Based on the IR Spectra, need help filling out the following table. Thank you :) Wavenumber (cm-1) Band intensity Assignment e.g. 3286 e.g. Medium, broad e.g. N-H stretching
Based on the IR Spectra, need help filling out the following table. Thank you :) Wavenumber (cm-1) Band intensity Assignment e.g. 3286 e.g. Medium, broad e.g. N-H stretching
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter28: High-performance Liquid Chromatography
Section: Chapter Questions
Problem 28.5QAP
Related questions
Question
Based on the IR Spectra, need help filling out the following table. Thank you :)
| Wavenumber (cm-1) |
Band intensity |
Assignment |
| e.g. 3286 | e.g. Medium, broad | e.g. N-H stretching |
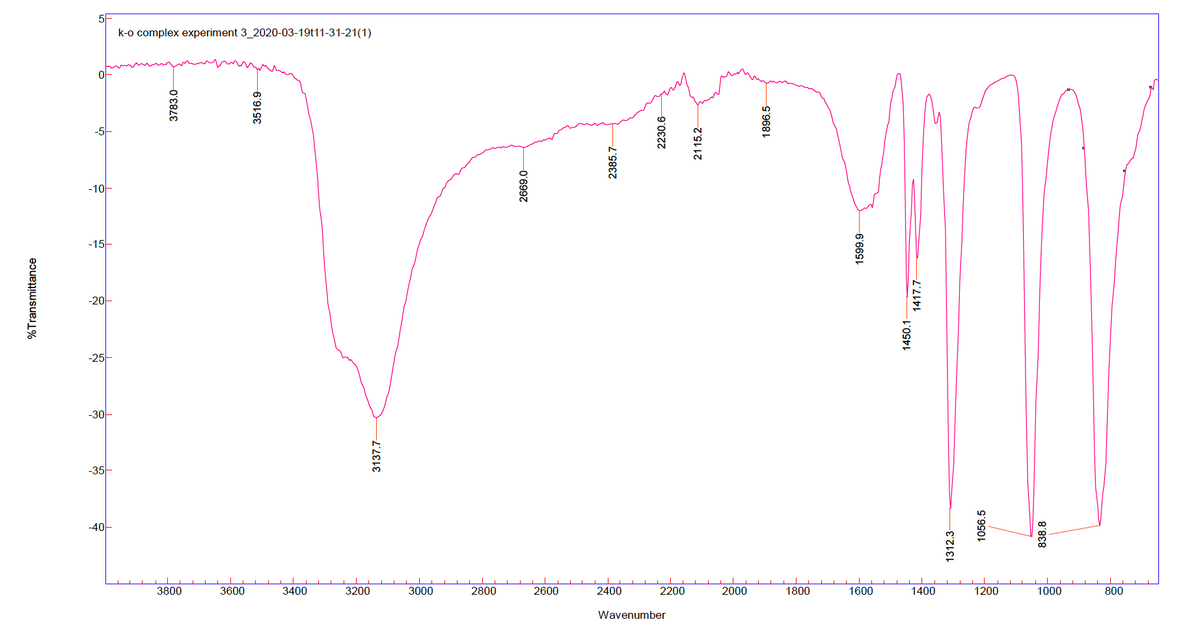
Transcribed Image Text:k-o complex experiment 3_2020-03-19t11-31-21(1)
OF
-5-
-10-
-15-
-20아
-25-
-30-
-35-
-4아
3800
3600
3400
3200
3000
2800
2600
2400
2200
2000
1800
1600
1400
1200
1000
800
Wavenumber
%Transmittance
3783.0-
3516.9-
3137.7-
2669.0-
2385.7-
2230.6-
2115.2-
1896.5–
1599.9–
1450.1–
1417.7-
1312.3-
1056.5
838.8
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
