Q: a) -СCH-CH-OH,-Br,-H, -CH-CHз b) -СООН, СООCH3,-CH-ОН,-ОН с) -СH-NHCH3,-CH-NHz-NHz,-CN
A: Answer:- This question is solved by using the simple concept of determination of priority order…
Q: Each of the following reactions has been reported in the chemical literature and proceeds in good…
A: Note: Since you have posted a question with multiple subparts, we will solve the first three sub…
Q: Na a) 2 CH3-CH2CI 1) NaNH2 b) CH3-CECH 2)CH3CH,CI Br 1) ? c) CH3-CECH CH3-CH2-CH Br 2) ? d) HgSO,…
A:
Q: H2N-CHC-OH CH2 H2N-CHC-OH CHOH CH3 H2N-CHC-OH CH2 HN- H2N-CHC-OH CH2 OH
A: Amino acids:- These are basically the building blocks of protein. They contain an acid(COOH)…
Q: CH;-CH,-CH, CH,-CH3 1. CH3-CH-CH,- сH - CH, CH3 CH3 CH3 2. CH,- с - сH,-CH - CH-CH, CH3
A: Write the IUPAC NAME of the given structure-
Q: Classify each monosaccharide by the type of carbonyl group and the number of carbons in the chain.
A: Based on the type of functional group, carbohydrates are classified into two categories: Aldoses:…
Q: Draw the products formed in each reaction.
A: Note: Since you have posted a question with multiple subparts, we will solve the first three…
Q: 1. NaBH4 (е) CH,=CH-CH,--CH3 2. Но 1. (CH;),СHCH2-AlН-СH2CH(CH3)2, -78°C (f) -CH2-C-OCH3 2. H.О
A:
Q: OH CH3 II CH2CH3 НО HO- -H- ČO,H ČO2H II IV CH3 CO2H H- -HO- Но -H-
A: Fisher
Q: nplete tihe reactio H20 HC=CH H*, Hg2+ Cu propanol 250 °C KMNO4 C=C-CH3 нн H*/ t°C NABH4 H3c-C…
A: The detail solution for this reactions is provided below in attach image.
Q: Question attached
A: In the presence of strong acids, such as sulfuric acid and heat, alcohol dehydrates to an alkene.…
Q: HgSO4 H2SO4, H2O 1T (1) Br Вн, THF H,Oг, NaOH 10 NANH2 H =H 1S NH3 H20 HCI (1 equiv.) diethyl ether…
A: NaNH2 abstracts the acidic hydrogen from alkyne which reacts with alkyl halide to gives alkyne (1S).…
Q: Br Br- -CH-CH;, НО -H ОН CH2CH3 ŅH2 CH CH3 F- -CH; NH2 Cl キ, F НF Cl- -H H2N. Cl H2N- -CH3 НС Н H
A: Explanation- 1) Disteriomers- having different configuration around the more than one carbon atom .…
Q: How are the Fischer projection formulas in each pair related to each other? Are they identical or…
A: Enantiomers are those organic compounds which are non superimposable mirror images of each other.…
Q: HO- CH2CHCH,-OH |CH;CHCH,–OH CH,CH2CH2 –OH CH,CH2OCH3 OH OH
A: surface tension depends on intermolecular force of attraction between compound. as the…
Q: CHCH2),CH3 NaOH (CH2),CH3 CH;OH CH3(CH2) OCH3 OH CH,(CH),CH, OCH,
A: Trans-esterification:- This process helps in formation of esters and glycerol from the reaction of…
Q: а. CH3CH,OH `CO2H H2SO4 .CO2H b. `OH H2SO4 CO2H NaOCH3 С.
A:
Q: CH,CH,OH :0: :0: || Но — С — CH,— СH, — С — ОН :0: - - CH,-S– CH, H,0 KCl Cl4
A:
Q: CH, — CH —— сн, — сн, — ОН - сосн, CH3 CH, — с CH, — сONHCH, — сн, C1 CH; CH, - CH — с — соосH, - -…
A:
Q: CH,-C-0-CH, + Figure 39 t (s) II CH-C-CI + A CH-NH2 CH-C-NH-CH, + HCI H* B CH-C-OH + CH-OH H20 он CI…
A: A nucleophile is a nucleus loving species. An electrophile is an electron loving species. A good…
Q: Draw the products of each Sharpless epoxidation.
A: Sharpless asymmetric epoxidation: Titanium-catalyzed asymmetric epoxidation of primary allylic…
Q: NH₂ OH b. d. CHO
A: If the highest priority are on the same side of alkene then we called it as Z and if they are on…
Q: Label each stereogenic center as R or S.
A: Given,
Q: OH CH,CHCH,CC=CH CH, CH3 Çi C1 CH 3-CH-CH 2-CH-C-CH-CH 2-CH2-OH tH-CH, C1 CH3-CH,-CH;-CH2-NH,
A:
Q: k H3C-CH2-CH2-C-CH2-ċ-CH3 IUPAC 1. OH IUPAC OH Common Name OH H3C-CH2-CH-CH2-C-H m. IUPAC OH…
A: Applying concept of IUPAC nomenclature of organic molecules we can determine IUPAC nomenclature of…
Q: HC. N. NH aHC 'N' N. H-C-H H- OH H- -OH H- OH NH2 H- H. N 9- -0-CH2 но OH
A: Oxidoreductases - these are a class of enzymes that catalyze oxidoreduction or redox reactions .…
Q: CHO HNO3 1 H FOH Н—он NH2NHPH 2 -OH OH CH2OH NABH4 3
A: HNO3 is a strong oxidizing agent and NaBH4 is a reducing agent.
Q: HNO3 HCI H3CO -C(CH3)3 ? H2SO4 Fe or Sn HS A) H2N- -C(CH3)3 B) H3CO- -C(CH3)3 C) H2N D) H2N H3CO-…
A: Nitration is a process in which a nitro group is introduced into an organic compound. A mixture of…
Q: What are the products of the following reactions?
A: Gilman Reagent or Organolithiumcuprates: This is prepared by the reaction of alkyl halide with the…
Q: d.-COOH, -CH, OH, -H, -CHO e-CI, -CH,.-SH, -OH (-C=CH, -CH(CH,)2. -CH2CH3. -CH-CH,
A: Given: D. -COOH, CH2OH,-H,-CHO E. -Cl, -CH3,-SH, -OH F. -CCH, -CH(CH3)2, -CH2CH3, -CH=CH2 To find:…
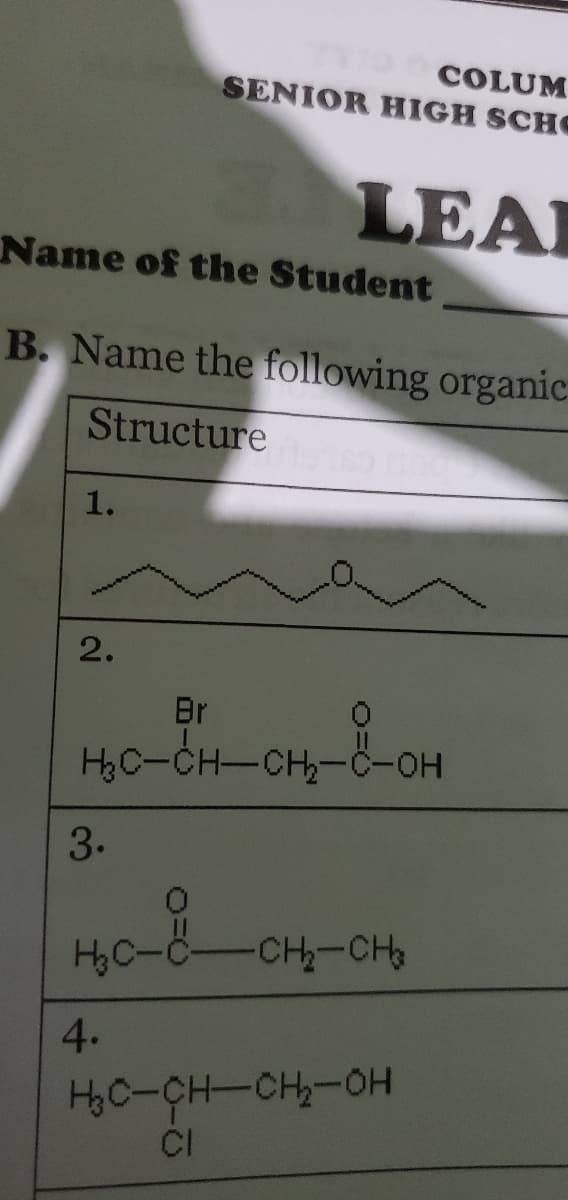
Q: Name the following compounds: 1. 2. Br Hc-CH-CH-ö-OH 3. -CH-CH 4. H,C-CH-CH-OH CI 5. HC-C=C-CH,-CH,…
A: According to our policy we should solve one question (If any subparts present then we should solve…
Q: H,C- -CH- -CH3 1 CH3 2 H;C- CH-CH ČH,-CH; H,C-CH,-CH,-ÇH-CH;-CH;-CH; 3 ČH;-CH3
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: е. d. 2 eq. HBr H2O, H,SO4 HgSO4 H, BH3 Pd/BaSO, H,O2, OH Li conc. KMNO, NH3 H,O H2 O3, -78 °C Pd/C…
A: The answer is given as follows
Q: Of the possible products shown for the following reaction, are there any that will not be formed
A: The first product in the above pic will not form in the reaction as we have change in the…
Q: O CH3 CH3 CH,0° CH,OH CH3 HCI (а) (b) CH;OH CH3 H3C H;C 8 CH3 CH;0° CH,OH CH3 HCI (d) CH,OH
A: SOLUTION: Step 1: Hello. Since the question contain multiple sub-parts, the first three are solved.…
Q: CH, - CH, - CH, - CH- NH - CH, - CH, - CH, 3. CH, - CH,
A: Interpretation- IUPAC name of these two compounds Concept- first compound is simple aliphatic…
Q: H HH 1 H-C-C-C-H Н Br H 2. CH;CH,CHCICH3 F 3.F. F Н Br H H 4. H-C-C-C-C-CI H H HH 5 CH3CC1,F CI CI…
A: Given that : We have to write the names of the following set of alkyl halides :
Q: 19 H3C- -CH, CI 20 Br Br HC Ec- -CH-CH-CH,-CH, 21 HC-C CH
A: The organic compounds are named according to the IUPAC nomenclature. It is done according to some…
Q: CH3 Brz/H,O A) CH3 CH3 1) BH/THF B) 2) H-О, NaOH CH3 CH3 CH3COOH C) CH3
A: Reaction is called bromination of alkenes. Solvent used is water. In this reaction solvent is very…
Q: CHO MF poc's CHO CHO what is The Me caníssumel Inclol) DMF. POCI
A: DMF is dimethylformamide. Detailed mechanism is given in step 2.
Q: CH,OH 38. CHCH,CH3 Br
A: We are authorised to solve only one question at a time. Please post rest of the questions…
Q: CH3 Но CH3 CH3 CH3 -H- Но- - HO- H- OH -OH -H- CH3 H- OH H- -ОН Но- -H- CH3 CH3 CH3 I II III IV
A: Meso compounds contain chiral centers/stereogenic centre though they are optically inactive. Meso…
Q: 0-CH,-CH3 H. 0-CH3 CH3-C-0-CH3 CH3-CH2-0-CH,-OH 0-CH3 OH 0-CH2 CH3 HO-CH, CH,…
A: Alcohols add reversibly to aldehydes and ketones to form hemiacetals. This reaction can continue by…
Q: Give the complete IUPAC name for each of the following compounds:
A: IUPAC name of (a) Butan-2-ol IUPAC name of (b) 2-iodo-3,3-dimethylpentan-2-ol IUPAC name of (c)…
Q: H2N-CHO-OH CH2 H2N-CHC-OH CH2 CH2 H2N-CHC-OH H. C=0 NH2 CHa
A: Given: Different- different structure of amino acids. To find: Common name of amino acids. Solution:…
Q: COOCH3 OH Br делзеный дух ОН NH CI CH3 CH3
A:
Q: 3 ÇH3 CH3 CH3 CH3 CH,-c -H CH,-C-OH; CH;-C +:OH2 ČH; CH3-C CH;=C ČH3 ČH; CH; 2 CH3 CH;-C-CHCH, Hi…
A: In 2, there is no loss of H+, number of hydrogen in reactant and product are same.
Q: -CH2OH b. -CH,Br -CH(CH3)2 -CH3 -CH2CH,OH -CH¿OH -CH,CH,CH,Br а. -H -CH3 -CI -OH c. -CH,CH,Br
A: Priority is on the basis of atomic number of atoms as here in CH2OH ist atom is carbon next is 2…
Q: CH3 CICH,ÖCH,CH,CH(CH,), CH,CH,Č-CHCH, CH, Č,H, ČH2CI CH3 OH .CHO `CHO оно BRCH,CH CCH, CH,CH,CH,CHO
A: Given molecules are : Provide the IUPAC names for the following compounds = ?
Step by step
Solved in 2 steps

- Most naturally occurring amino acids have chiral centers (the asymmetric a carbon atoms)that are named (S) by the Cahn–Ingold–Prelog convention (Section 5-3). The commonnaturally occurring form of cysteine has a chiral center that is named (R), however.(a) What is the relationship between (R)-cysteine and (S)-alanine? Do they have the oppositethree-dimensional configuration (as the names might suggest) or the same configuration?(b) (S)-Alanine is an l-amino acid (Figure 24-2). Is (R)-cysteine a d-amino acid or anl-amino acid?(Thanks for the help if you can break down the steps and provide drawings(needed) during the answer since i am trying to see if i have solved this in the right way not copy :)) Draw the heme environment in oxymyoglobin and explain how the geometric data for the attached oxygen molecule may be understood. Fe O (bond length) = 1.809 Å; O-O (bond length ) = 1.246 Å; Fe - O - O (bond angle) = 123 \degmmole of H2SO4?
- 1. Show how to separate mixture of salt from oil. 2. Explain why alkanes are hydrophobic?Q.3: A polydisperse sample of PS is prepared by mixing three monodisperse samples in the following proportion: 1g (10,000) molecular weight, 2g (50,000) molecular weight and 2g (100,000) molecular weight. Calculate number average and weight average molecular weight.Name the following XeF6



