Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter14: Mass Spectrometry
Section: Chapter Questions
Problem 14.37P
Related questions
Question
100%
What functional group is at 763ppm?
For HNMR at about 2.3ppm is it a quintuplet?
How do I get the structure from this?
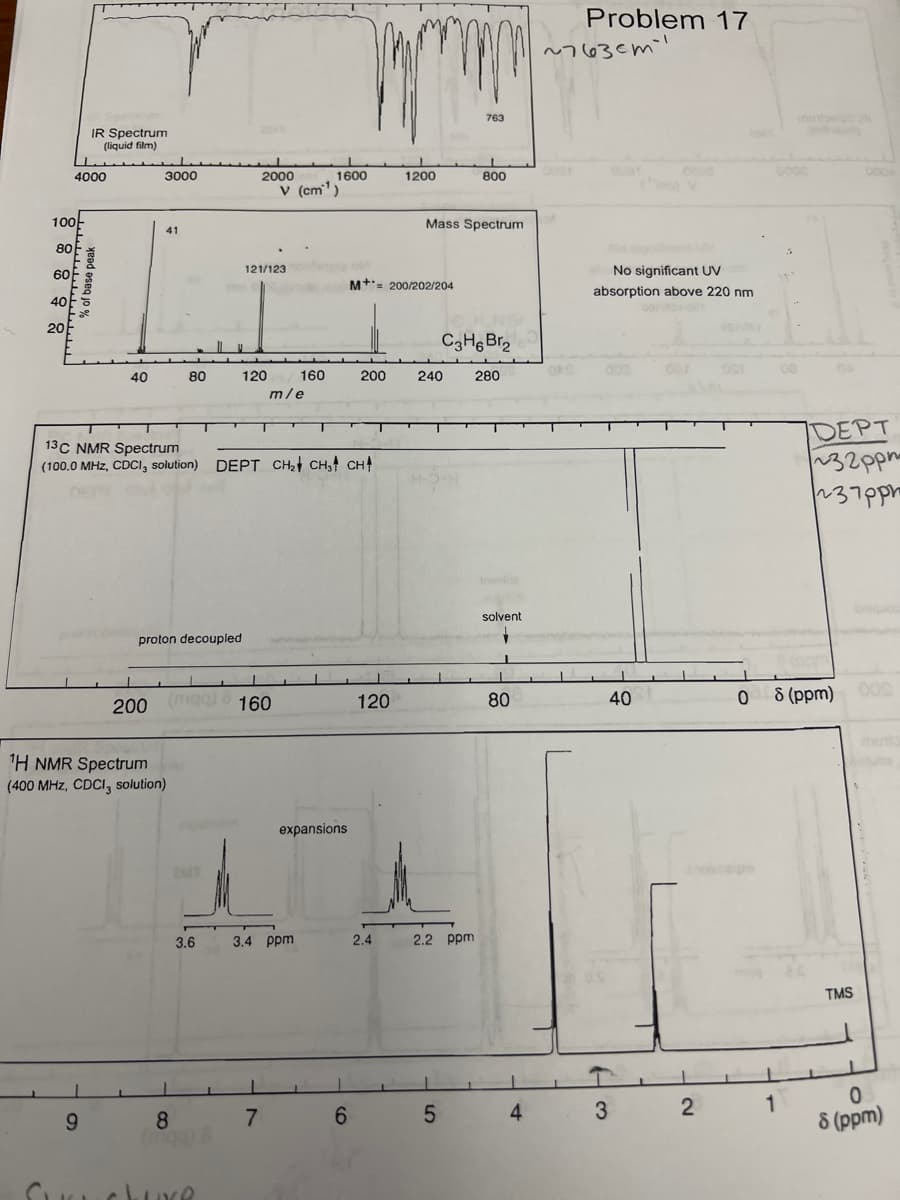
Transcribed Image Text:100k
80
60
40
20
4000
IR Spectrum
(liquid film)
% of base peak
9
40
3000
41
200
proton decoupled
¹H NMR Spectrum
(400 MHz, CDCI, solution)
80
8
3.6
2000
Suuchuxe
121/123
120
13C NMR Spectrum
(100.0 MHz, CDCI, solution) DEPT CH₂ CH3 CH
1
(mga) 160
v (cm¹)
7
m/e
160
1600
3.4 ppm
expansions
6
200
M+ 200/202/204
T
120
2.4
1200
Mass Spectrum
240
763
2.2 ppm
5
800
C3H6Br₂
280
solvent
✓
80
4
Problem 17
~7630m
OAS
No significant UV
absorption above 220 nm
13
40
2
0
1
8 (ppm) 009
20
000
DEPT
~32ppm
237ppm
TMS
0
8 (ppm)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning