Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter8: Addition Via Carbocation
Section: Chapter Questions
Problem 6CTQ
Related questions
Question
Please use the table
Which is the major and minor product for this molecule reacting with cl2

Transcribed Image Text:"For each halogen, reactivities with four types of alkane C-H bonds are normalized to the reactivity of th
2-151 O
(tert-Butyl bromidej
90%
:
Less (primary)
0.06%mi
10%
Expected (statistical) ratio
C-H bond reactivity
Experimental ratio
More (tertiary)
99.94%
In Summary Increased reactivity goes hand in hand with reduced selectivity in radied.
stitution reactions. Fluorine and chlorine, the more reactive halogens, discriminate ba
the various types of C-H bonds much less than does the less reactive bromine (Table 3.
Relative Reactivities of the Four Types of Alkane C-H Bonds
in Halogenations
Table 3-6
F•
e Cl.
(25°C, gas)
C-H bond
Br
(150°C, g
(25°C, gas)
CH3-H
RCH-H"
R,CH-H
R;C-H
0.5
1
0.004
0.002
1.2
1
1
1.4
4
80
primary C-H bond.
1700
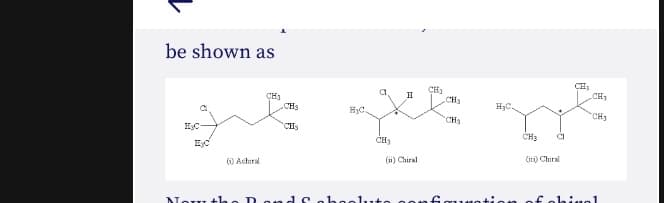
Transcribed Image Text:be shown as
CH1
.CHa
CH
CH
II
CH3
H3C
HyC.
CHs
HC-
EHD.
CH3
CH3
CI
0 Actiral
(w) Chiral
() Chital
Nou tha
D ond Saba
figuwotio
ohiuol
1.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning