Q: 6.24 Consider this reaction: 2CH;OH(I) + 302(g) 4H,O(1) + 2CO2(g) AH = -1452.8 kJ/mol
A: The given reaction is : 2CH3OH (l) + 3O2 (g) ---------> 4H2O (l) + 2CO2 (g) , ΔH = -1452.8 KJ/mol…
Q: mass of empty calorimeter (g) 22.39 mass of hot water (g) 119.4 initial temp (Celsius) 64.43 mass…
A: When ice would be placed in hot water , hot water would release heat and ice would gain heat . At…
Q: Consider these reactions: Reaction 1: H2 (g) + Cl2 (g) → 2HC1(g) AH = –184.6 kJ Reaction 2: 20F2(g)…
A:
Q: Balanced Chemical Equation Reaction Type: At completion of reactions: Grams of calcium hydroxide…
A: The answer are as follows:
Q: A tank attached to a grill contains 9 kg of propane (C3H8) which burns to form CO2 and water: C3H8…
A: Given, Mass of propane = 9kg. Mass of propane is converted into moles as follows:
Q: 2 NO(g) + Cl2(g) 2 NOC((g)? [PNOCIP (PNO3]2 [Pc½] (PNO3]2 (PC12] (PNOCI? Кр Кр [PNOC1|2 [PCk] [PNO3?…
A: The given equilibrium reaction is as follows: In this reaction, nitrogen monoxide reacts with the…
Q: Questions
A: 2PbS + 3O2 -------> 2PbO + 2SO2 Divided by 2 PbS + 3/2O2 ------> PbO + SO2 Multiply by 5…
Q: Consider these reactions, where M represents a generic metal.…
A: The given equations are as follows. 2M(s)+6HCl(aq)⟶2MCl3(aq)+3H2(g)…
Q: Consider these reactions: Reaction 1: H2 (g) + Cl2 (g) → 2HC1(g) AH = -184.6 kJ Reaction 2: 20F2 (g)…
A: -> If ∆H =+ve => reaction is endothermic reaction. -> if ∆H = -ve => reaction is…
Q: What are the significant concepts about balancing chemical reactions?
A: 1. Balananced chemical equation obeys Law of conservation of mass. 2. Which states that mass is…
Q: Consider a reacting mixture consisting of 5 molecules of oxygen and 2 molecules of hydrogen reacting…
A: The question is based on stoichiometry of a reaction highlighting concept of limiting reagent. A…
Q: reactant? True or False?
A: Theoriyical yeild is determined by limiting reagent, rather than from excess reagent
Q: Which statement is true about the following reaction? N + He → "O 13.992 4.00 15 16.99 86 + H amu…
A:
Q: Question attached
A: Since carboxylic acid act as an Acid , so it will neutralized with base NaOH or KOH and reaction…
Q: Consider these reactions, where M represents a generic metal.…
A: Answer: We have to calculate enthalpy of formation of the given reaction.
Q: fill in the blanks to properly balance this equation __Agl+ __Na2S= __Ag2S+ __NaI
A: We are to balance the given equation __Agl+ __Na2S= __Ag2S+ __NaI We will use simple hit and trial…
Q: Consider these reactions: Reaction 1: H₂(g) + Cl₂(g) 2HCl(g) AH-184.6 kJ Reaction 2: 20F₂(g) O₂(g) +…
A:
Q: Consider the following reaction Ca(s) + 2HCI(aq) --> CaCl2(aq) + H2(g) How many moles of CaCl, can…
A: 2 moles
Q: Balance this equation: Al +O2->Al203 Al [ Choose ] 02 [ Choose ] A1203 [ Choosc Balance this…
A: In a balanced chemical equation, number of different atoms present in reactant and product side…
Q: Reaction 3: N₂(g) + 20₂(g) Reaction 2 can be described as an [Select] [Select] reaction 2NO₂(g) ΔΗ =…
A:
Q: 2Mg(S)+O2(g) →2MgO(s) a)-Formula and Description of the Substances b)-Balanced Chemical Equation…
A: a) Mg - Magnesium O2 - oxygen MgO - Magnesium oxide Mg is a solid, O2 is a gas and MgO is a solid…
Q: moles KMnO 4 to grams KMnO 4 18. Define these terms : a. limiting reactant b. excess reactant c.…
A: 1 mole = 6.022×1023 this number is known as Avogadro number To determine the mass of KMnO4 in 18.9…
Q: Which one of the equations below is an exothermic reaction? A) CO2 (g) → C (s) + O2 (g) AH° = 394…
A: Exothermic reactions are those reactions in which heat is released during a chemical reaction and…
Q: Methanol, CH3OHCH3OH , is formed from methane and water in a two‑step process. Step 1:Step…
A:
Q: The reaction: Zn + 2HCI ZnCl2 + H2 is an example of synthesis decomposition combustion single…
A: The element which comes above hydrogen in the reactivity series can displace hydrogen from acids.
Q: Data Table 1: NaHCO, Reaction Data Mass of evaporating dish (g) 47.19919 Mass of evaporating dish +…
A:
Q: Consider these reactions, where M represents a generic metal.…
A:
Q: Here's a one-batch sample of Just Lemons lemonade production. Determine the percent yield and amount…
A: Limiting reactant in a chemical reaction is that reactant which is entirely consumed during the…
Q: Methane is an odorless, colorless, flammable, greenhouse gas. It is used primarily as fuel to make…
A: This Is the reaction of Combustion reaction where CH4 is Burned in the presence of Air or say O2.…
Q: Use the following reaction to complete the statement: 1 N2 + 3H2 -> 2NH3 1.5 moles of nitrogen…
A: Moles of N2 = 1.5 Moles of H2 and NH3 =?
Q: Consider the following unbalanced chemical reaction: Al (s) + HCI (g) → AICI3 (g) + H2 (g)
A:
Q: Use the given information to determine the enthalpy of the reaction
A:
Q: mass in grams of phosphorus trichloride
A:
Q: Making Calcium Sulfate: MgSO4 (aq) + CaCl2 (aq) -------> CaSO4 (s) + MgCl2 (aq) Given Data: mass…
A:
Q: 4. The reaction: 4 Al (s) + 3 02 (g) → 2 Al203 (s) ; AH° = -3351 kJ Is and therefore heat is by the…
A:
Q: Classify the following reaction • NaCl + AgF → NaF + AgCI O Synthesis (composition) reaction O…
A: Since we know that by Double Replacement Reaction , AB + CD → AD + CB
Q: How many moles of each reactant were used in the reaction? (can you explain how to do it) Equation:…
A: Number of moles of reactent and Number of moles of products formed, Is totally depends on Balanced…
Q: Consider the following graph of the reaction 2 SO3(g) 2 2 SO2(g) + O2(g) AH = +197.8 kJ 60 503 502…
A: Actually when the volume of the system is decreased, the concentration of all the gas increases…
Q: How much energy in kJ is required to decompose 765 g of PCI3, according to the reaction below?…
A: Given that, 4PCl3 (g) →P4 (g) + 6Cl2 (g) , ΔH°rxn = +1207 kJ i.e, 4mol PCl3 (g) →1 mol P4 (g) + 6…
Q: How many reactants are present in this chemical reaction? BaS --> Ba + S
A:
Q: How much energy in kJ is required to decompose 765 g of PC13, according to the reaction below?…
A: When standard enthalpy change of a reaction is given, energy required for decomposition of a…
Q: Calculate the amount (mol) of sucrose that reacted. Molar mass of sucrose = 342.3 g/mol ΔT = 2.14…
A: NOTE: Since there is no reaction given in the question, hence based on the type of the data given in…
Q: Consider these reactions: Reaction 1: H2(g) + Cl2(g) 2HC1(g) AH = –184.6 kJ Reaction 2: 20F2(g) →…
A: In given question we have three reactions and we need to tell which one of these reaction is…
Q: What are the significant concepts in chemical reaction? What are the major takeaways?
A: The significant concepts in chemical reaction - Chemical reactions involve breaking chemical bonds…
Q: Reaction Type:_ At completion of reactions: Grams of methane Grams of oxygen Grams of carbon dioxide…
A: We need to write the mass of reactants and product after completion of reaction.
Q: U. Consider the unbalanced equation for the double displacement of aqueous solutions of barium…
A: Limiting reagent : It is the reactant which consumes firstly during the reaction. The amount of…
Q: 2. The calculated amount of product that should be produced based on the amounts of reactants is…
A: 2. C) Theoretical yield. The theoretical yield is defined as the calculated maximum amount of the…
Q: Consider the following balanced reaction. What mass (in g) of CO2 can be formed from 288 mg of O2?…
A: Given :- C3H7SH(l) + 6O2(g) → 3CO2(g) + SO2(g) + 4H2O(g) mass of O2 = 288 mg or 288 × 10-3 g To…
Q: Assume that combustion of C6H8O6 is a complete reaction. So, which of the followings show the…
A: Balanced chemical reaction : A reaction us called balanced when the atoms of all the elements in the…
Q: Given the data in the table below, determine ΔH°rxnfor the reaction 4NO (g) + 6 H2O…
A: In this question, we will determine the value of ∆H° reaction. We know the formula, product enthalpy…
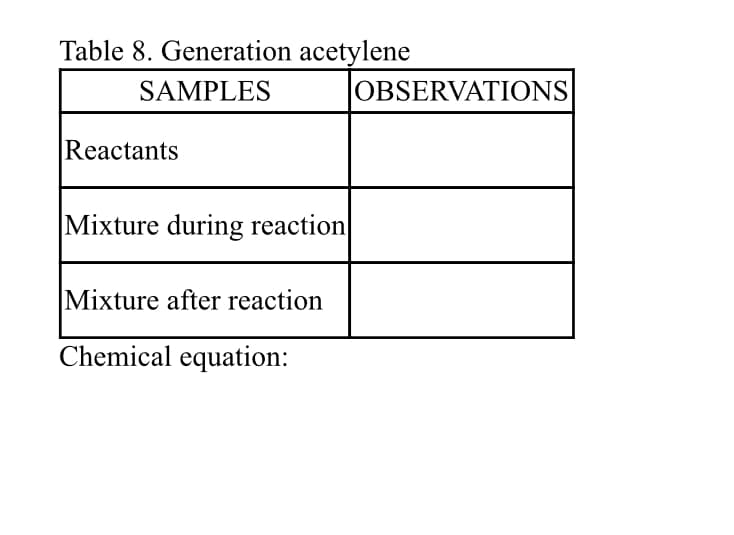
complete the table
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Which of the following statement are related to the reaction below? i. It is a process involve covalent bonds ii. Peptide bond links carbon of acid group to nitrogen of amine group iii. A typical organic molecule in this reaction are a basic amino group and an acidic carboxyl group iv. Consume water when each molecule held together by intermolecular bondsdrow the reaction..D. OXIDATION OF ALCOHOLS1. Place 1 mL of each of the following alcohols in separate clean test tubes: ethanol,benzyl alcohol and ethylene glycol.2. To each alcohol sample, add 2 mL of acidified potassium dichromate solution.Mix by shaking gently and observe any changes. Warm gently if necessary to startthe reaction.Record results in the table below.Alcohol Color changes observed Ethyl AlcoholBenzyl AlcoholGlycerol Equations for the oxidation of each alcohol:a. _________________________________________________________________b. _________________________________________________________________c. _________________________________________________________________E. FORMATION OF ESTERS1. To 2 mL of ethyl alcohol, add 1 mL of glacial acetic acid and 2 drops ofconcentrated sulfuric acid (CAUTION). Warm gently, then pour into a beakercontaining 20 mL of ice water previously made alkaline with sodium carbonatesolution. Stir well and note the…
- Some sulfuric acid is spilled on a lab bench. You can neutralizethe acid by sprinkling sodium bicarbonate on it and thenmopping up the resulting solution. The sodium bicarbonatereacts with sulfuric acid according to:2 NaHCO31s2 + H2SO41aq2¡Na2SO41aq2 +2 H2O1l2 + 2 CO21g2Sodium bicarbonate is added until the fizzing due to the formationof CO21g2 stops. If 27 mL of 6.0 M H2SO4 was spilled,what is the minimum mass of NaHCO3 that must be addedto the spill to neutralize the acid?Differentiate Precipitation and Agglutination reactions based on: Time duration of the procedure. Reactants involved. EXPLAIN EACH PLEASE THANK YOUExplain the meaning of “the reaction goes to less than 100% completion”
- Reaction of interest : S2O82-(aq) + 3I- (aq)→ 2SO42-(aq) + I3-(aq). rate= k[S2O82-]1[I-]1.Zn│Zn2+ (1.0 M)║Cu2+ (0.10 M)│Cu Ecell measurement .698 Reaction quotient, Ecell calculationThe reaction A(aq) + B(aq) C(aq) + D(aq) has a ΔHrxn = -85 kJ/mol. This reaction is endorthermic or exothermic . If the temperature of the reaction mixture is increased, equilibrium will shift towards the reactans or products. This change will decrease or increase the concentration of the reactants and K will increase or deacrease
- (a) What are the units usually used to express the rates ofreactions occurring in solution? (b) As the temperatureincreases, does the reaction rate increase or decrease?(c) As a reaction proceeds, does the instantaneous reactionrate increase or decrease?For the reactions: 2CH4(g)⇌C2H2(g)+3H2(g)2CH4(g)⇌C2H2(g)+3H2(g) ; Kc =0.170 mol2 L−2 at 1800 ∘C N2(g)+3H2(g)⇌2NH3(g)N2(g)+3H2(g)⇌2NH3(g); Kp =4.35×10−3 bar−2 at 291 ∘C∘C . Find Kp and Kc respectively for the reactions above.PbCrO4 is a common pigment called chrome yellow (or school bus yellow). It has a solubility of 1.71 x 10-4 g/L. The solubility product of PbCrO4, Ksp = ________. Enter the result in scientific notation to 1 decimal. e.g. enter 5.6x10-5 as 5.6E-5.

