Q: What product is formed when attached compound is treated with either Ag2O, NH4OH or Na2Cr2O7, H2SO4,...
A:
Q: Show complete solution. Round everything to 4 decimal places. Box all final answers Ethylene gas (25...
A: The chemical change involves a change in the bond energies, as some bonds are formed and some are ...
Q: Explain Retro Diels–Alder Reaction ?
A: To study the Retro Diels -alder reaction
Q: Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes. What is t...
A: Given Citric acid is responsible for the tartness of citrus fruits, especially lemons and limes. Str...
Q: 1.0L
A: “Since you have asked multiple questions, we will solve the first question for you. If youwant any s...
Q: What is the oxidation state of an element, like Cu or F2? O-1 02
A:
Q: 7. What mass of nitrogen is required to react with 16 grams of oxygen? 2N2+ 502 --> 2N205 (Hint: 3 s...
A: as per Q&A guidelines of portal I solve first question because it comes under multiple question ...
Q: What is the correct name for the structure shown below? Select one: O a. N-ethyl-2,3-dimethyl-2-hexa...
A:
Q: 6. No element exists with mass number larger than about 300. Explain Why?
A:
Q: Which metal is least reactive based on the activity series?
A: The least reactive metal based on the activity series is Cu. In the activity series the metals are...
Q: 13B.1(a) The line marked A in Fig. 13.3 is the fluorescence spectr benzophenone in solid solution in...
A: 13B.1(a) This problem belongs to the vibrational spectroscopy of the heteronuclear diatomic molecule...
Q: Explain the Dehydration of Alcohols to Alkenes ?
A: Alcohol gives alkenes by the removal of the water molecule has to given below,
Q: Perform the calculation and round the answer to the correct number of significant figures. 14.65...
A: Round the difference or sum to the same number of decimal places as the measurement with the fewest ...
Q: Draw the open structures of the molecules in the table below using the Lewis dot structure or VSEPR ...
A: [PtCl4]2- has a square planar geometry. The dipoles in [PtCl4]2- are directed in different directio...
Q: At 350 K, the rate of decomposition of a gaseous compound was 10.01 Pa s−1 when 10.0 per cent had re...
A: The decomposition reaction involves the breaking of larger molecules into smaller fragments. Example...
Q: If a glass of water and a lake have the same temperature, which has more thermal energy?
A:
Q: In former times, gas volume was used as a way to measure temperature using devices called gas thermo...
A: The ideal gas equation is PV = nRT P = pressure of gas V = volume of gas n = number of moles R = un...
Q: 18. Reacting 8.85 g of SiO2 with excess carbon produces 2.25g of SiC. What is the percent yield? SIO...
A:
Q: Discuss the dissolving soap in water ?
A: Soap is a mixture of sodium salts of fatty acids which are formed when fats or oils are saponified o...
Q: The attached reaction does not afford the major product that is given.Explain why this is so, and dr...
A: The alkyl halide is a tertiary alkyl halide, and the reagent is a strong base. The reaction is an el...
Q: If you could answer number 1 and explain it piece by piece tat would be great!
A: The reaction given is Fe(s)+S(I)→FeS(s) from the above balanced reaction , it is clear that 1 mol F...
Q: Draw curved arrows for the following acid base reaction. Using the pKa values, predict the direction...
A: Acidity can be best explained using pKa values which is inversely proportionate to the acidity. Thu...
Q: A hydrogen bond can form between adjacent to each other. a. two hydrogen atoms b. two oxygen atoms c...
A: a. Two hydrogen atoms share one electron resulting in the formation of a single covalent bond. There...
Q: Complete the reactions given below
A: The Diels-Alder reaction is an important organic chemical reaction where the reactants include a sub...
Q: How many molecules of oxygen are found in 3.00 moles of oxygen?
A:
Q: Question attached
A: An eclipse conformation infers that the substituents attached to two adjacent atoms are so close tha...
Q: describe the properties of transition metals that make them important as biomolecules
A: Transition metals involve 'd' block elements. They play an important role in biological processes. T...
Q: The pressure above a pure sample of solid Substance X at - 184. °C is raised. At what pressure will ...
A: The given temperature is (-184 + 273.15)K = 89.15 K
Q: A 10.0 mL sample of CO2 gas is enclosed in a gas-tight syringe at 22 oC. If the syringe is immersed...
A: Charle's Law states that volume of a gas is directly proportional to the absolute temperature at con...
Q: Plz do Asap...!
A: Given that, v=kAB The given stoichiometry reaction is given below. Let a and b be the initial concen...
Q: Why do most chemical reactions do not occur in a single step?
A: In a chemical reaction, the reactants react to form the products. For example, hydrogen and chlorine...
Q: Question 34 of 38 How many moles of precipitate will be formed when 33.4 mL of 0.300 M AGNO3 is reac...
A: The reaction is 2AgNO3(aq)+CaI2(aq)→2AgI(s)+Ca(NO3)2(aq) CaI2 is excess , so AgNO3 is limiting reage...
Q: H*/H,O Li C2H5NH2/-78°C b) 2-Pentin HBr/ROOR/hv c)
A: Hydration of alkene is done by treating alkene with H+/ H2O Internal alkyne can be converted to tra...
Q: In the reaction: 2H2O → 2H2 + O2 how many grams of O2 are formed from decomposing 18...
A:
Q: ODD-ONE OUT: Considering possible isomerism, choose which one is not part of the group.
A: square planner complexes: four atoms positioned at the corners of a square on the same plane around ...
Q: Draw both enantiomers for each biologically active compound.
A: enantiomer is one of two stereoisomers that are mirror images of each other that are non-superposabl...
Q: 3. Consider the Pourbaix diagram of copper a Identify the immunity, passivation, and corrosion areas...
A: (a) (b) Cu2+ + 4H+ + 2O2 → Co2O + 2H2O It is true because of slope line depends on both E and pH.
Q: Synthesize the following syntheses starting from the starting compounds using the necessary organic ...
A: Organic reaction mechanisms
Q: NO2 dimerises to form N2O4. Why?
A: In the formation of the chemical bond with the same or different elements, all elements tend to comp...
Q: these 2?
A: “Since you have asked multiple question, we will solve the first question for you. If you want any s...
Q: Explain nuclear magnetic resonance spectroscopy ?
A: SPIN AND MAGNETIC PROPERTIES THE NUCLEUS CONSIST OF ELEMENTARY PARTICLES CALLED NEUTRONS AND PROTONS...
Q: How does the density of the gas depend on the molar mass of the gas?
A: Number of moles is any substance can be determined from its given mass and molar mass No.of molesn=M...
Q: suppose you accidentally drop a glass bowl full of sugar onto the floor, causing the bowl to shatter...
A:
Q: How do you draw an ionic Lewis structure?
A: There are many trends in inorganic chemistry. These trends are atomic size, reactivity, metallic cha...
Q: Synthesize the following yntheses storling from the istorting compounds. using necessory organic od ...
A:
Q: Draw a structural formula for the product of the reaction of acetophenone with reagent Q.NaBH4 in CH...
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic addit...
Q: Write a molecular equation for the precipitation reaction that occurs (if any) when each pair of aqu...
A: In general, the reactants reacts to give the products. When two solutions are mixed with each other ...
Q: 2) The first law states that the energy of the universe is a constant (AU = q + w) while the second ...
A:
Q: Is it possible to use U-238 in chain reaction?
A: Uranium-238 is also denoted as U-238 or 238U. It is the most common uranium isotope found in nature....
Q: The fluoride ion reacts with water to produce HF. Will a concentrated solution of NaF in water be ac...
A:
Give the IUPAC name for attached compound.
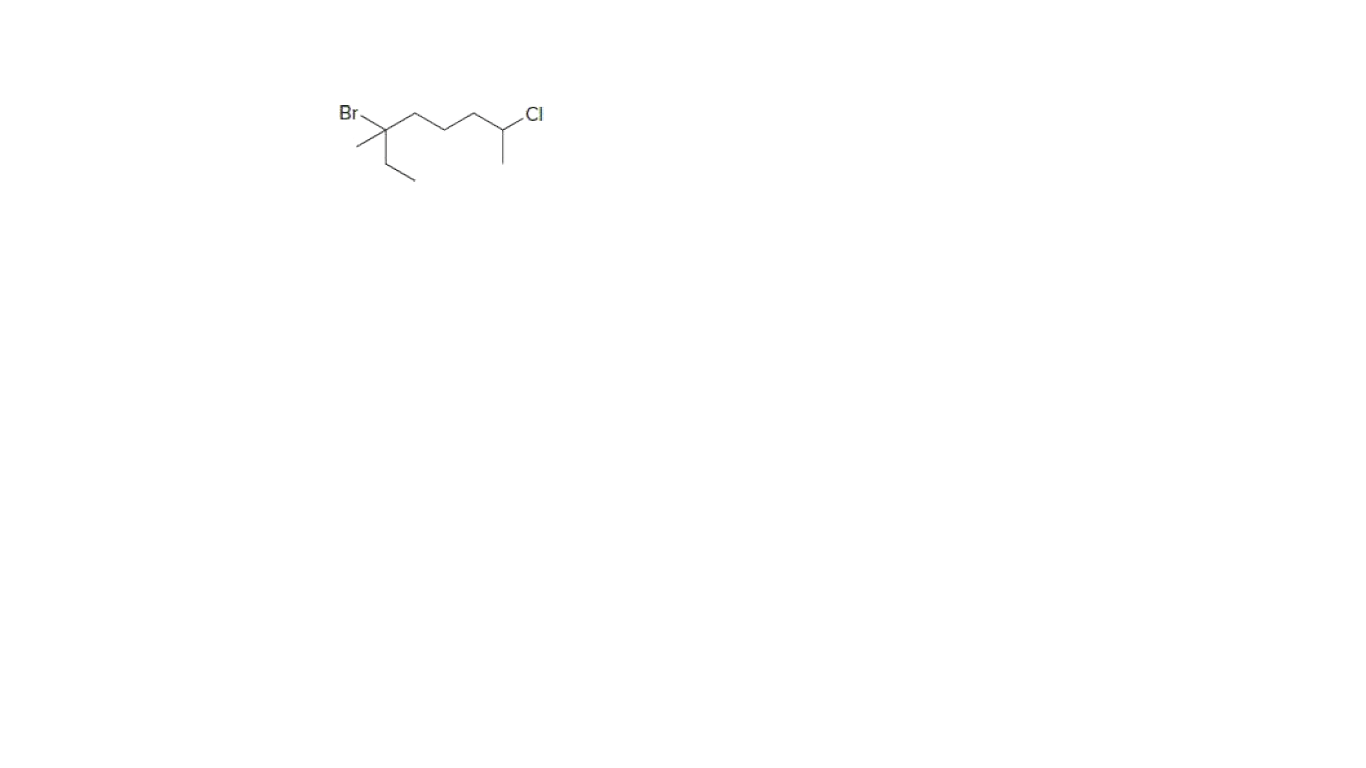
Step by step
Solved in 2 steps with 1 images


