Based on the Maxwel Boltzman Distribution Function, find the mean of the following quantities : a) W, Wx, Wx, Wx* b) Wy, W,, W,3, w, c) W, W,2, W 3, W, d) W, W², w 3.Wª 4.
Based on the Maxwel Boltzman Distribution Function, find the mean of the following quantities : a) W, Wx, Wx, Wx* b) Wy, W,, W,3, w, c) W, W,2, W 3, W, d) W, W², w 3.Wª 4.
Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter1: Gases And The Zeroth Law Of Thermodynamics
Section: Chapter Questions
Problem 1.76E
Related questions
Question
give me step by step answer
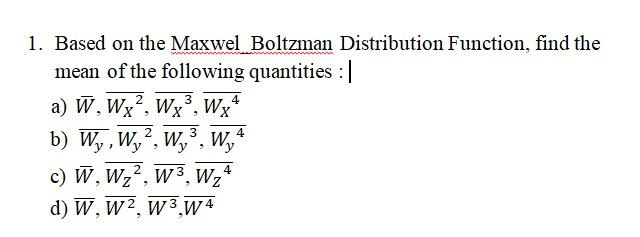
Transcribed Image Text:1. Based on the Maxwel Boltzman Distribution Function, find the
mean of the following quantities :|
a) W, Wx², Wx°, Wx*
b) W,, W,, W,, W,
c) W, Wz2, w 3, Wz°
d) W, W2, w 3 W4
4
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 4 images

Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,