Q: It a sample of benzoic acid (m.p. 121-122 degree C) was contaminated with a small amount of phenol…
A: All the necessary explanation has been given in the following step.
Q: Complete the table below for calculating the molar mass of the compound tetraphosphorus decaoxide.…
A: Given table is :
Q: Which of the following reactions will produce the a-amino acid? (Select all apply, ) NH, H2 COOH Pd…
A: Here we have to predict the reactions in which cases amino acids are formed.
Q: created x10 Is SOCI, being created or destroyed by the chemical reaction? destroyed neither created…
A: Given:
Q: Use the table below to answer the questions. In your own words, define half-life. b. Which isotope…
A: Since you have asked multiple questions, we will solve the first question for you as per our…
Q: 50 40 kcal/mol 30 20 - 10 course of rx No activation energy is required A. True B. False
A: We have to predict whether given Statement is correct or not.
Q: Consider the reaction described by the chemical equation shown. 3C,H, (g) → C,H,(1) AHan = -633.1 kJ…
A: ∆S°reaction = standard entropy of reaction = ∆S°product - ∆S°reactant ∆Sproduct = Standard molar…
Q: Calculate the ionic strength of a solution that is 0.15 mol kg-1 in KCI(aq) and 0.30 mol kg-1 in…
A: Given: Molality of KCl = 0.15 mol kg-1 Molality of CuSO4 = 0.30 mol kg-1
Q: 5. Draw the structures of a three-carbon chain structure with following functional groups. a. An…
A: There are different types of functional groups (groups of atoms that are responsible for the…
Q: An enzyme has an optimal pH of 7.1 and an optimal temperature at 37° C. Which statement describes a…
A:
Q: you react benzoic acid with a base, the resulting ion will be soluble in water. If you add HCl to…
A: The answer is as follows:
Q: Which of the following ions is the smallest? Ca2+ O Mg2+ O Sr2+ Re2+
A:
Q: Use this data to answer the following questions. Write the rate law for this reaction. rate = k O ロロ…
A: The rate law can be found in two ways- 1- graphical method 2- taking the concentration at different…
Q: estion 7 of 23 > Identify the chirality center(s) (sometimes called chiral atom) in the compound…
A: Identify chirality centre and number of stereoisomers ?
Q: 4. Calculate the heat that is transferred when 98.999g of metal is heated from 20.1°C to 125.0°C.…
A:
Q: Complete the table below for calculating the molar mass of the ionic compound chromium(II) fluoride.…
A: Calculation of molar mass of the chromium(II) fluoride: The molar mass of Cr2+ = 51.99 g/mol The…
Q: what happens when an amide reacts with water?
A:
Q: CO, (g) + H,O (1) CH,O, (aq) + 0, (g) Suppose that 10.0 g of CO, reacts with 7.00 g of H,0 to give…
A:
Q: Hydrolysis of an ester (in presence of acid catalyst) forms --- and ---. Question 16 options:…
A:
Q: The equilibrium constant Kc for the folowing reaction is 7.17 at 350 K 2XY(g) ---> Y2(g)+X2(g)…
A:
Q: The skeleton of the bicarbonate ion, HCO, , is shown here. Draw the complete structure. Add…
A: Formal charge = valence e- - unbonded e- - 1/2 bonded e-
Q: Name: ID: HOAC H OH C) AcO AcO- COAC HBr H OAC. Ag,0 NaOH, H20 HOH H NABH4 D) но HO он н он H H20 H…
A: An aqueous solution of Br2 when added to sugar. Br2 is mild oxidizing agent and easily oxidizes…
Q: Question 3 Calculate the final total borate concentration (in mM) in the buffer formed by mixing…
A: Here we have to find total borate concentration in buffer solution containing 0.1650L of 110 mM of…
Q: Name the following polypeptide, tising its abbreviated name. CH,SH CH,OH CH2 H,N-C-C-N-C-CN-C-C-OH…
A:
Q: What is the mass percent (m/m) of NaCl in a solution made by dissolving 5.16 g of NaCl in 79.9 g of…
A: Since, you have multiple questions so I will solved first question for you please post separately…
Q: 5. A 85.75g sample of a clear liquid is cooled from 68.3° to 32.1°C when it loses 5850 Joules of…
A: Specific heat capacity is amount of heat needed to increase the temperature of one gram of substance…
Q: 69 mL of.125 M solution of an acid HA is titrated with .125 m solution of NaOH calculate the PH at…
A:
Q: 3. Fill in the following chart. Sample Type of Matter (Element, Compound, Mixture) Compound Efement…
A: The samples given are Ocean water, Glucose (C6H12O6), Apple juice, Oxygen (O2), Blood, Water (H2O),…
Q: Question 4 of 23 > Monosaccharides Disaccharides Polysaccharides Answer Bank CH,OH CH OH HOCH2 OH…
A: Carbohydrates are optically active polyhydroxy aldehydes or ketones or such type of substances which…
Q: In the best Lewis structure of OCI2, around the oxygen there are and O 4 bonding pairs and zero lone…
A: Note: As per our guidelines, we are supposed to answer only one question when multiple questions are…
Q: Which of the following ions is the smallest? O Ca2+ O Mg2+ Sr2+
A:
Q: [References) Use the References to access important values if needed for this question. What volume…
A:
Q: For the following reaction, 0.262 moles of sulfur dioxide are mixed with 0.284 moles of water.…
A:
Q: Calculate the mass in grams of a metal Q ( Molar mass= 88.23 g/mol) that will be deposited from a…
A:
Q: Highest S Lowest S° Answer Bank Ne(g) HF(g) F, (g) Question Source: McQuarrie, Rock, And Gallogly 4e…
A:
Q: Which of the following is not a valid resonance structure for N3-? A) -NEN o ENEN-N] =N: B) N:…
A: Given structures are : Which of the following is not a valid resonance structure of N3- = ?
Q: 32. A solution of ammonia, NH3, has pH = 11.50. What is the ammonia concentration? (The pK of NH4*…
A:
Q: alculate the (a) electrical energy, (b) AG°, and (c) K of the reactio 1. Fe*2(aq) / Fe+ (aq) //…
A:
Q: Which of the compounds is more soluble in an acidic solution than in pure water? AgCl Cd(OH)2 CACO3…
A:
Q: The purity of the aspirin sample is tested with iron (III) chloride. A purple color resulting from…
A: Aspirin is acetyl salicylic acid. It is a white crystalline substance. Purity of aspirin is done by…
Q: 3. A gas mixture contains 50 mol% H2 and 50 mol% He. 1.00-L samples of this gas mixture are mixed…
A: #3: Volume parts of H2(g) in the mixture = 1.00 L * (50/100) = 0.50 L Since He(g) is an inert gas,…
Q: What will happen to the NO concentration at equilibrium if the [NO2] is raised NO2(g) NO(g) +…
A: The above problem is based is related to chemical equilibrium.
Q: Which of the following equations represent redox reactions? Which do not represent redox reactions?…
A: A reaction in which all the atoms have same oxidation number on both the sides of the reaction is…
Q: 2. The decomposition of N205 (2 N2O5 → 4 NO2 + O2) was carried out at several different…
A: Recall the given reaction, 2 N2O5 → 4 NO2 + O2We have to calculate the activation…
Q: In the best Lewis structure of OCI2 , around the oxygen there are and O 6 bonding pairs and 2 lone…
A: In this question we have to tell the bonding pair and the lone pair.
Q: For each resonance structure, use curved arrows to show the delocalization of electron pairs that…
A:
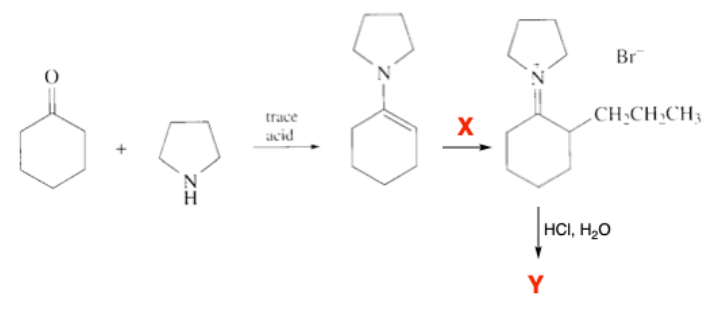
Q: what are the reagents? write out the mechanism
A:
Q: For given is the chemical equation and the data in the table below. Determine the Activation Energy…
A: We can solve this problem by using this equation K= A e-Ea/RT Where K= Rate constants A=…
Q: The ratio of carbon-14 to carbon-12 in a piece of wood discovered in a cave is R = 1/915. Estimate…
A:
Q: Consider the reaction. 2 A(g) B(g) Kp = 6.26 x 10-s at 500 K If a sample of A(g) at 2.90 atm is…
A:
detrmine the ff:
1. reagent x
2. product y
Step by step
Solved in 2 steps with 1 images


