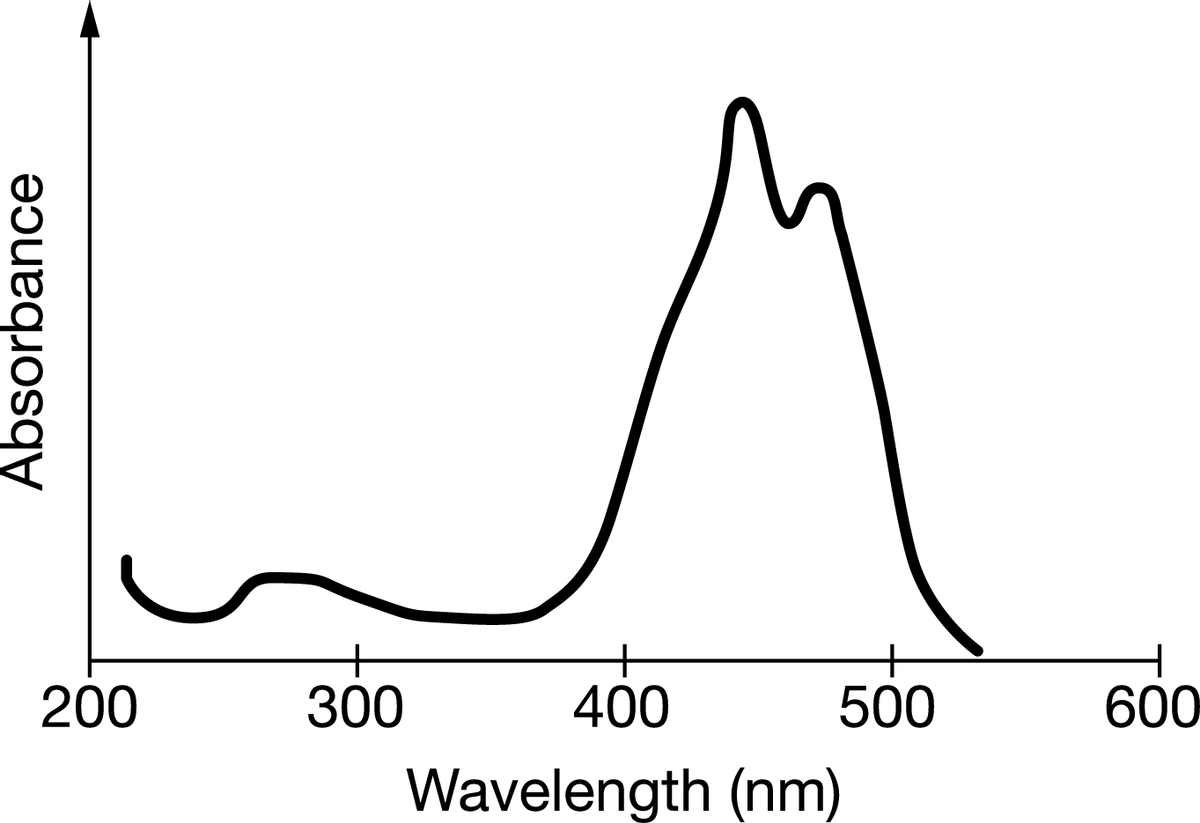
Beta-carotene is an organic compound with an orange color. The diagram above shows the ultraviolet spectrum of beta-carotene. Which of the following statements is true about the absorption bands in the spectrum?(see attached image) a.) The absorption band between 250250 and 320 nm320 nm is due to transitions in electronic energy levels, and the absorption band between 380 and 520 nm is due to transitions in molecular vibrational levels. b.) The absorption band between 250 and 320 nm is due to transitions in molecular vibrational levels, and the absorption band between 380 and 520 nm is due to transitions in molecular rotational levels. c.) The two main absorption bands are associated with transitions in electronic energy levels. The band in the region corresponding to shorter wavelengths shows a lower absorbance than the band in the region corresponding to longer wavelengths. d.) The two main absorption bands are associated with transitions in molecular vibrational levels. The band in the region corresponding to shorter wavelengths shows a lower absorbance than the band in the region corresponding to longer wavelengths.
Beta-carotene is an organic compound with an orange color. The diagram above shows the ultraviolet spectrum of beta-carotene. Which of the following statements is true about the absorption bands in the spectrum?(see attached image) a.) The absorption band between 250250 and 320 nm320 nm is due to transitions in electronic energy levels, and the absorption band between 380 and 520 nm is due to transitions in molecular vibrational levels. b.) The absorption band between 250 and 320 nm is due to transitions in molecular vibrational levels, and the absorption band between 380 and 520 nm is due to transitions in molecular rotational levels. c.) The two main absorption bands are associated with transitions in electronic energy levels. The band in the region corresponding to shorter wavelengths shows a lower absorbance than the band in the region corresponding to longer wavelengths. d.) The two main absorption bands are associated with transitions in molecular vibrational levels. The band in the region corresponding to shorter wavelengths shows a lower absorbance than the band in the region corresponding to longer wavelengths.
Principles of Instrumental Analysis
7th Edition
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Chapter34: Particle Size Determination
Section: Chapter Questions
Problem 34.9QAP
Related questions
Question
Beta-carotene is an organic compound with an orange color. The diagram above shows the ultraviolet spectrum of beta-carotene. Which of the following statements is true about the absorption bands in the spectrum?(see attached image)
a.) The absorption band between 250250 and 320 nm320 nm is due to transitions in electronic energy levels, and the absorption band between 380 and 520 nm is due to transitions in molecular vibrational levels.
b.) The absorption band between 250 and 320 nm is due to transitions in molecular vibrational levels, and the absorption band between 380 and 520 nm is due to transitions in molecular rotational levels.
c.) The two main absorption bands are associated with transitions in electronic energy levels. The band in the region corresponding to shorter wavelengths shows a lower absorbance than the band in the region corresponding to longer wavelengths.
d.) The two main absorption bands are associated with transitions in molecular vibrational levels. The band in the region corresponding to shorter wavelengths shows a lower absorbance than the band in the region corresponding to longer wavelengths.
Transcribed Image Text:200
300
400
500
600
Wavelength (nm)
Absorbance
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning