Based on Table 31.1 above, what is the total binding energy of the nucleus of lodine-131. Enter the numerical value in units of MeV. Type your answer. What element is formed after decay? bromine (Br) tellurium (Te) selenium (Se) xenon (Xe) astatine (At) 9 lodine-131 is injected into patients for treatment of thyroid cancer. How many days after injection does it take for 69% of a dosage to decay away? Type your answer. CO
Based on Table 31.1 above, what is the total binding energy of the nucleus of lodine-131. Enter the numerical value in units of MeV. Type your answer. What element is formed after decay? bromine (Br) tellurium (Te) selenium (Se) xenon (Xe) astatine (At) 9 lodine-131 is injected into patients for treatment of thyroid cancer. How many days after injection does it take for 69% of a dosage to decay away? Type your answer. CO
Chapter7: Statistical Data Treatment And Evaluation
Section: Chapter Questions
Problem 7.17QAP
Related questions
Question
ANSWER ALL PARTS FOR A THUMBS UP
Transcribed Image Text:References
Group 1
Period
2 3
5 6 7 8 9
4
11
12
13
14
16
17
18
Не
4.
Li
10
Ne
Ве
3
11
12
13
14
Si
15
16
18
Ar
Na
Mg
Al
4
19
20
21
23
26
Cr Mn
27
28
29
30
Zn
31
32
34
33
Se
As
Ca
Sc
Co
Cu
35
36
Kr
Br
Fe
Ni
Ga
Ge
37
Rb
38
Sr
40
Źr
39
41
42
43
Tc
46
44
45
Rh
Pd
Ru
49
47
48
Cd
In
Ag
51
50
Sb
Sn
Nb
Мо
52
53
54
Te
I
Xe
55
56
57
La
72
Hf
73
Ta
6.
74
75
Re
76
Os
77
78
79
Cs
80
81
82
83
84
Po
85
At
86
Rn
Ва
Ir Pt AuHg 6 B
W
87
Fr
88
Ra
89
Ac
* | 104 105 | 106 107 108 109 110||111 112 113 114 115 116 117 118
Rf
7
Db
Sg
Bh
Hs
Mt
Ds
Rg | Cn | Nh
FI
Mc
Lv
Ts
Og
*
58
59
60
61
62
63
67 68
69
Tm
če|| Pr | Nd Pm Sm Eu Ğd Ťb by Ho Er
64
65
66
71
70
Yb
Lu
90
Th
91
Pa
92
93
Np
94
95
Am Cm
Pu
96
97
98
99
100 101
No
102 103
Md
Bk
Cf
Es
Fm
Lr
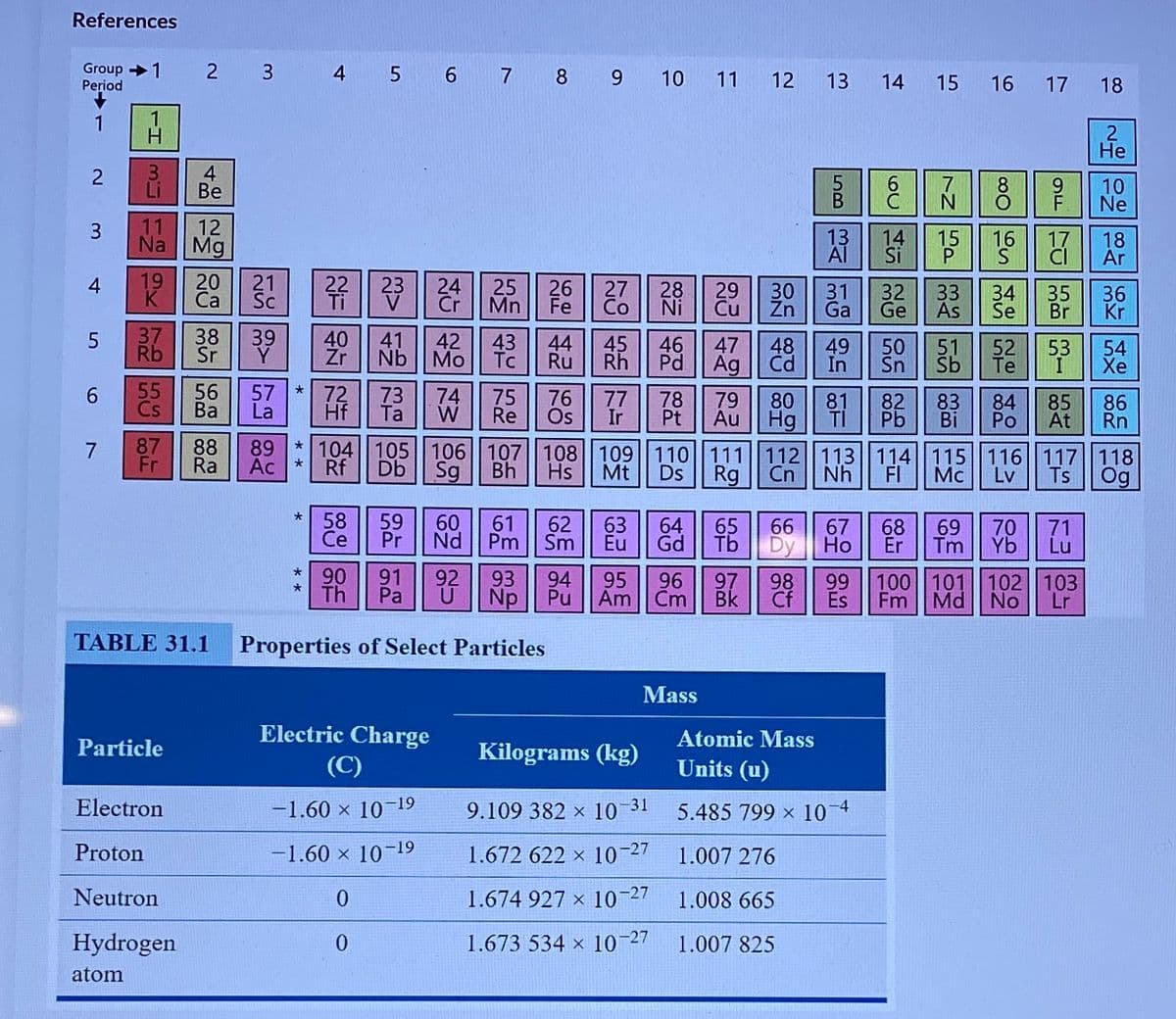
TABLE 31.1
Properties of Select Particles
Mass
Electric Charge
Atomic Mass
Particle
Kilograms (kg)
(C)
Units (u)
Electron
-1.60 × 10–19
9.109 382 × 10 31
5.485 799 x 10 4
Proton
-1.60 × 10-19
1.672 622 × 10-27
1.007 276
Neutron
1.674 927 x 10-27
1.008 665
Hydrogen
1.673 534 × 10-27
1.007 825
atom
15
10
2.
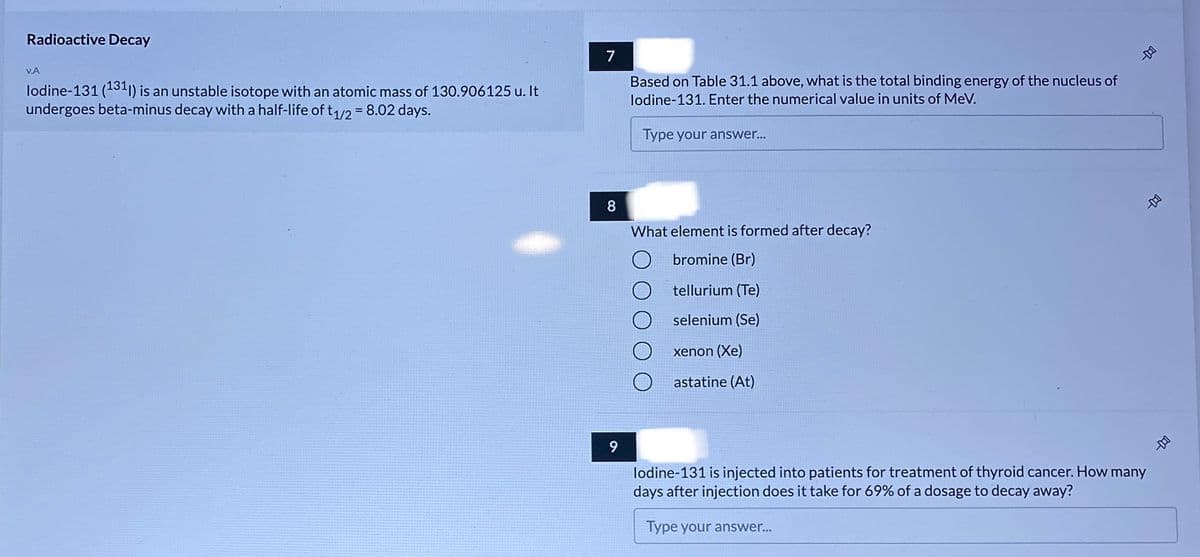
Transcribed Image Text:Radioactive Decay
7
VA
lodine-131 (131"1) is an unstable isotope with an atomic mass of 130.906125 u. It
undergoes beta-minus decay with a half-life of t1/2 =8.02 days.
Based on Table 31.1 above, what is the total binding energy of the nucleus of
lodine-131. Enter the numerical value in units of MeV.
Type your answer...
8
What element is formed after decay?
bromine (Br)
tellurium (Te)
selenium (Se)
xenon (Xe)
astatine (At)
9
lodine-131 is injected into patients for treatment of thyroid cancer. How many
days after injection does it take for 69% of a dosage to decay away?
Type your answer...
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning



Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning