Q: Identify the expected major product of the following reaction. Br2 ? hv Br Br Br Br. Br +En +En +En ...
A: The presence of light is the most favourable condition for the radical reaction to occur. If there a...
Q: Aprotic solvents contain -OH groups and can function as hydrogen bond donors. А True В False
A: Aprotic solvents are polar liquid compounds that have no dissociable hydrogen atoms. These solvents ...
Q: Calculate the pH for each case in the titration of 50.0 mL of 0.220 M HC1O(aq) with 0.220 M KOH(aq)....
A: Note - Since you have asked a question with multiple sub-parts, we will do the first three sub-parts...
Q: Calculate the pH of a solution formed by mixing 100.0 mL of 0.20 M HCIO with 200.0 mL of 0.30 M KCIO...
A: pH of the solution = 8.01
Q: All of the following are extended coverage perils under an HO-2, EXCEPT: A: Burglar damage.
A: All of the following are extended coverage perils under an HO-2, EXCEPT Burglar damage .burglary is ...
Q: 8. Balanced chemical equation for the formation of zinc chloride from zinc and HC1
A: Interpretation: We have tp balance the chemical reaction between Zn and HCl .
Q: c. The absorption data in the table below were collected for two complexes of Cr(III). From these da...
A: In both the complexes Cr is present in +3 oxidation state so both the complexes are d3 and octahedr...
Q: What must be the coefficient of O2 in this reaction 2 NaClO3 → 2 NaCl + ____O2 ?
A: According to the Law of conservation of mass " all atoms of different elements must be equal on both...
Q: H. CH3 HO.
A: Given structure is : Identify the chiral center and determine if it is the R- or S- stereoisomer = ...
Q: A) 30mL of a 0.2M solution of tripeptide Glu-Lys-His is adjusted to pH=9.5. The solution is then tit...
A:
Q: 2. Calculate the pH of a solution that contains 150 mL of 0.25 M NaF and 252 ml of 0.30 M HF. K, = 3...
A: Given volume of HF is 252 mL and strength is 0.30 M Given volume of NaF is 150 mL and strength is 0...
Q: How can you interpret the Rf value to study any molecule? Please shortly answer at your own words. ...
A:
Q: (References] An 18.6 mL volume of hydrochloric acid reacts completely with a solid sample of MgCOg. ...
A: Molarity of HCl can be calculated by using formula - Molarity = mole/volume(L) First of all calculat...
Q: Write the balanced net ionic equation for the reaction of aqueous sodium sulfide with aqueous copper...
A: Net ionic equation is writen using balanced molecular reaction.
Q: For each of the following species, determine the number of protons and the number of neutrons in the...
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: Using Faraday constant, how many grams of copper will attach to the cathode of an electrolysis cell ...
A: Given, Current = I = 12 mA Time period = t = 1.1 hour Ion of Cu in electrolytic solution = Cu2+ ...
Q: The melting point of calcium is 851 oC, its solid-state specific heat (Csolid) is 0.632 J/goC, and i...
A: To calculate the energy , we would thermochemistry relations . 1.) So calculate the energy required ...
Q: Draw all resonance structures for the nitrite ion , NO2-.
A: Rules for drawing resonance structure 1)Only the electrons move, the nuclei of atoms never move an...
Q: If 14. 8 mL of 0. 100 M NaOH solution are needed to react with 25. 0 mL of an unknown HCl s...
A: Given data is as follows: The volume of NaOH V1= 14.8 mL The molarity of NaOH M1= 0.100 M The volume...
Q: 21 October 2021 at 3:57 pm Chemistry practise a) Predict the products of a chemical reaction between...
A:
Q: Write the balanced equation for the neutralization reaction between H, SO, and KOH in aqueous soluti...
A: An acid and a base with react together to form water and a salt ,this type of reaction is called n...
Q: MacBook Air 9.4 Provide synthetic schemes for the following transformation. The only sources of carb...
A: We have to provide the synthesis for the following given reactions as follows in step 2:
Q: 0.150 mole of NaCl is dissolved into 246 grams of pure water. What is the mole fraction of NaCl? O 0...
A: there are two question,but student want to solve question-2, so i am solving question-2.
Q: Consider the equilibrium N2(g) + 3 H2(g) → 2 NH3(g) at a certain temperature. An equilibrium mixture...
A: Kc is the ratio of product of concentration of products raised to their stoichiometric coefficient ...
Q: Write a general statement about what happened to temperature when the microwave was turned on. 2 ...
A: 1. Temperature will increase. 2. Yes, the number of molecules make a difference in the change of te...
Q: Phosphorus in urine can be done by treating with molybdenum (VI) and then reducing the phosphomolybd...
A: To plot the corrected absorbance as a function of the concentration of P standard.
Q: Calculate the average atomic mass
A:
Q: What is the pH of a 0.380 M solution of NACN (Ka of HCN is 4.9 x 1010)?
A:
Q: Practise questions a) Predict the products for a reaction between sulfuric acid and magnesium hydrox...
A: Sulfuric Acid is H2SO4 Magnesium Hydroxide is Mg(OH)2 Hydrochloric Acid is HCl The molar mass of M...
Q: Draw the structure of the major organic product of the reaction. -NH2 CI H3C-
A: Acid chloride reacts with the amine to form an amide.
Q: Why is the protection of the amine function needed in this reaction?
A: Practical organic chemistry
Q: What is the pH of a 0.680 M solution of Ca(NO2)2 (Ka of HNO2 is 4.5 x 104)?
A:
Q: THE SEARCH FOR LIFE CONTINUES Scientists have identified two similar molecules as water-like candida...
A: A question based on general chemistry, which is to be accomplished.
Q: Make an electron-flow-mechanism for this synthetic scheme. This involves predicting major and by-pro...
A: It is an example of electrophilic aromatic substitution reaction
Q: Explain the concept of partial molar volume, and justify the remark that for a binary solution, the ...
A: We need to explain the concept of partial molar volume, and justify the remark that for a binary sol...
Q: Identify the oxidized substance, the reduced substance, the oxidizing agent, and the reducing agent ...
A: Oxidation :- The process of loss of electrons or increase in oxidation number is known as oxidation ...
Q: How many π bonds are there in 1,2-propadiene (H-C=C=C-H)?
A: π bond :- A chemical bond which is formed by partial or side wise overlapping of atomic orbitals of...
Q: Archaeologists used pieces of burned wood, or charcoal, found at the site to date prehistoric painti...
A: Given, half life of C-14 = 5730 years. Exponential decay equation : A(t) = Ao ekt Since half life is...
Q: To what temperature in K must a variable volume container of helium gas be cooled to obtain a final ...
A: We know that : V1/T1 = V2/T2 ...(1) (Charles' law) Here, V1 = initial volume = 2.86 L, T1 = initial...
Q: Heat Transfer and Calorimetry 4. A 10.0 g metal block is heated to 100.0 °C and then added to 20.0 g...
A: Given, Mass of sample metal = 10.0 g Mass of water in calorimeter = 20.0 g The initial temperature o...
Q: is when bodies of water contain abundance of nutrients caused by humans. O A outrophication OR therm...
A: Eutrofication is the excessive growth of algal blooms in water body due to excessive enrichment of t...
Q: 2. Copper has two stable isotopes with the following masses and % abundances: Cu-63 (62.93 amu, 69.0...
A: Calculation of average masses: As per our guideline we have to answer first question only.
Q: Please help explain how to find pKas of ionizable groups.
A: pKa's are experimentally determined values for the amino acid side chains, and is a defined literatu...
Q: what atomic or hybrid orbitals make up the pi bond between c2 and o in acetaldehyde, how many sigma ...
A:
Q: Show the step-by-step process. Do not use shortcut methods. Make it as detailed as it can be. ENCOD...
A: Chiral centre is that carbon which is attached to the four different groups. If any one group is sam...
Q: Question 12 of 28 Which atom in the C-Br bond has a partial negative charge (8-)? A) C B) Br C) Both...
A: Given is C-Br bond.
Q: Which precaution must be taken when working with bromine? Work under a well-ventilated hood ...
A: Wear a respirator.
Q: DRAW the structure given the IUPAC name 4-Bromo-4-ethyl-2-methylhexane
A:
Q: What is the IUPAC name for the following compound? CH;CH, CH3-CH,-CH-CH,-CH-CH-CH-CH; Select one: O ...
A: Given : Structure containing 2 Chlorine group. To find : IUPAC name
Q: Write the balanced equation for the neutralization reaction between H, SO, and KOH in aqueous soluti...
A: We have to write the balance equation for the neutralization reaction between sulfuric acid and pota...
IUPAC Names for
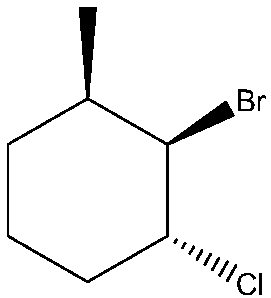
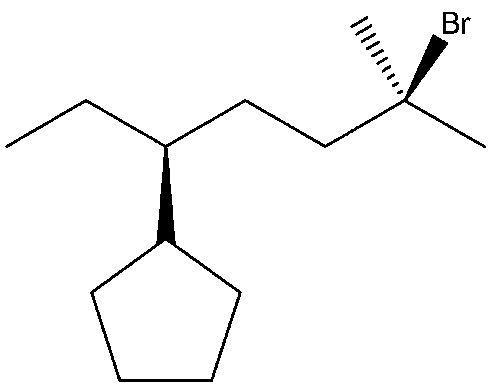
Step by step
Solved in 2 steps with 2 images


