Calculate for the binding energy in MeV per nucleon of 207 Ph if the actual mass of the atom is 206.975 amu. Express your answer in 2 decimal places. Do not write the units. 82 Mass of proton = 1.0078 amu; Mass of neutron = 1.0087 amu Speed of light = 3.00 x 108 m/s 1 amu = 1 g/mol = 1.66 x 10-27 kg 1 MeV = 1.60 x 10-13 J
Calculate for the binding energy in MeV per nucleon of 207 Ph if the actual mass of the atom is 206.975 amu. Express your answer in 2 decimal places. Do not write the units. 82 Mass of proton = 1.0078 amu; Mass of neutron = 1.0087 amu Speed of light = 3.00 x 108 m/s 1 amu = 1 g/mol = 1.66 x 10-27 kg 1 MeV = 1.60 x 10-13 J
Chemistry & Chemical Reactivity
10th Edition
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Chapter25: Nuclear Chemistry
Section: Chapter Questions
Problem 36PS
Related questions
Question
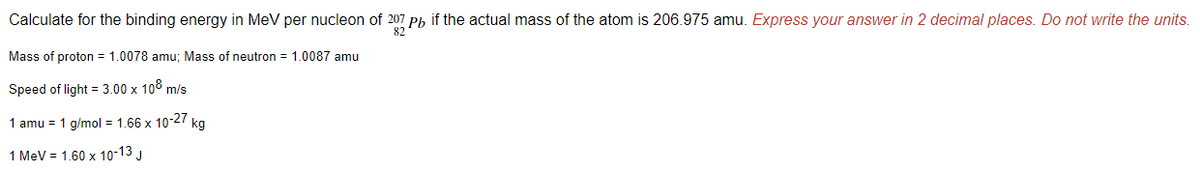
Transcribed Image Text:Calculate for the binding energy in MeV per nucleon of 207 Ph if the actual mass of the atom is 206.975 amu. Express your answer in 2 decimal places. Do not write the units.
82
Mass of proton = 1.0078 amu; Mass of neutron = 1.0087 amu
Speed of light = 3.00 x 108 m/s
1 amu = 1 g/mol = 1.66 x 10-27 kg
1 MeV = 1.60 x 10-13 J
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning