Chapter14: Conjugated Compounds And Ultraviolet Spectroscopy
Section14.SE: Something Extra
Problem 41AP: Although the Diels–Alder reaction generally occurs between an electronrich diene and an...
Related questions
Question
2d.
Transcribed Image Text:10:03 1
Today
All Photos
10:02 AM
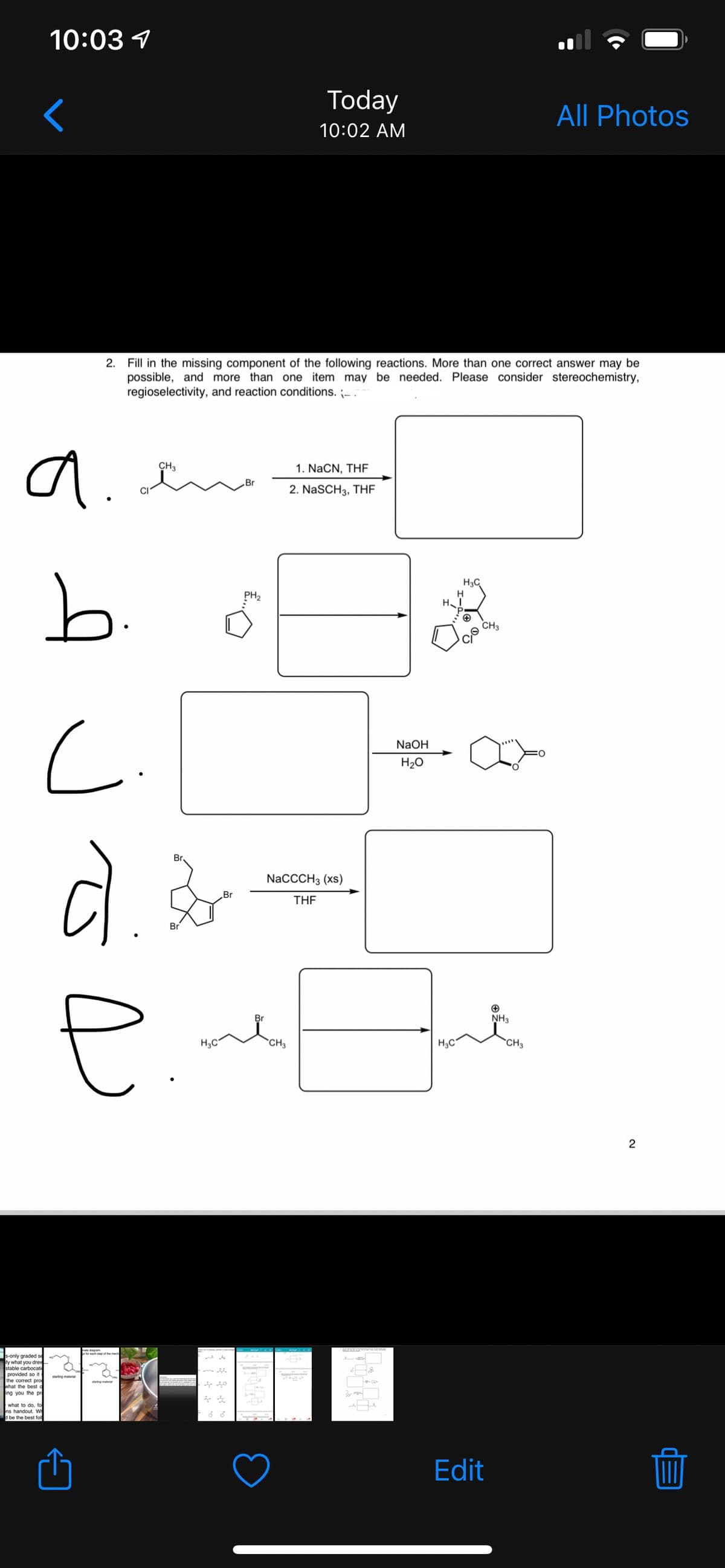
2.
Fill in the missing component of the following reactions. More than one correct answer may be
possible, and more than one item may be needed. Please consider stereochemistry,
regioselectivity, and reaction conditions. -.
a.
CH3
1. NaCN, THF
2. NaSCH3, THF
b.
H3C
PH2
H
CH3
NaOH
H20
Br
NaCCCH3 (xs)
Br
THE
Br
NH3
H3C"
CH3
H3C
*CH3
2
s-only graded se
ty what you drew
stable carbocati
provided so iti
the correct pro
what the best c
ing you the pr
ting mat
what to do, fo
ns handout. WH
be the best fol
Edit
** * * *
C.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

