] HO MeONa
A:
Q: In the following reaction, how will the hydroxide ion most likely react with propanoic acid? OH™ OH…
A: According to the Arrhenius theory, the substance that gives hydrogen ion H+ in the aqueous solution…
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: Explain why the a protons of an ester are less acidic than the a protons of a ketone by ~5 pkg…
A: The stronger acids have lesser value of pka and weaker acids have larger value of pka.
Q: following reactions is representation för the heutralization of acetic acid by sodium hydroxide? (c)…
A: Introduction: We have to tell which reaction is neutralization reaction.
Q: 1. The following reactivity order has been found for the basic hydrolysis of p-substituted methyl…
A: Order of reactivity of various para substituents.
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A: Bronsted acid- base theory:- According to this theory, an acid is a species which can transfer a…
Q: 4. reactivity towards hydrolysis HO. NH2
A: In this question, we will see the reactivity order ( for least Reactive 1 and for most reactivity…
Q: 16. 8 Ci D Nombre: 17. Nombre: HO 18. Nombre: Br 19. -4-ethyl-2,3-dimethyl-5-iodobenzoic acid www…
A:
Q: e) For the following carbonyl compounds, please rank their relative acidity (i.e. 1 = most acidic, 6…
A: Solution is given below
Q: Predict the major product(s) when hexanoyl chloride is reacted with: a. acetic acid, with pyridine…
A:
Q: 8. In the reaction below, why does the amine nitrogen (#1 in red) undergo protonation with H2SO4…
A:
Q: Which of the following will give a negative result with Br2/CCI4? O a) O b) O d) H. Which of the…
A: Br2 in the solvent of CCl4 give addition reaction on alkene and alkyne ( called trans addition…
Q: 1. KMNO4, HO¯, heat 2. H3O*
A:
Q: 2. Draw the structure of the ESTER A and identify the missing reagents that are required to complete…
A: We have to draw the following missing reagents that are
Q: The bicyclic ketone shown below is first treaterd with a base, then ethyliodide is added to form the…
A: We have given the different bases conditions but the best suitable condition for transformation is…
Q: The synthesis of an azo dye is shown below. Please answer the following questions. NH2 N2 1) NażCO3…
A: In this question, we will see which nitrogen is most basic Nitrogen in Compound - 3 You can see…
Q: Which reaction would give a carboxylic acid? a) c) en H 1) LIAIH 2) H₂O* H₂ Ni b) d) MgBr 1) CH, 0…
A:
Q: 24) What is the product of the following reaction? C6H5CO₂Et + CH,CH,CO Et A) CH,CCH₂CH₂COET 00 B)…
A: Conjugate base - When an acid dissociates it gives a proton and an anion, the resulting anion is…
Q: Predict the product of the following reaction sequence. Me 1. МеОН, Н* ? Ph 2. PCC 3. CH3CH,CH,Li Ме…
A: Organic reaction.
Q: Answer questions a-c about the Bronsted acid-base reaction below using the identifying letters A-D…
A: According to Bronsted Lawry's concept of acid and bases. The acids are the substance that donates a…
Q: c) Which of the following phenolic compounds has the lowest pKa value? Explain. co.o HO, но ÓH A В
A:
Q: Complete this reaction of a carboxylic acid with a strong base. reaction: C,H,COOH + NAOH C,H,COO…
A: We have to complete the given reaction
Q: Which among the group has the most alkaline reaction? Aniline Kb = 3.8 x 10-10 Methylamine Kb = 4.4…
A: Inorganic chemistry.
Q: 1. Which carboxylic acid(s) would decarboxylate when heated to 100-150 °C? OH Ha II a. I b.I с. П d.…
A: Decarboxylation of the carboxylic acid: The carboxylic acid that contains a carbonyl group at the…
Q: Following is an outline of a synthesis of the bronchodilator carbuterol, a beta-2 adrenergic blocker…
A: Given is an outline of a synthesis of the bronchodilator carbuterol, a beta-2 adrenergic blocker…
Q: 1. Which nitrogen-containing compound of each pair would you expect to be stronger base? Indicate…
A:
Q: Rank the following carboxylic acid derivatives according to their increasing pKa values. HO. Но. HO.…
A: Acidic strength is Inversely proportional to Pka value
Q: Alkyl Halides (X = CI, Br or I): Assume AICI, is present if needed %3D 2 of 2 D E H un acid catalyst…
A: In this question, we want to enter a code letter or number of each steps for this given…
Q: Which letter is near the most acidic protons in the molecule below. 0000 В с D А H3C UI A of o CH3 D
A:
Q: Feagents are strong bases. Because of this, they cannot be prepared from compounds that contain…
A: 1. Ans : 3 ( Acidic hydrogens are present in the staring material. Therefore Grignard generation is…
Q: Explain the trends in the acidity of phenol and the monofluoro derivatives of phenol. OH он он он F…
A: Higher the pKa lower is the acidity. If the conjugate base is stabilized, then it is more acidic.
Q: Which of the following sequences ranks the structures below in order of increasing acidity? OH Дон 1…
A: The stability of the heterocyclic compounds such a pyrrole and pyridine can be directly correlated…
Q: کی رنوسیل Ph 22 C-O ester Ph OH + CI CI CI RO RO benzyl alcohol 23 24a) 24b muidelines 1 and 2 may…
A: Functional group is area substituent in a molecule which tells characteristic of compound.
Q: Please help with this question question 9
A:
Q: 48. Neutral iron (III) chloride is uscd to distinguish between phenols and non-phenolic aromatic…
A: Given 48 First Statement : Neutral Iron(III) chloride is used to distinguish between phenols and…
Q: 3. Complete the reactions below: CH OH thium aluminium hydride PCC (excess) Tollen's Reagent A1 > A2…
A: PCC is mild oxidising agent, tollen reagent oxidised aldehyde group and lithium aluminium hydride is…
Q: help
A: 2-propanol is a weak base. so, it tendency to react with strong acid .but we have a weak acid named…
Q: Which set of conditions will accomplish the transformation shown below? HO HO. O NAOH in water…
A: The details solution for this reaction is provided below in attach image.
Q: Chemistry IDENTIFY PRODUCT A TO C Br BugSnH, AIBN A Toluene, 80 °C Ph-
A:
Q: Write an equation for the proton transfer reaction that occurs when the following acid reacts with…
A:
Please find below the reaction mechanism.
Step by step
Solved in 2 steps with 2 images

- Electrostatic potential maps of anisole and thioanisole are shown. Which do you think is the stronger acid, p-methoxybenzoic acid or p-(methylthio)benzoic acid? Explain.Show how the synthetic scheme developed in Problem 23.67 can be modified to synthesize this triiodobenzoic acid X-ray contrast agent.Propose a detailed mechanism for the reaction below (in the attached picture), showing the structure of thestable intermediate and using curved arrows to indicate electron flow in each step
- To prepare butanoic acid from 1-propanol, which sequence of reagent(s) is/are best employed? a) (1) NaCN; (2) H2O; (3) [H+] b) LiAlH4 c) K2Cr2O7 in acid d) (1) PBr3; (2) NaCN; (3) H2O; (4) [H+]based on this video: on Hinsberg Test Tests for Amines - MeitY OLabs (8.5min) https://www.youtube.com/watch?v=j5jgMUWri8U Write the reaction (two-step, in skeletal) of each test amine when tested in the Hinsberg Test.How to approach and solve the problem, thank u
- 10.14 Complete the following reactions: Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be. Encode (not hand-written)!Give typed explanation 27. Arrange the following molecules in order of increasing acidity CH3CO₂H, H₂O, CH₂=CH₂, CH3CH₂OHChoose the set of letters corresponding to the BEST answer. -gives two moles of QRS upon hydrolysis -the most reactive towards nucleophilic acyl substitution -can be formed from the alcoholysis of TUV -has the most acidic proton
- The alkaline hydrolysis of the following compound yields: CH3CON(CH2CH3)2synthesize each from benzene and sketch the following: H-NMR; C-NMR; MS, and IR for the compounds: 1. ortho form of aminophenol 2. meta form of salicylic acidAlcohols can undergo a lot of different reaction mechanims. If the alcohol group (OH) is attaached to an aromatic core, how will the chemistry change as compared to a typical alkyl alcohol? A) The OH group will become more polarised and more nucleophilic. B) The OH group will become more susceptible to oxidation C) The OH group will become more polarised and therefore basic D) The OH group will become more polarised and therefore acidic.



