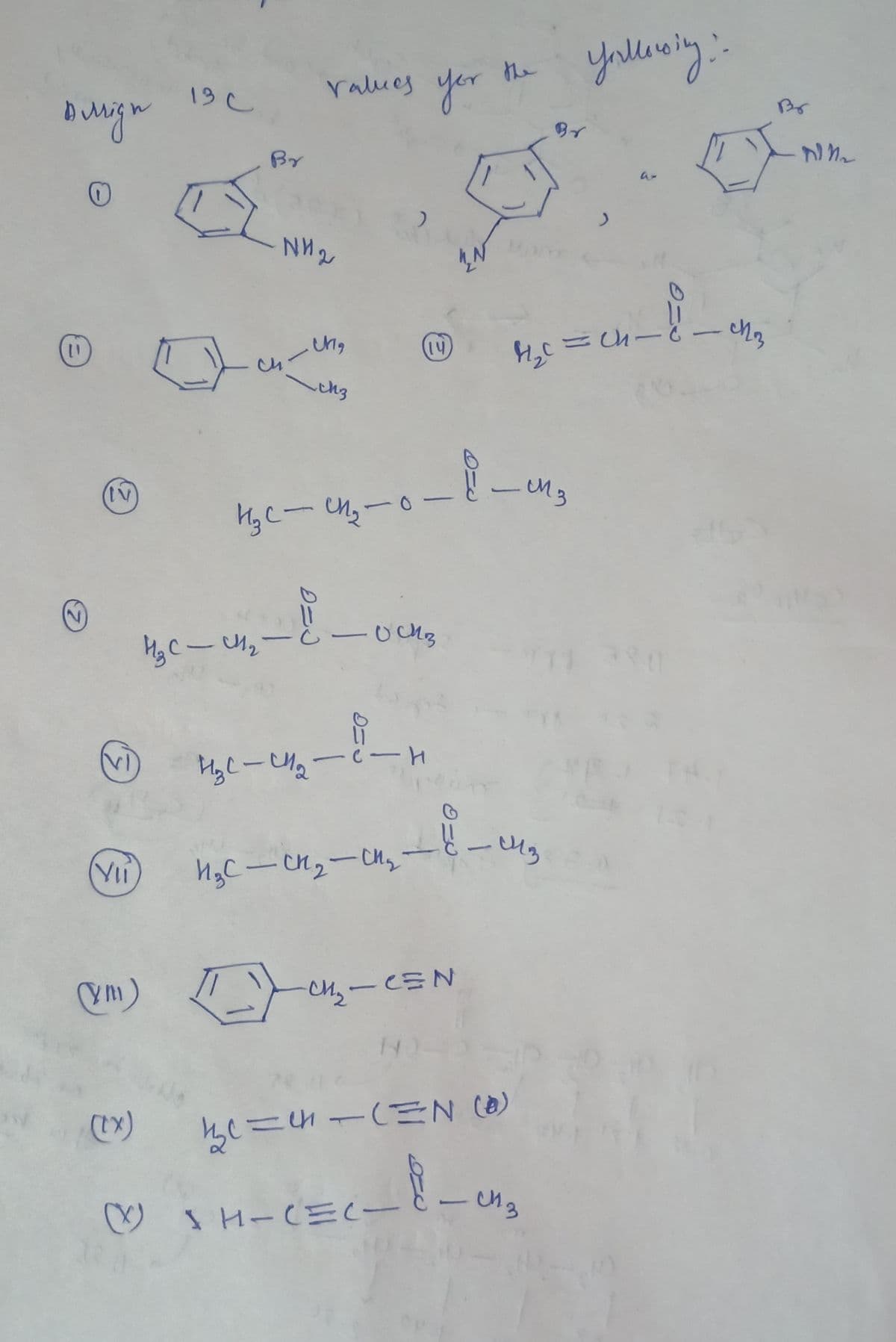
Q: Hge-Ua-ーH (Yi HgC-rュ-u,-と-u。 Ym) -CEN | 3.
A: Compound 1 Compound - 2 Compound 3
Q: 4. For the compound below, indicate the most probable multiplicity profile (ie, singlet, doublet,…
A: We have to predict the multiplicity of the protons.
Q: assign the NMR spectra of the compound diphenyl tertiary alcohol. including chemical shift
A: Protons near to alcohol group experience more downfield. Therefore a₁ a2 a3 and a will experience…
Q: Loudon | Parise SEVENTH EDITION presented by Macmillan Learning C,H,0, has IR absorptions of…
A:
Q: T D M M PPM M D 6H D 2H 1H PPM Lon
A: Spectrum 1 : 1,3-dibromo butane. Spectrum 2 : Isobutyl chloride.
Q: 2. The following spectra for A and B correspond to two structural isomers with a molecular formula…
A: A singlet at δ1.16 in spectrum A disappears when the sample is shaken with D2O. This shows that that…
Q: 6. The MS andđ 13C NMR spectra for two constitutional İsomers with 4 degrees of unsaturation are…
A:
Q: 1H NMR (The blue numbers represent integration) 8. 6 5 4 3 2 1. HSP-40-179 ppm 13C NMR 200 180 160…
A:
Q: Consider the data for an unknown compound with the molecular formula C,H002- • IR: 2400-3200 cm-,…
A: Molecular formula – C9H10O3
Q: 13 12 11 10 4
A: More deshielded protons are coming at higher ppm value in 1H-NMR spectrum.
Q: Compound A: C5H12O Vmax ¹H NMR 13C NMR Compound B: C6H11 NO Vmax ¹H NMR 3330s, 2950s cm-1 8 0.95…
A: Please find the attachment.
Q: The spectra (IR, 'H NMR, "C NMR and MS) of an unknown compound are shown in the following spetra.…
A: Note : δ 167 belongs to carbonyl. δ 145 to 125 having four signal corresponds to benzene ring. δ 61…
Q: ignore (solvent) 190 180 170 160 150 140 130 120 110 100 90 80 70 60 50 40 30 20 190.20 159.87 136…
A:
Q: For the molecules below how many signals for an 1H and 13C NMR will show?
A:
Q: The data for four 13C NMR spectra are given below. Match each spectrum with one of the following…
A: In 13 C NMR , Number of signals are equal to number of chemically non-equivalent carbons.
Q: I. Identify the structure best consistent with the data and II. Justify your choice in 1-2 short…
A: I.C7H16O (DBE = 7-16/2+1 = 8-8 = 0). DBE is o. Therefore epoxide is not required compound. II. The…
Q: 2) Propose a structure for C4H100. The 1H NMR data is as follows [splitting, chemical shift…
A: Firstly, calculate the double bond equivalent (D.BE) to know the number of pi bonds or rings. The…
Q: Predict the proton spectrum chemical shift, coupling and integration of the following molecule?…
A: Since you have asked multiple questions, we will answer only first question. For the remaining…
Q: (c) 'H NMR spectrum; Iron and Chlorine are NMR inactive, 31P, I = ½ 100% abundant. PCI3 Cl.. ..H Fe…
A: Solution -
Q: What structure is consistent with these spectra? 12 10 6 PPM 2 200 180 160 140 120 100 80 60 40 20…
A: Every nuclei will have spin and if an external magnetic field is applied, there will be an energy…
Q: 13C NMR Spectrum Resolves into two signals at higher field (50.0 MHz, CDCI, solution) DEPT C3H1,Br…
A: The molecular formula of the Organic compound is C9H11Br. The double bond equivalent (DBE) can be…
Q: 1a. Do the chemical shifts of the benzene protons in the iodinated product confirm the structure…
A: In 1H NMR (nuclear magnetic resonance) spectrum protons having different chemical environments show…
Q: Draw all six isomers of formula C4H8 (including stereoisomers).(b) For each structure, show how many…
A: C4H8 has 4 carbon atoms in it and 8 hydrogen atoms. From the molecular formula we can see that it is…
Q: Draw the proton NMR spectrum you would expect for butan-2-one. How well do the proton chemical…
A: Given compound, Butan-2-one
Q: Consider the following 13C and DEPT NMR spectra: f g c d ab 13C-NMR DEPT 13C-NMR 180 60 200 120 100…
A: C13 NMR is an analytical technique which is based on the fact that nucleus C-13 isotope is…
Q: Use the available spectra to deduce the identity of an unknown compound. Relative integrations are…
A: NMR spectroscopy is a very important tool for the determination of the structure of the organic…
Q: Which is the most likely structure for this NMR data? It would be helpful to refer to Table 15.1 and…
A: Answer In given NMR data ans spilling of H is satisfied by the…
Q: 6. The MS and 13C NMR spectra for two constitutional Isomers with 4 degrees of unsaturation are…
A:
Q: Two isomers have NMR chemical shifts of 5.5 and 7.0. What would be the coalescence time in a 500 MHz…
A: The formula for the coalescence time is given as follows: k=1ζπ∆ν2=1ζ2.22∆ν=1ζ where Δν: (Difference…
Q: NMR Spectrum for Question 2: HAC-CH-CHộ- gHO- 16 14 12 10 8. 6. 4 2 -2 -4 ppm HSP-00-115 00
A: Given NMR spectrum, Given compound - CH3CH2CH2COOH
Q: a. Explain why the 1C-NMR of cis-1,2-dimethylcyclohexane shows only one methyl resonance even though…
A: (a) The 13 C-NMR of cis-1, 2-dimethylcyclohexane shows only one methyl resonance(peak) even though…
Q: (c) 'H NMR spectrum; Iron and Chlorine are NMR inactive, 31P, I = ½ 100% abundant. PCI3 Fe CI PCI,
A:
Q: For each set of spectral data below, draw the skeletal structure of the molecule that is consistent…
A:
Q: 1. Identify and interpret the main peak in the mass spectrum of cyclohexanol. 100 80 60 40 20 0.0…
A:
Q: Methylacetate 2. Predict IR signal and their values in the assigned molecule (Part B). Also predict…
A: Ester compound shows an characteristic carbonyl absorption at 1735 cm-1 and 1H-NMR spectrum gives an…
Q: The following are all isomers of C10H140. Match the most appropriate structure to the NMR spectra…
A:
Q: . For each set of spectral data, propose an acceptable structure that is consistent with the data.…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: The carbon NMR chemical shifts of diethylmethylamine, piperidine, propan-1-ol, andpropanal follow.…
A:
Q: B) Rank by number of signals in 13C NMR spectrum. The compound with least number of signals is 1…
A: Number of signals in 13C NMR = Number of sets of carbon with different electronic environment
Q: 4. Describe the 'H NMR signals (size, shape, shifl) and "C NMR signals (size and shift) that would…
A:
Q: 8. Show how you would distinguish among the following three compounds H. OCH3 HO, H3CO W H3CO X Y…
A:
Step by step
Solved in 2 steps with 3 images

- Allenes are compounds with adjacent carbon-carbon double bonds. Many allenes are chiral, even though they don’t contain chirality centers. Mycomycin, for example, a naturally occurring antibiotic isolated from the bacterium Nocardia acidophilus, is chiral and has [α]D = -130. Explain why mycomycin is chiral.Assign R or S configuartion to the following molecules.c-e only
- Which of the following is not a monodentate ligand ? Select one: a. OH- b. H2NCH2CH2NH2 c. H2O d. CN-Org. Chem. 1. EA26. Can you please draw out all four , and explain what a stereoisomer is in relation ? ThanksQ- e acth _Jigg cintion con® Xowk Qov chlovuns 2 acid Ml o 25 (940210 calculote Ao Concendiekim of Mx TP Mo Wi concenAvation of geidig O, es M
- 32. Which of the following is INCORRECT? A. CH3COOH H+ + CH3COO- B. HF H+ + F- C. H2O H+ + O2- D. C2HO4- H+ + C2O42-Draw the structure of TRIPHat pH 5. Show the stereochemistry of each Calpha chiral center.The following reaction has a ΔSsystem < 0 O2(g) → 2O(g) T or F can you explain why this is false?
- Draw examples of the following: (a) A meso compound with the formula C8H18 (b) A meso compound with the formula C9H20 (c) A compound with two chirality centers, one R and the otherOne of the following molecules (a)–(d) is d-erythrose 4-phosphate, an intermediate in the Calvin photosynthetic cycle by which plants incorporate CO2 into carbohydrates. If d-erythrose 4-phosphate has R stereochemistry at both chirality centers, which of the structures is it? Which of the remaining three structures is the enantiomer of d-erythrose 4-phosphate, and which are diastereomers?Identify A–F in the following reactions.

