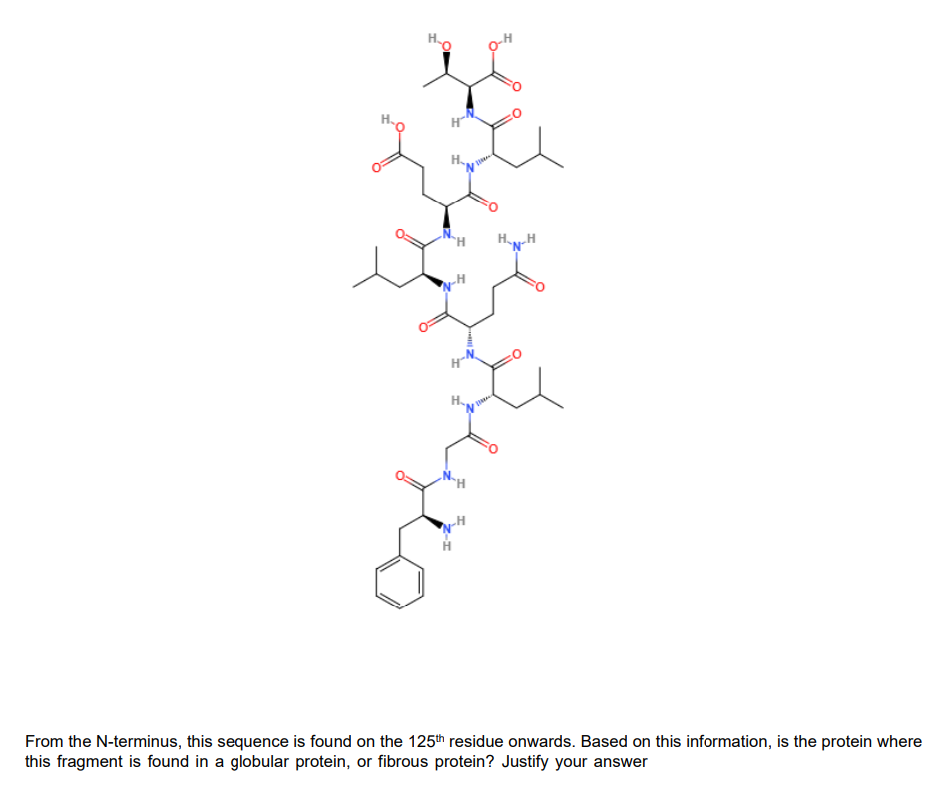
o-H H. Hy-H H. From the N-terminus, this sequence is found on the 125th residue onwards. Based on this information, is the protein where this fragment is found in a globular protein, or fibrous protein? Justify your answer
Q: V-B. Which of the following peptides would be more soluble at the indicated pH? 1. (Gly)20 or…
A: The twenty amino acids are classified into majorly the following classes: Polar Nonpolar Positive…
Q: titration curve. Match each numbered arrow on the curve (from (1) to (5)) with the appropriate amino…
A: Amino acids have two functional groups namely carboxyl and amino group. When the pH is less than pKa…
Q: Determining the amino acid sequence in a protein usually in- volves treating the protein with…
A: Treatment of a polypeptide with eleven amino acids with one reagent gives the following fragments :…
Q: Amino sequence:…
A: There are 37 amino acid residues are present in the polypeptide chain, which consists of several…
Q: Upload a drawing of Gly-Met-Asn-Glu-His Label the alpha carbons Label the R groups as…
A: Alpha carbon: It is the carbon connecting the amino group and carboxylic group in amino acid. Acidic…
Q: Protein: Hemoglobin Circle and underline each codon, amino acid sequence, make a mutation of the 3rd…
A: Here the not cleared the mutation type, are point mutations, frame shift mutation, addition mutation…
Q: proteins? a. High pressure drives protein conformational equilibria towards the physically larger…
A: Protein denaturation is the process in which the proper structure of the protein which is either in…
Q: How to calculate the molecular weight of the given protein sequence;…
A: The given peptide is composed of glutamine, alanine, cysteine, glutamine, glutamine and isoleucine.…
Q: Accoding to the figure below, which amino acid will be added next to the growing polypeptide chain?…
A: A codon is a trinucleotide sequence of DNA or RNA that translates to a certain amino acid. The…
Q: CH3CO-ETATKAELLAKYEATHK-CONH2 4. A polypeptide comprised of 17 amino acid res- idues with the…
A: Polypeptide: These are chains of amino acids with peptide bonds that connect them. Dehydration of…
Q: The number of distinct protein folds in limited. Why might this be so?
A: Protein folding is the physical process by which a polypeptide folds into its characteristic…
Q: Drag the correct amino acid into the corresponding box according to the roles they play. Reset Help…
A: Amino acids are the biological molecules which combines with each other to form proteins /…
Q: Give a brief description of what the term ‘native protein’ state refers to. Your answer should make…
A: Introduction: Proteins are biological polymers made up of amino acids connected covalently by…
Q: hi, I had a question about tertiary structure. I was wondering if the properties of the R group are…
A: Proteins are made of a long chain of amino acids. There are 20 amino acids in the protein. There are…
Q: H. State if/how Aureliano's mutation changes the amino acid sequence and describe the effect that…
A: Mutations are changes in the DNA sequences. It may be due to many reasons. There are many type of…
Q: 62. Which of the following cases DOES NOT illustrate protein denaturation? A. Breakdown of the…
A: Antibody: It is also known as Immunoglobulin. It is a protective protein that helps our body to…
Q: Formation of the tertiary structure of a protein - what are the main physical forces affecting the…
A: Biomolecules are organic molecules made up of mainly carbon and hydrogen but there are other…
Q: Loop regions play important roles in the secondary structure of protein. Define loop region and…
A: Protein is a naturally occurring polymer that is made up of amino acids linked by peptide bonds.…
Q: unfold the protein
A: Protein are consist of polymers of amino acids linked by amide/peptide bonds, which is called as…
Q: 5. amino acids classified to biological and -- -
A: Amino acids are the monomers that build up the proteins. They contain two functional groups namely…
Q: Functional conformation: Ultimately the functional conformation of a protein is determind by its…
A: The conformation of protein can be defined as an organization of the overall molecular shape in the…
Q: Protein Description A 35 kDa monomer Disulfide-linked homodimer comprised of 19 kDa monomers…
A: Protein is made by amino acids and in living organisms, 22 types of amino acids exist and each has a…
Q: 10) Denaturation of a protein
A: Proteins are complex structures that play a variety of important roles in the body. These are…
Q: protein folding briefly
A: Protein is a polymer of amino acids connected together via peptide bond. In a cell, proteins are…
Q: TERTIARY STRUCTURE (A) (B) (C) Fg Eet Galand Sen 20e Figure 6. Examples of the arrangement of…
A: Biomolecules are organic compounds found in living organisms. All living organism will have these…
Q: 63. Which of the following statements is CORRECT about protein denaturation? A. Complete protein…
A: The correct option is (b), loss of protein conformation causes loss of protein function.…
Q: Protein structure.Circle one of the three amino acid sequences that is most likely to form a stable…
A: The common secondary structure of the protein are alpha-helix and beta-sheets. The alpha-helix are…
Q: HN H3Ñ® Where on this amino acid does it attach to a primary sequence of a protein and where is the…
A: Given amino acid, Histidine is a basic amino acid. The structure of Histidine is presented as a…
Q: 9. Protein denaturation and renaturation. Probability of successful protein renaturation. Reversible…
A: Denaturation of protein occurs due the influence of denaturants on protein. Denaturants can be heat,…
Q: Draw serine
A: Those organic molecules which possess two functional groups carboxylate and amino group and a…
Q: Ile-Ala-His-Thr-Tyr-Gly-Pro-Phe-Glu-Ala-Ala-Met-Cys-Lys-Trp-Glu-Ala-Gln-Pro-Asp-Gly-Met-Glu-Cys-Ala-…
A: Most common secondary are the α-helix and the β-pleated sheet. Both the secondary structure is…
Q: The folding of some protein was monitored as a function of the time it takes for the protein to…
A: The proteins can be modified by different modifications and this modification can affect the…
Q: .Ala-Phe-Lys-Val-Val-Glu rom the above polypeptide, what amino acid/s go/goes inside the cell after…
A: We’ll answer the first question since the exact one wasn’t specified. Please submit a new question…
Q: geometry on both cand N. Trigonal plannes -- NHZ CH ーCーN eーN CH2 HN CH-C-O4 (d- carbon) HooC…
A: According to our guideline we can answer maximum 3 subparts of a question. So upload the questions…
Q: Does your protein 3GRS have a quaternary structure??? talk about the tertiary structure of 3GRS.…
A: All molecular models (atomic coordinate file) based on the X-ray crystallographic data of the…
Q: 1. The amino acid sequence for the protein lysozyme is given below. Estimate the isoelectric point…
A: Isoelectric point is the pH where the net charge on a protein is zero.
Q: How does the mutation change/affect the structure of the Hb heterotetramer (ie how is quaternary…
A: A mutation occurs when the sequence of DNA changes. Mutations may occur as a result of DNA copying…
Q: 30 A DNA sequence encoding a five-amino acid polypeptide is given below.…
A: Protein molecules are those molecules that are made up of chains amino acids in a specific order.…
Q: What would be shortest time a 250 amino acid protein would be able completely fold to the native…
A: Proteins are the building block of the body. They are macromolecules made up of amino acids as the…
Q: V-B. Which of the following peptides would be more soluble at the indicated pH? 1. (Gly)20 or…
A: There are two amino acids that are classified into 4 classes: 1. Nonpolar(aliphatic and aromatic) 2.…
Q: Protein denaturation disrupts the noncovalent interactions in a protein. Which level of protein…
A: By subjecting proteins or nucleic acids to an external stress or substance such as a strong acid or…
Q: Protein folding with PDI and Peptidyl-prolyl isomerase
A: Protein folding is a very sensitive process by which a polypeptide chain folds to become a…
Q: why this position in your protein is important and outline the effects the mutation will have on the…
A: Function of 3GRS: Glutathione acts as an important antioxidant in your body. That means it helps…
Q: Attach an amino acid monomer below. Name of the amino acid: Abbreviated name:
A: Amino acids at the building blocks of proteins.
Q: All standard amino acids have an asymmetric carbon except lysine
A: Amino acids are the building blocks of proteins. They are compounds containing carbon, nitrogen,…
Q: A common strategy in the regulation of protein function is to alter its structure. Describe two…
A: The proteins are the final product of gene expression. The deoxyribonucleic acid (DNA) is…
Q: In hinge region, what is the most prominent amino acid existing? A B C D E proline G F
A: The antibodies are released from the B-lymphocytes that are performing specific immunological roles…
Q: 2. Of the two sequences, which is more likely to have helical structure. Explain. a)…
A: The amino acids are the protein structure alphabet; they can be arranged to create an almost…
Q: Referring back to the quaternary level proteins, list and describe the modifications that can be…
A: The structure of protein includes sequence of amino acids in a polypeptide chain. The formation of…
Step by step
Solved in 3 steps

- Upload a drawing of Gly-Met-Asn-Glu-His Label the alpha carbons Label the R groups as hydrophobic or hydrophilic Label the acidic and basic R-groups Label the peptide bonds Label the N terminus and the C terminusQuestion:- Give a brief description of what the term ‘native protein’ state refers to. Your answer should make a specific reference to how the structural organisation of this state is accomplishedProtein Structure and Function A common strategy in the regulation of protein function is to alter its structure. Describe two specific strategies used by the cell to alter a protein’s structure, thereby altering its function.
- Leu-Trp-Phe-Met-Ala-Ile-Val- Draw the structure of the peptide at pH7.4. and Indicate the hydrogen bonds formed in the alpha helix.how many amino acids and omega bonds? How many amino acids appear to have undergone a post-translational modification? how many amino acids which would be classified as polar uncharged amino acids?Beta ( ? ) sheets are a type of secondary structure in proteins. A segment of a single chain in an antiparallel ? sheet has a length of 80.5 Å . How many residues are in this segment?
- Reposting - What would the tertiary structure of the dipeptide Asp-Ser be if it was made into a polypeptide chain? (Would it form a beta pleated sheet, an alpha helix, etc) Why would it do this? What properties of this polypeptide causes this? This sub part still needs to be solved - What would the tertiary structure of Pro-ala and Glycl-L-alanine be?Draw out this peptide using condensed or line-bond structures: His-Thy-Phe-Cys-Glu examine your drawing of peptide for each amino acid, indicate what type(s) of interactions it can contribute to protein tertiary structureAla-Arg-Val-His-Asp-Gln Given the polypeptide chain above Estimate the net charge of the polypeptide chain at physiological pH (7.4) and at pH 5.0 . How many peptide bonds are there? What kind of polypeptide is it?
- Hydropathy plot analysis of your protein of interest reveals a single, prominent hydrophobic peak. However, you later discover that this protein is soluble and not membrane associated. Explain how the hydropathy plot may have been misleading.Protein: HemoglobinCircle and underline each codon, amino acid sequence, make a mutation of the 3rd codon in the nucleotide sequence and circle the affected areas, show the amino acid area with the mutation.Lastly, describe the impact on the protein."MVHLTPEEKSAVTALWGKVNVDEVGGEALGRLLVVYPWTQRFFESFGDLSTPDAVMGNPKVKAHGKKVLGAFSDGLAHLDNLKGTFATLSELHCDKLHVDPENFR"Ala-Phe-Pro-Cys-Clu-Arg-Arg-Pro-His-Gly-Lys-Clu Calculate the net charge on the polypeptide at a. pH 1.5 b. pH 8 c. pH 13

